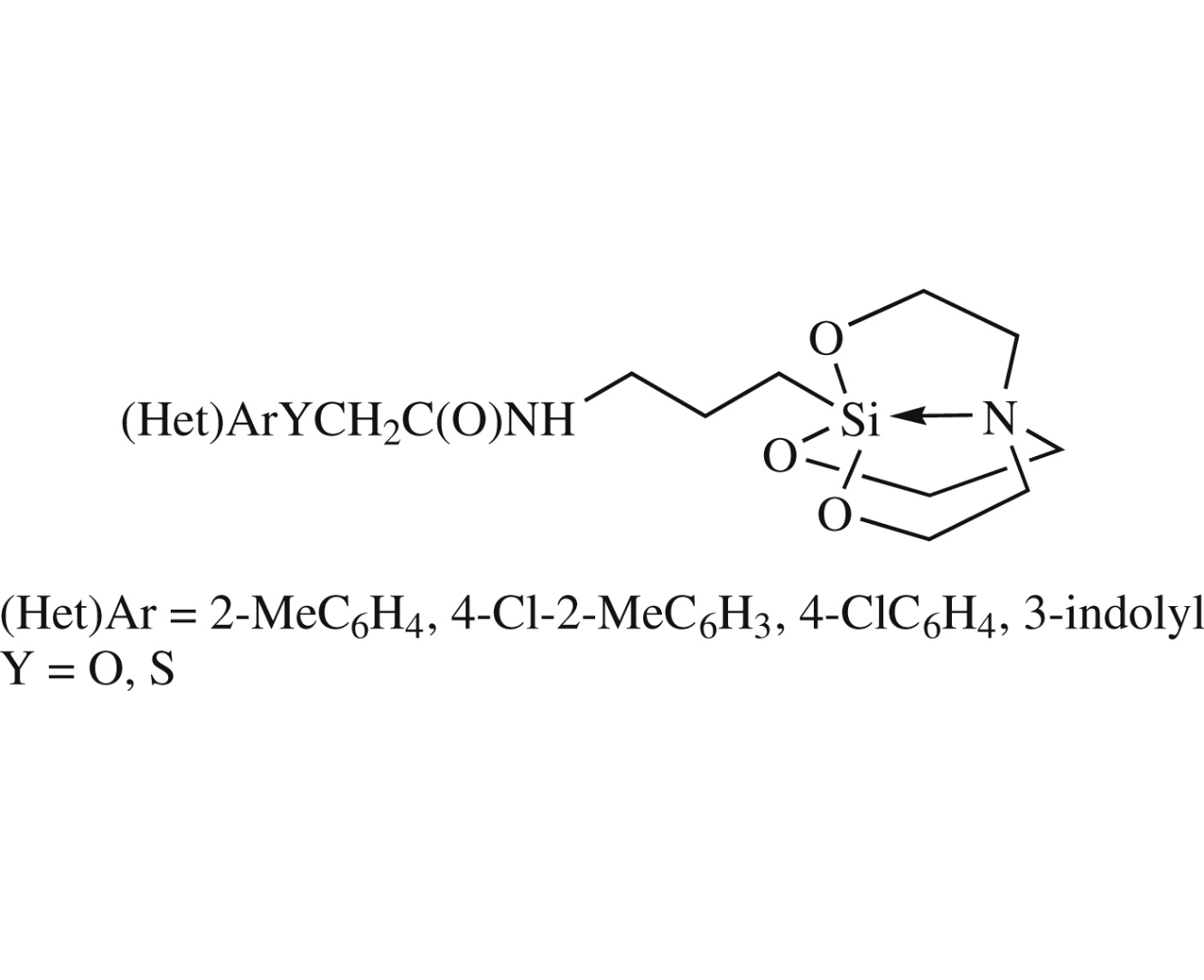
Abstract
Silatranes bearing an amide group in the axial chain have been synthesized by the reaction of 1-(3-aminopropyl)silatrane with arylchalcogenylacetic acids (Het)ArYCH2CO2H [(Het)Ar=2-MeC6H4, 4-Cl-2-MeC6H3, 4-ClC6H4, 3-indolyl; Y=O, S], and their structures have been established by 1H,13C, 15N, 29Si NMR and IR spectroscopy. When 4-chlorophenylsulfonylacetic acid was employed, the reaction proceeded unexpectedly via the 4-chlorophenyl methyl sulfone intermediate and resulted in methyl 4-[3-(1 silatranyl)propylamino]phenyl sulfone.
References
1.
10.1016/j.mencom.2019.11.029_sbref0005a
Voronkov
Silatrany v meditsine i sel'skom khozyaistve (Silatranes in Medicine and Agriculture),
2005
2.

Puri J.K., Singh R., Chahal V.K.
Chemical Society Reviews,
2011
3.

Adamovich S.N., Novokshonov V.V., Ushakov I.A., Elshina V.G., Oborina E.N.
Russian Chemical Bulletin,
2018
4.

Lazareva N.F., Lazarev I.M.
Russian Chemical Bulletin,
2018
5.

Materna K.L., Brennan B.J., Brudvig G.W.
Dalton Transactions,
2015
6.

Huang K., Hsieh C., Kan H., Hsieh M., Hsieh S., Chau L., Cheng T., Lin W.
Sensors and Actuators, B: Chemical,
2012
7.

Hsieh S., Chao W., Hsieh C.
Journal of Nanoscience and Nanotechnology,
2009
8.

Singh G., Saroa A., Girdhar S., Rani S., Sahoo S., Choquesillo-Lazarte D.
Inorganica Chimica Acta,
2015
9.

Han A., Li L., Qing K., Qi X., Hou L., Luo X., Shi S., Ye F.
Bioorganic and Medicinal Chemistry Letters,
2013
10.

Ye F., Song X., Liu J., Xu X., Wang Y., Hu L., Wang Y., Liang G., Guo P., Xie Z.
Chemical Biology and Drug Design,
2015
11.

Voronkov M.G., Baryshok V.P.
Pharmaceutical Chemistry Journal,
2004
12.

Mirskova A.N., Adamovich S.N., Mirskov R.G., Voronkov M.G.
Russian Chemical Bulletin,
2014
13.
S. N. Adamovich, A.N. Mirskova and O. P. Kolesnikova, RU Patent 2623034, 2016.
14.
A. N. Mirskova, S.N. Adamovich and R. G. Mirskov, RU Patent 2642778, 2016.
15.

Adamovich S.N.
Applied Organometallic Chemistry,
2019
16.

Adamovich S.N., Mirskov R.G., Mirskova A.N., Voronkov M.G.
Russian Chemical Bulletin,
2012
17.

Gooßen L., Ohlmann D., Lange P.
Synthesis,
2008
18.

Lundberg H., Tinnis F., Adolfsson H.
Chemistry - A European Journal,
2012
19.

Lanigan R.M., Sheppard T.D.
European Journal of Organic Chemistry,
2013
20.
The Amide Linkage: Structural Significance in Chemistry, Biochemistry, and Materials Science, eds. A. Greenberg, C. M. Breneman and J. F. Liebman, John Wiley & Sons, 2000
21.

22.

Adamovich S.N., Mirskova A.N., Zel′bst E.A., Fundamensky V.S.
Journal of Structural Chemistry,
2017