Abstract
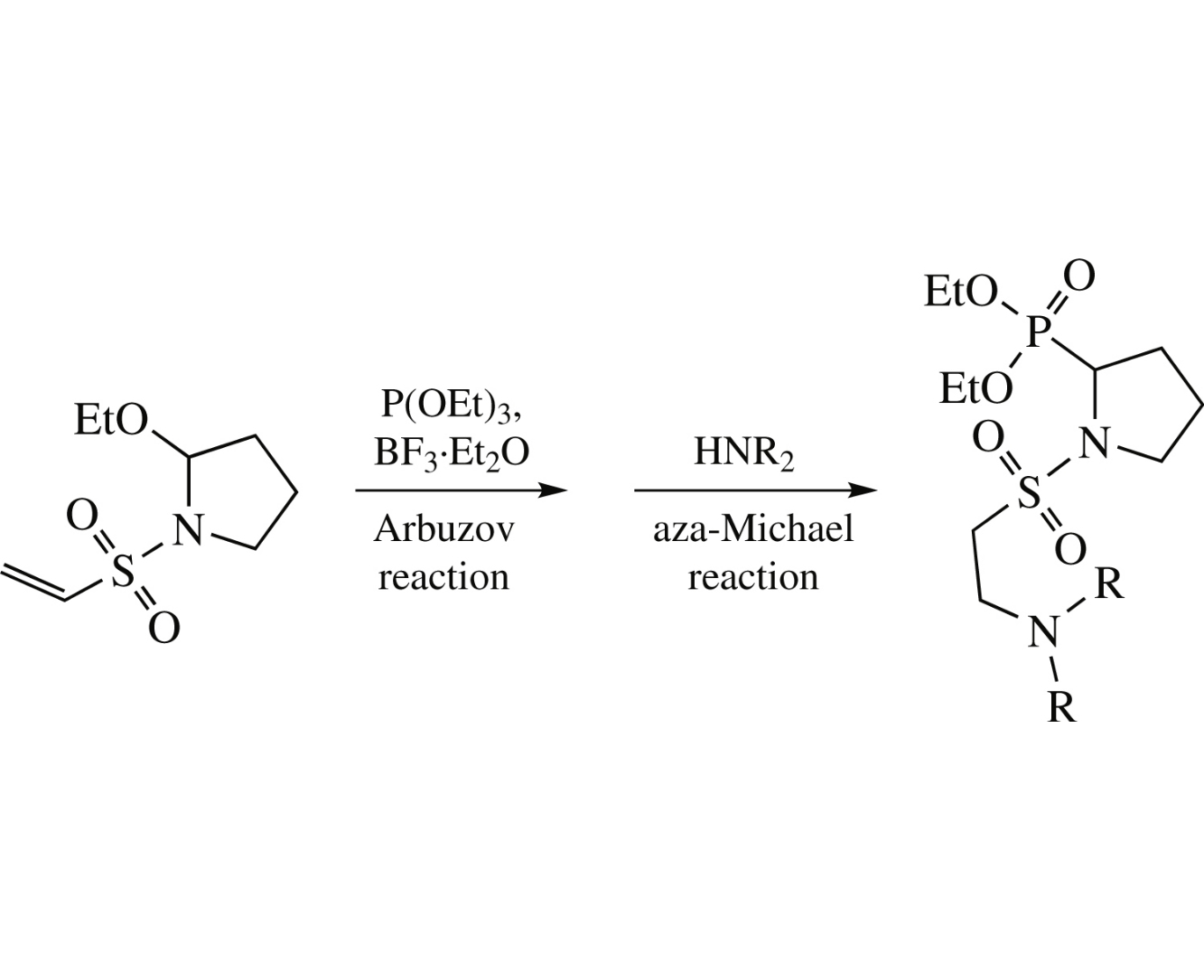
1-(2-Aminoethylsulfonyl)-2-phosphorylpyrrolidines have been synthesized via boron trifluoride-catalyzed Arbuzov reaction of 2-ethoxy-1-(vinylsulfonyl)pyrrolidine with triethyl phosphite followed by aza-Michael reaction of thus obtained 2-phosphoryl-1-(vinylsulfonyl)pyrrolidine with secondary amines. The cytotoxicity of the prepared 1-(2-aminoethylsulfonyl)-2-phosphorylpyrrolidines against M-Hela tumor cell line is comparable with that of tamoxifen, whereas the cytotoxicity against normal cell line is twofold lower.
References
1.
10.1016/j.mencom.2019.11.028_bib0005
Berlinck
Modern Alkaloids: Structure, Isolation, Synthesis, and Biology,
2007
2.
10.1016/j.mencom.2019.11.028_bib0010
Buckingham
Dictionary of Alkaloids,
2010
3.

