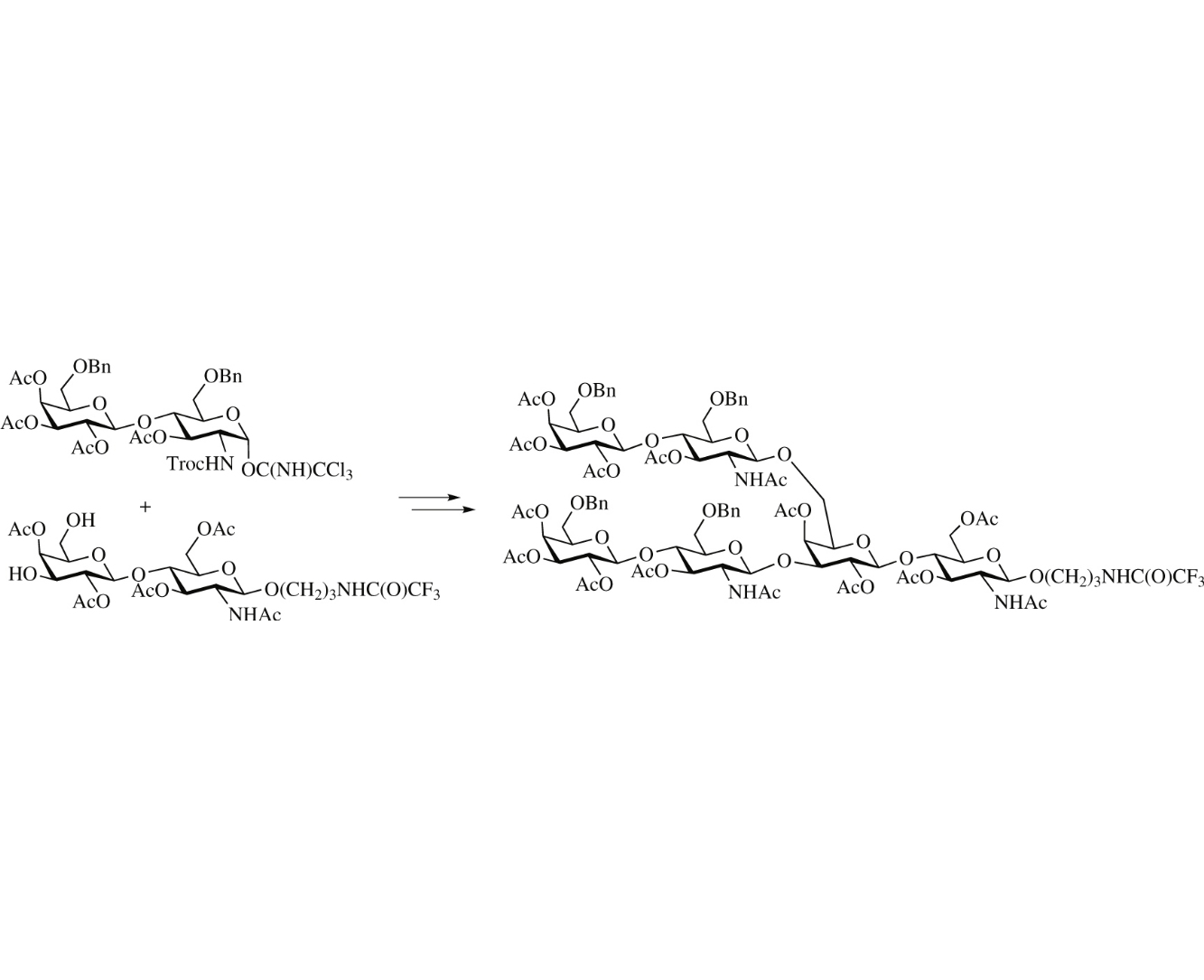
Abstract
The protected spacer-armed branched tri-N-acetyllactosamine has been synthesized as a building block for the synthesis of biantennary ABH blood group antigens. The key synthetic step is the simultaneous glycosylation of the 3′- and 6′-OH groups of an N-acetyllactosamine glycosyl acceptor by N-Troc-lactosamine trichloroacetimidate.
References
1.

Koscielak J.
Transfusion Medicine,
2001
2.
Pazynina G.V., Tyrtysh T.V., Bovin N.V.
Mendeleev Communications,
2002
3.

Meloncelli P.J., Lowary T.L.
Carbohydrate Research,
2010
4.

Meloncelli P.J., West L.J., Lowary T.L.
Carbohydrate Research,
2011
5.

Meloncelli P.J., Lowary T.L.
Australian Journal of Chemistry,
2009
6.

Karki G., Mishra V.N., Mandal P.K.
Glycoconjugate Journal,
2015
7.

Zimmermann P., Greilich U., Schmidt R.R.
Tetrahedron Letters,
1990
8.

Ryzhov I.M., Korchagina E.Y., Popova I.S., Tyrtysh T.V., Paramonov A.S., Bovin N.V.
Carbohydrate Research,
2016
9.

Blixt O., Razi N.
Methods in Enzymology,
2006
10.

Severov V.V., Belyanchikov I.M., Pazynina G.V., Bovin N.V.
Russian Journal of Bioorganic Chemistry,
2007
11.

Zinin A.I., Malysheva N.N., Shpirt A.M., Torgov V.I., Kononov L.O.
Carbohydrate Research,
2007
12.

Severov V.V., Pazynina G.V., Ovchinnikova T.V., Bovin N.V.
Russian Journal of Bioorganic Chemistry,
2015
13.

Sato S., Ito Y., Ogawa T.
Carbohydrate Research,
1986
14.

Jansson K., Frejd T., Kihlberg J., Magnusson G.
Tetrahedron Letters,
1988
15.

Kalidasan K., Su Y., Wu X., Yao S.Q., Uttamchandani M.
Chemical Communications,
2013
16.
10.1016/j.mencom.2019.11.026_bib0080
Schmidt
1994
17.

Huang C., Wang N., Fujiki K., Otsuka Y., Akamatsu M., Fujimoto Y., Fukase K.
Journal of Carbohydrate Chemistry,
2010