Abstract
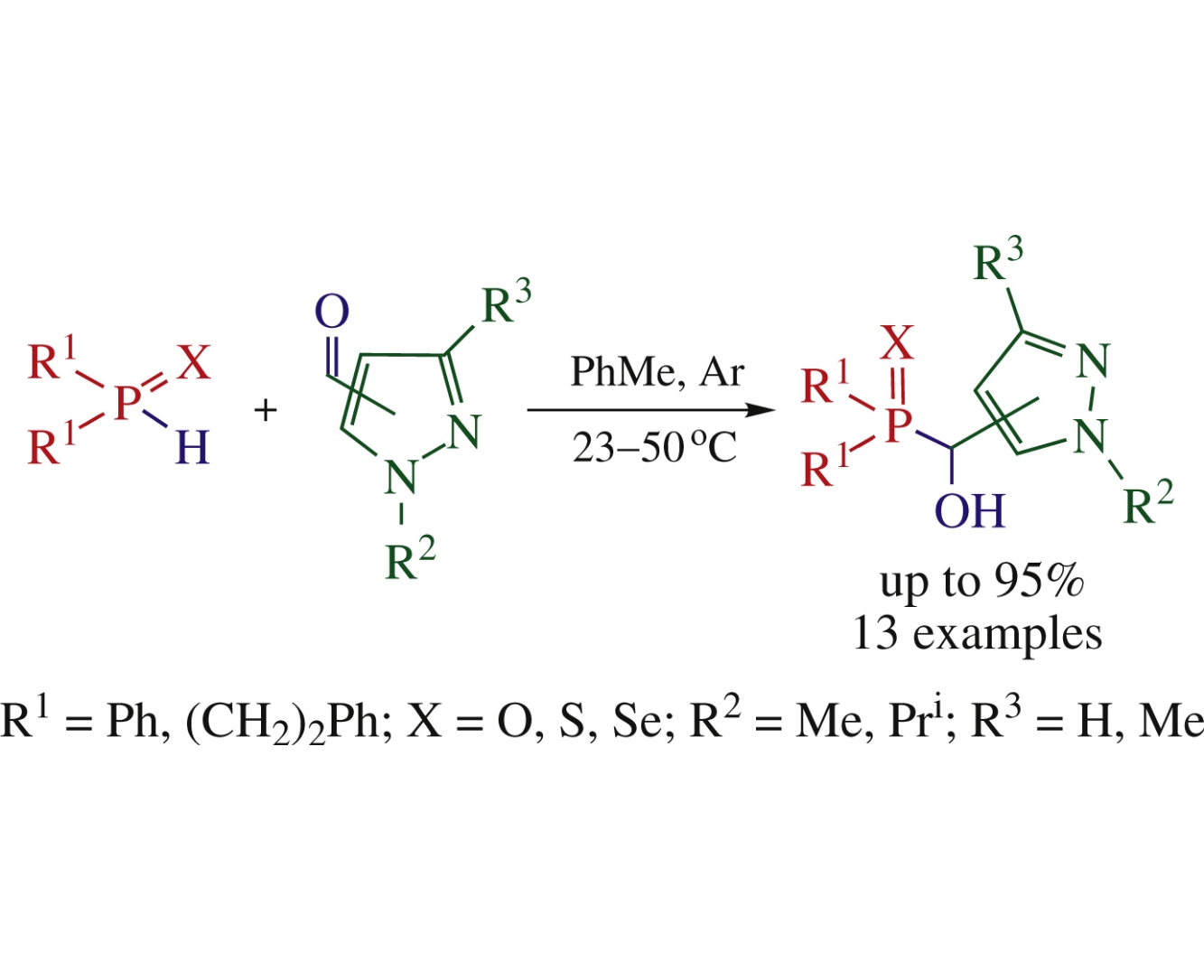
(Chalcogenophosphoryl)(hydroxyl)methyl-substituted pyrazoles were obtained by catalyst-free reaction between 4- and 5-pyrazolecarbaldehydes and secondary phosphine chalcogenides R2P(X)H [R=Ph, (CH2)2Ph, X=O, S, Se] at 23–50°C in toluene.
References
1.