4.

Moonen K., Laureyn I., Stevens C.V.
Chemical Reviews,
2004
5.

Mucha A., Kafarski P., Berlicki Ł.
Journal of Medicinal Chemistry,
2011
6.

Orsini F., Sello G., Sisti M.
Current Medicinal Chemistry,
2009
7.
W. Sirrenberg, I. Hammann and B. Homeyer, US Patent 3911058A, 1975.
8.
W. Sirrenberg and I. Hammann, US Patent 3856892A, 1999.
9.
J. Hassan, US Patent 6812224B2, 2004.
10.
R. L Bugianesi, G.A. Doherty, A. Gentry, J.J. Hale, C.L. Lynch, S.G. Mills and W. E. Neway III, US Patent 7479504B2, 2009.
11.

Van der Veken P., Soroka A., Brandt I., Chen Y., Maes M., Lambeir A., Chen X., Haemers A., Scharpé S., Augustyns K., De Meester I.
Journal of Medicinal Chemistry,
2007
12.

Belyaev A., Zhang X., Augustyns K., Lambeir A., De Meester I., Vedernikova I., Scharpé S., Haemers A.
Journal of Medicinal Chemistry,
1999
13.

Gilmore B.F., Carson L., McShane L.L., Quinn D., Coulter W.A., Walker B.
Biochemical and Biophysical Research Communications,
2006
14.

Sabidó E., Tarragó T., Niessen S., Cravatt B.F., Giralt E.
ChemBioChem,
2009
15.

Camp N.P., Hawkins P.C., Hitchcock P.B., Gani D.
Bioorganic and Medicinal Chemistry Letters,
1992
16.

Camp N.P., Perrey D.A., Kinchington D., Hawkins P.C., Gani D.
Bioorganic and Medicinal Chemistry,
1995
17.

Das H., Wang L., Kamath A., Bukowski J.F.
Blood,
2001
18.

Arizpe A., Rodríguez-Mata M., Sayago F.J., Pueyo M.J., Gotor V., Jiménez A.I., Gotor-Fernández V., Cativiela C.
Tetrahedron Asymmetry,
2015
19.

Hirata S., Kuriyama M., Onomura O.
Tetrahedron,
2011
20.

Shono T., Matsumura Y., Tsubata K.
Tetrahedron Letters,
1981
21.

Smolobochkin A.V., Muravyeva E.A., Vagapova L.I., Knyazeva I.R., Voronina J.K., Burilov A.R., Pudovik M.A., Gildebrant A.V., Sazykin I.S., Sazykina M.A., Gazizov A.S.
Chemistry and Biodiversity,
2019
22.
![Reaction of 4-Chloro-6-[1-(vinylsulfonyl)pyrrolidin-2-yl]benzene-1,3-diol with Some Amines](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Smolobochkin A.V., Anikina E.A., Gazizov A.S., Vagapova L.I., Burilov A.R., Pudovik M.A.
Russian Journal of General Chemistry,
2018
23.
Vagapova L.I., Voronina J.K., Syakaev V.V., Burilov A.R., Garifzyanov A.R., Pudovik M.A.
Mendeleev Communications,
2018
24.

Voloshina A.D., Semenov V.E., Strobykina A.S., Kulik N.V., Krylova E.S., Zobov V.V., Reznik V.S.
Russian Journal of Bioorganic Chemistry,
2017
25.
![Unusual nanosized associates of carboxy-calix[4]resorcinarene and cetylpyridinium chloride: the macrocycle as a glue for surfactant micelles](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Morozova J.E., Syakaev V.V., Shalaeva Y.V., Ermakova A.M., Nizameev I.R., Kadirov M.K., Voloshina A.D., Zobov V.V., Antipin I.S., Konovalov A.I.
Soft Matter,
2017