Keywords
allylsilanes
asymmetric synthesis
bicyclic δ-lactones
bioactive compounds
cyclopentanoids
cyclopentenones
epoxidation
ion-exchange resins.
lactonization
silylation
Abstract
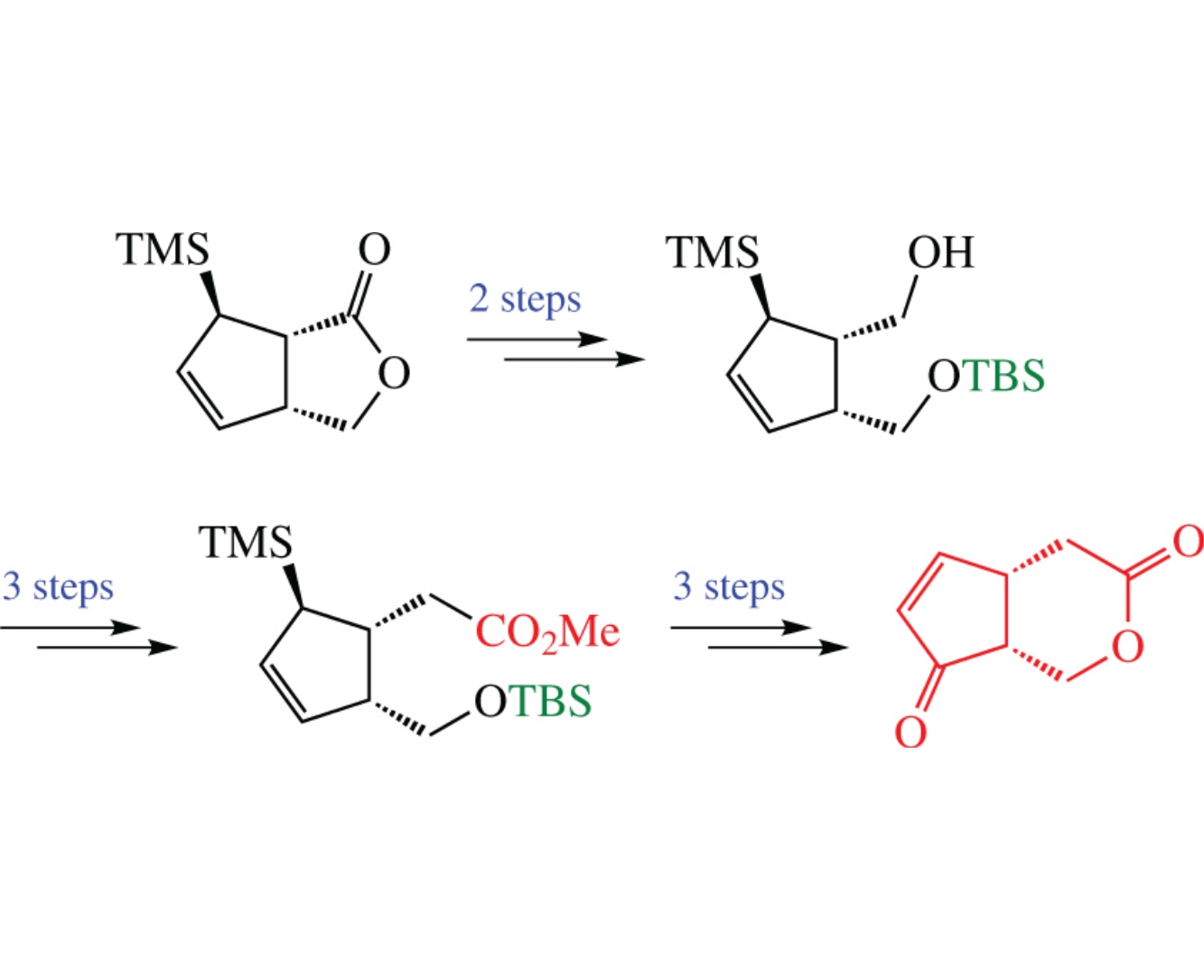
The title compound, as the new chiral building block for bioactive cyclopentenones, was prepared in 8 steps with 15% overall yield. The key steps involve selective homologation in intermediate [(1S,2R,5R)-5-trimethysilylcyclopent-3-ene- 1,2-diyl]dimethanol by regioselective silylation followed by oxidation and the Wittig reaction.
References
1.

OKADA M., EGOSHI S., UEDA M.
Bioscience, Biotechnology and Biochemistry,
2010
2.

Okada M., Ito S., Matsubara A., Iwakura I., Egoshi S., Ueda M.
Organic and Biomolecular Chemistry,
2009
3.
10.1016/j.mencom.2020.01.003_bib0015
Mander
Synlett,
2003
4.

O'Sullivan T.P., Zhang H., Mander L.N.
Organic and Biomolecular Chemistry,
2007
5.

Straus D.S., Glass C.K.
Medicinal Research Reviews,
2001
6.

Zanoni G., Castronovo F., Perani E., Vidari G.
Journal of Organic Chemistry,
2003
7.

Brummond K.M., Sill P.C., Chen H.
Organic Letters,
2003
8.
Gimazetdinov A.M., Khalfitdinova L.A., Miftakhov M.S.
Mendeleev Communications,
2013
9.

Wexler B.A., Toder B.H., Minaskanian G., Smith A.B.
Journal of Organic Chemistry,
1982
10.
![A simple and efficient synthesis of enantiomeric (3aRS,4RS,6aSR)-4-hydroxy-3,3a,4,6a-tetrahydro-1H-cyclopenta[c]furan-1-ones](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Gimazetdinov A.M., Gataullin S.S., Bushmarinov I.S., Miftakhov M.S.
Tetrahedron,
2012
11.

Gimazetdinov A.M., Al'mukhametov A.Z., Spirikhin L.V., Miftakhov M.S.
Tetrahedron Letters,
2017
12.
Al’mukhametov A.Z., Gimazetdinov A.M., Miftakhov M.S.
Mendeleev Communications,
2018