Goulioukina N.S., Makukhin N.N., Beletskaya I.P.
Russian Chemical Reviews,
2016
2.

Abakumov G.A., Piskunov A.V., Cherkasov V.K., Fedushkin I.L., Ananikov V.P., Eremin D.B., Gordeev E.G., Beletskaya I.P., Averin A.D., Bochkarev M.N., Trifonov A.A., Dzhemilev U.M., D'yakonov V.A., Egorov M.P., Vereshchagin A.N., et. al.
Russian Chemical Reviews,
2018
3.

Baiju T.V., Namboothiri I.N.
Chemical Record,
2017
4.

E. Ali T., M. Abdel-Kariem S.
Heterocycles,
2012
5.

Spink E., Ding D., Peng Z., Boudreau M.A., Leemans E., Lastochkin E., Song W., Lichtenwalter K., O’Daniel P.I., Testero S.A., Pi H., Schroeder V.A., Wolter W.R., Antunes N.T., Suckow M.A., et. al.
Journal of Medicinal Chemistry,
2015
6.
M. Chang, S. Mobashery, E. Spink, D. Ding, S. Testero, E. Leemans and M. A. Bouderau, Patent WO 2016049586, 2016.
7.

Zhang W., Tam C.P., Wang J., Szostak J.W.
ACS Central Science,
2016
8.
D. S. Goldfarb, Patent US 20090163545 A1, 2009.
9.

Kim Y., Yoon Y.
Bioorganic and Medicinal Chemistry Letters,
2014
10.

Yoon Y.
Bioorganic and Medicinal Chemistry Letters,
2014
11.

Lilley M., Mambwe B., Thompson M.J., Jackson R.F., Muimo R.
Chemical Communications,
2015
12.

Kee J., Oslund R.C., Couvillon A.D., Muir T.W.
Organic Letters,
2014
13.

Modranka J., Jakubowski R., Różalski M., Krajewska U., Janecka A., Gach K., Pomorska D., Janecki T.
European Journal of Medicinal Chemistry,
2015
14.

Nayab Rasool S., Subramanyam C., Janakiramudu D.B., Supraja P., Usha R., Raju C.N.
Phosphorus, Sulfur and Silicon and the Related Elements,
2018
15.
O. Gebauer, H. Gayer, U. Heinemann, S. Herrmann, S. Hillebrand, H.-l. Elbe, R. Ebbert, U. Wachendorff-Neumann, P. Dahmen and K.-H. Kuck, Patent US 20050187224, 2005.
16.
U. Doeller, M. Maier, A. Kuhlmann, D. Jans, A.M. Pinchuk, A.P. Marchenko and G. N. Koydan, Patent WO 2005082917, 2005.
17.
P. C. Miller, J.M. Curtis, J.M. Molyneaux and T. J. Owen, Patent WO 2000046229, 2000.
18.

Shidlovskii A.F., Peregudov A.S., Bulychev Y.N., Chkanikov N.D.
Pharmaceutical Chemistry Journal,
2009
19.

Sudhapriya N., Balachandran C., Awale S., Perumal P.T.
New Journal of Chemistry,
2017
20.

El-Sayed N.F., Ewies E.F., El-Hussieny M., Boulos L.S., Shalaby E.M.
Zeitschrift fur Naturforschung - Section B Journal of Chemical Sciences,
2016
21.

Rani V.E., Ravindranath L.K.
Open Pharmaceutical Sciences Journal,
2016
22.
10.1016/j.mencom.2019.11.027_sbref0020e
Rani
Am. J. Org. Chem.,
2016
23.

