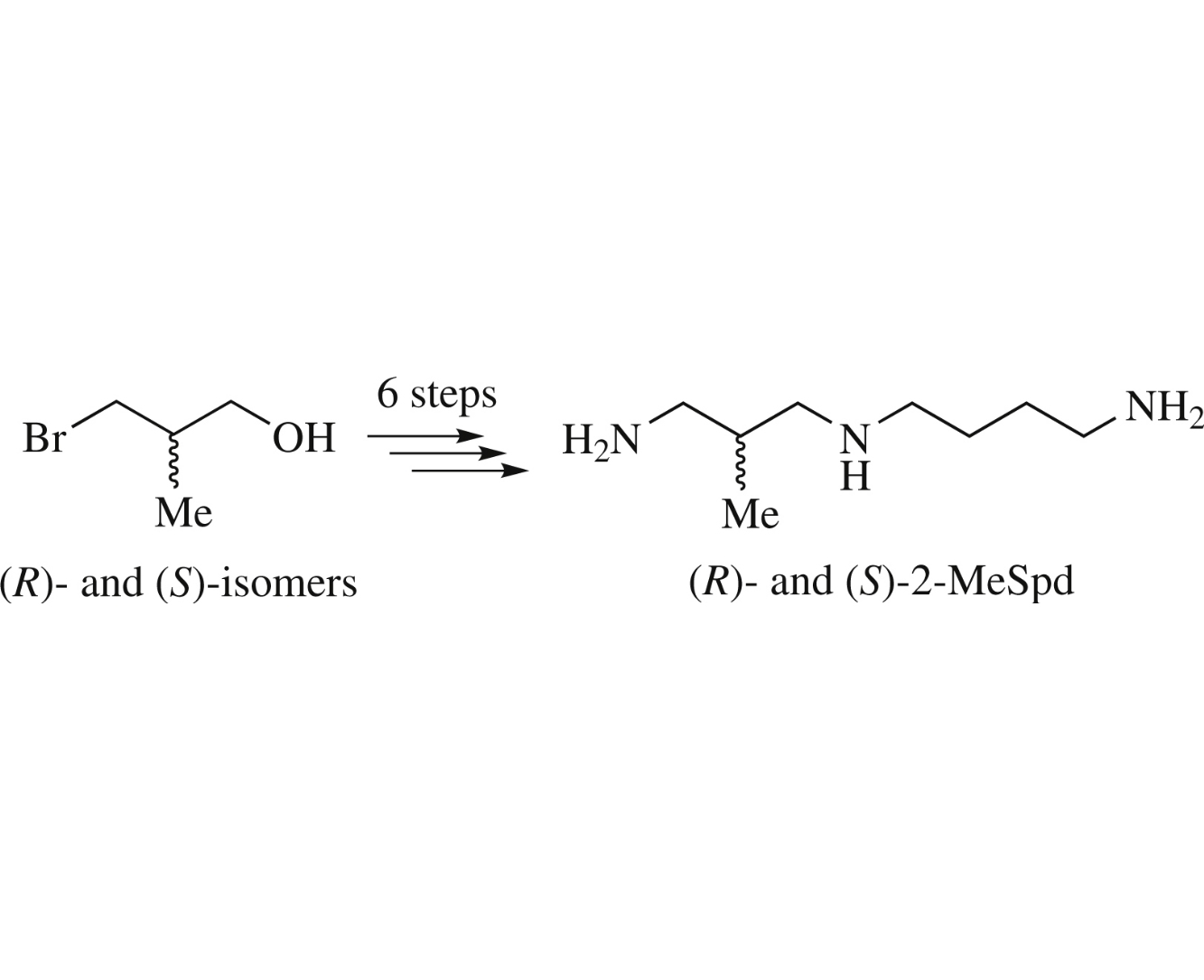
Abstract
The title compounds, (R)- and (S)-isomers of 1,8-diamino-2-methyl-4-azaoctane, were prepared from commercially available enantiomers of 3-bromo-2-methylpropan-1-ol, each in six steps and good overall yields.
References
1.

Casero R.A., Murray Stewart T., Pegg A.E.
Nature Reviews Cancer,
2018
2.

3.

Hyvönen M.T., Keinänen T.A., Khomutov M., Simonian A., Weisell J., Kochetkov S.N., Vepsäläinen J., Alhonen L., Khomutov A.R.
Journal of Medicinal Chemistry,
2011
4.

Keinänen T.A., Hyvönen M.T., Alhonen L., Vepsäläinen J., Khomutov A.R.
Amino Acids,
2013
5.
Grigorenko N.A., Vepsalainen J., Jarvinen A., Keinanen T., Alhonen L., Janne J., Khomutov A.R.
Mendeleev Communications,
2005
6.

Khomutov M.A., Keinanen T.A., Hyvonen M.T., Weisell J., Vepsalainen J., Alhonen L., Kochetkov S.N., Khomutov A.R.
Russian Journal of Bioorganic Chemistry,
2015
7.

Grigorenko N.A., Khomutov A.R., Keinänen T.A., Järvinen A., Alhonen L., Jänne J., Vepsäläinen J.
Tetrahedron,
2007
8.

Chattopadhyay M.K., Park M.H., Tabor H.
Proceedings of the National Academy of Sciences of the United States of America,
2008
9.

Hyvönen M.T., Khomutov M., Petit M., Weisell J., Kochetkov S.N., Alhonen L., Vepsäläinen J., Khomutov A.R., Keinänen T.A.
ACS Chemical Biology,
2015
10.

Duranti A.
European Journal of Medicinal Chemistry,
2000
11.
Tsedilin A.M., Fakhrutdinov A.N., Eremin D.B., Zalesskiy S.S., Chizhov A.O., Kolotyrkina N.G., Ananikov V.P.
Mendeleev Communications,
2015
12.
Khomutov M.A., Hyvönen M.T., Simonian A.R., Weisell J., Vepsäläinen J., Alhonen L., Kochetkov S.N., Keinänen T.A., Khomutov A.R.
Mendeleev Communications,
2018