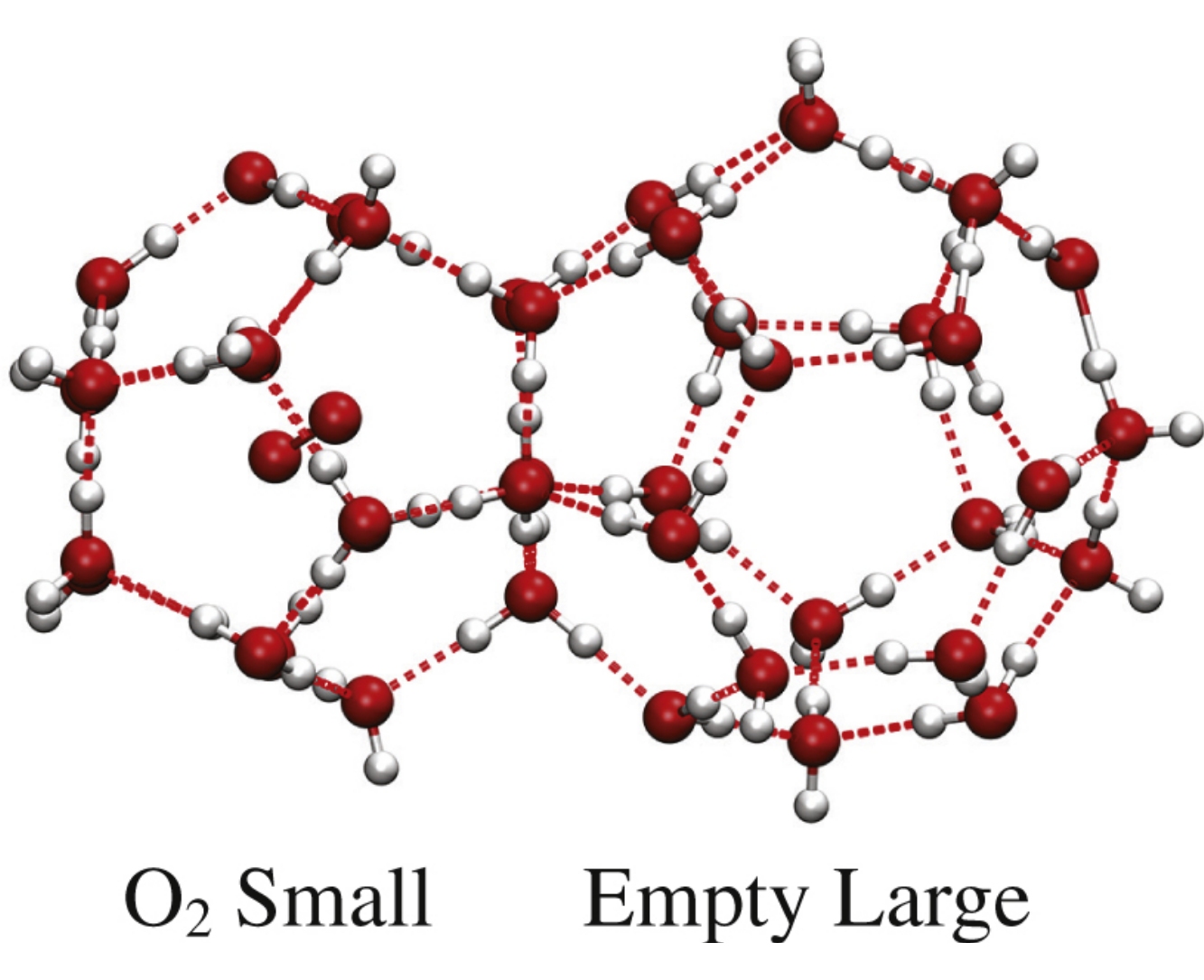
Abstract
The possibility of formation of double O3+N2, O3+O2, O3+CO2 hydrates and their structural and energetic properties have been estimated using the DFT calculations in order to expand the hydrate application in ozone storage technology. It was shown that N2 is the most suitable helper gas that could stabilize water framework in the presence of ozone.
References
1.

Subbotin O.S., Bozhko Y.Y., Zhdanov R.K., Gets K.V., Belosludov V.R., Belosludov R.V., Kawazoe Y.
Physical Chemistry Chemical Physics,
2018
2.

Subbotin O.S., Adamova T.P., Belosludov R.V., Mizuseki H., Kawazoe Y., Belosludov V.R.
Journal of Structural Chemistry,
2012
3.

Nakajima T., Kudo T., Ohmura R., Takeya S., Mori Y.H.
PLoS ONE,
2012
4.

Shishido K., Muromachi S., Nakamura R., Takeya S., Ohmura R.
New Journal of Chemistry,
2014
5.

Watanabe K., Nagashima H.D., Nakamura R., Ohmura R.
ACS Sustainable Chemistry and Engineering,
2018
6.
M. J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G.A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H.P. Hratchian, A.F. Izmaylov, J. Bloino, G. Zheng, J.L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J.A. Montgomery, Jr., J.E. Peralta, F. Ogliaro, M. Bearpark, J.J. Heyd, E. Brothers, K.N. Kudin, V.N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J.C. Burant, S.S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J.M. Millam, M. Klene, J.E. Knox, J.B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E. Stratmann, O. Yazyev, A.J. Austin, R. Cammi, C. Pomelli, J.W. Ochterski, R.L. Martin, K. Morokuma, V.G. Zakrzewski, G.A. Voth, P. Salvador, J.J. Dannenberg, S. Dapprich, A.D. Daniels, O. Farkas, J.B. Foresman, J.V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Revision A.01, Gaussian, Inc., Wallingford, CT, 2009.
7.

Plumley J.A., Dannenberg J.J.
Journal of Computational Chemistry,
2011
8.

Zatsikha Y.V., Blesener T.S., Goff P.C., Healy A.T., Swedin R.K., Herbert D.E., Rohde G.T., Chanawanno K., Ziegler C.J., Belosludov R.V., Blank D.A., Nemykin V.N.
Journal of Physical Chemistry C,
2018
9.

Cao X., Su Y., Liu Y., Zhao J., Liu C.
Journal of Physical Chemistry A,
2013
10.

Khan A.
Journal of Chemical Physics,
1999
11.

Belosludov R.V., Mizuseki H., Souissi M., Kawazoe Y., Kudoh J., Subbotin O.S., Adamova T.P., Belosludov V.R.
Journal of Structural Chemistry,
2012
12.

Curtiss L.A., Frurip D.J., Blander M.
Journal of Chemical Physics,
1979
13.

Kumar P., Sathyamurthy N.
Journal of Physical Chemistry A,
2011
14.
10.1016/j.mencom.2019.11.036_bib0070
Bozhko
J. Phys.: Conf. Ser.,
2018