Leemans E., Mahasenan K.V., Kumarasiri M., Spink E., Ding D., O’Daniel P.I., Boudreau M.A., Lastochkin E., Testero S.A., Yamaguchi T., Lee M., Hesek D., Fisher J.F., Chang M., Mobashery S., et. al.
Bioorganic and Medicinal Chemistry Letters,
2016
24.
10.1016/j.mencom.2019.11.027_sbref0025a
Malinowska
Pol. Merkuriusz Lek.,
2009
25.

Kupcewicz B., Sobiesiak K., Malinowska K., Koprowska K., Czyz M., Keppler B., Budzisz E.
Medicinal Chemistry Research,
2012
26.

Budzisz E., Krajewska U., Rozalski M., Szulawska A., Czyz M., Nawrot B.
European Journal of Pharmacology,
2004
27.

Budzisz E., Miernicka M., Lorenz I., Mayer P., Krajewska U., Rozalski M.
Polyhedron,
2009
28.

Varaksina E.A., Taydakov I.V., Ambrozevich S.A., Selyukov A.S., Lyssenko K.A., Jesus L.T., Freire R.O.
Journal of Luminescence,
2018
29.

Vitukhnovsky A.G., Ambrozevich S.A., Korshunov V.M., Taydakov I.V., Lyssenko K.A., Metlin M.T., Selyukov A.S.
Journal of Luminescence,
2018
30.

Metlina D.A., Metlin M.T., Ambrozevich S.A., Taydakov I.V., Lyssenko K.A., Vitukhnovsky A.G., Selyukov A.S., Krivobok V.S., Aminev D.F., Tobokhova A.S.
Journal of Luminescence,
2018
31.

Grotjahn D.B., Combs D., Van S., Aguirre G., Ortega F.
Inorganic Chemistry,
2000
32.

Alshakova I.D., Korobkov I., Kuzmina L.G., Nikonov G.I.
Journal of Organometallic Chemistry,
2017
33.

Geng Z., Zhang J., Li N., Chen J., Huang X., Zhang S., Li H., Tao J., Wang X.
Tetrahedron,
2014
34.

Galkina M.A., Bodrin G.V., Goryunov E.I., Goryunova I.B., Ambartsumyan A.A., Vasil’eva T.T., Protopopova P.S., Saifutiarova A.E., Uryupin A.B., Brel V.K., Kochetkov K.A.
Russian Chemical Bulletin,
2016
35.

Ambartsumyan A.A., Sviridova L.A., Vorozhtsov N.I., Goryunov E.I., Bodrin G.V., Goryunova I.B., Uryupin A.B., Vasil’eva T.T., Chakhovskaya O.V., Kochetkov K.A., Nifant’ev E.E.
Doklady Chemistry,
2013
36.
10.1016/j.mencom.2019.11.027_sbref0060a
Abranmov
Dokl. Akad. Nauk SSSR,
1950
37.
10.1016/j.mencom.2019.11.027_sbref0060b
Organophosphorus Reagents
A Practical Approach in Chemistry,
2004
38.

Guin J., Wang Q., van Gemmeren M., List B.
Angewandte Chemie - International Edition,
2014
39.

Gusarova N.K., Reutskaya A.M., Ivanova N.I., Medvedeva A.S., Demina M.M., Novopashin P.S., Afonin A.V., Albanov A.I., Trofimov B.A.
Journal of Organometallic Chemistry,
2002
40.

Ivanova N.I., Gusarova N.K., Nikitina E.A., Albanov A.I., Sinegovskaya L.M., Nikitin M.V., Konovalova N.A., Trofimov B.A.
Phosphorus, Sulfur and Silicon and the Related Elements,
2004
41.
10.1016/j.mencom.2019.11.027_sbref0065c
Gusarova
Synthesis,
2006
42.

Gusarova N., Ivanova N., Volkov P., Khrapova K., Larina L., Smirnov V., Borodina T., Trofimov B.
Synthesis,
2015