Abstract
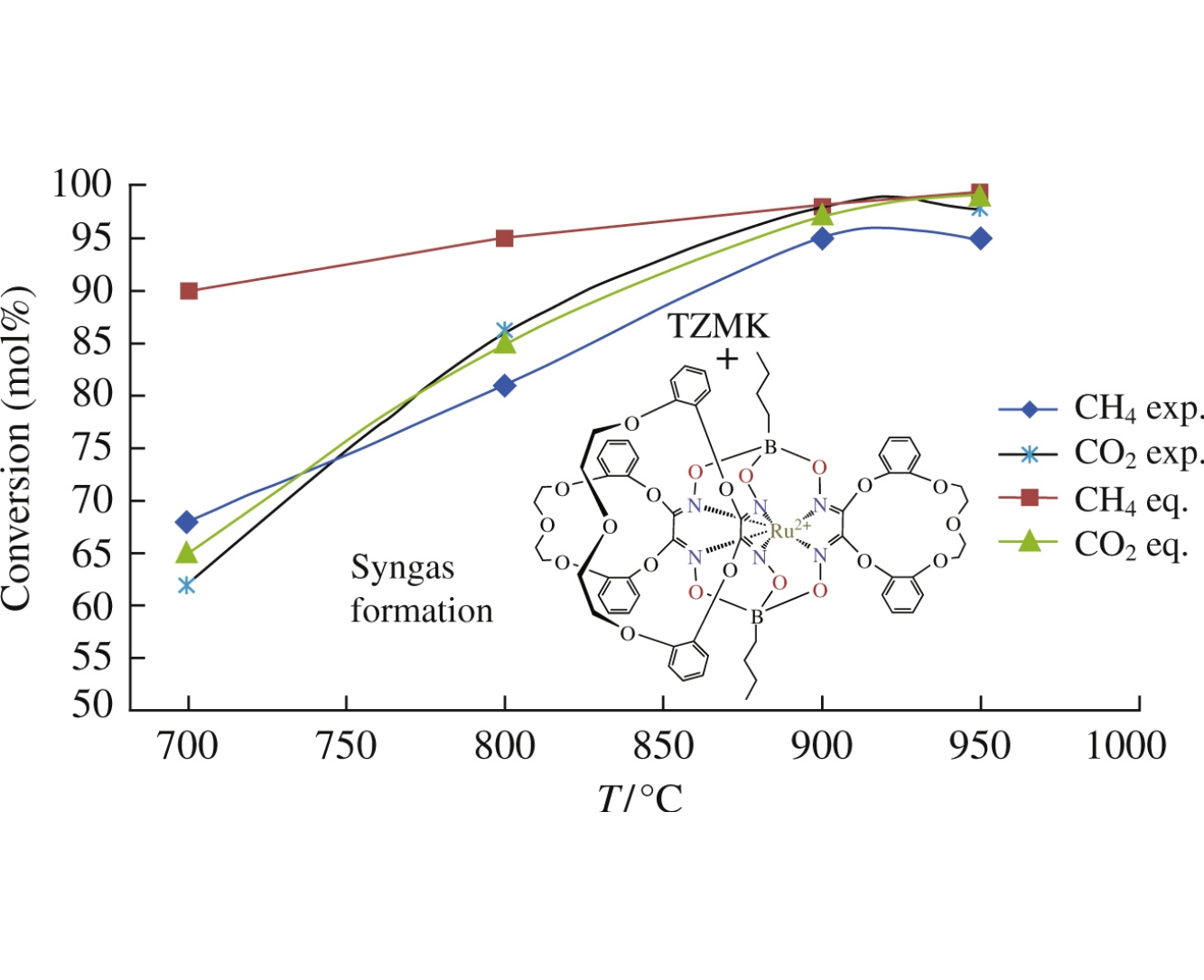
The Fe, Co and Ru clathrochelates bearing terminal polar groups were immobilized as the precursors of single atom catalysts on a surface of highly porous ceramic material used as the support. The obtained materials, which were evaluated for a syngas production from CH4 based on its partial oxidation and dry reforming reactions, did not catalyze the partial oxidation of CH4, while that based on immobilized RuII clathrochelate served as the active and selective catalyst of dry reforming conversion of CH4+CO2 mixture into a syngas containing the equimolar amounts of H2 and CO.
References
1.
L. V. Belunova, V.N. Gribkov, A.I. Chernyak, G.T. Mizyurina, O.A. Mordovin and E. E. Mukhanova, Patent US 5.569.423, 1996.
2.

Shchetanov B.V., Ivakhnenko Y.A., Babashov V.G.
Russian Journal of General Chemistry,
2011
3.
Dospekhi dlya Burana. Materialy i teknologii VIAM dlya MKS ‘Energiya–Buran’ (Armour for ‘Buran’. Materials and Technologies of VIAM for the International Space Station ‘Energy–Buran’), ed. E. N. Kablov, Nauka i Zhizne Station ‘Energy–Buran’) ted.
4.
10.1016/j.mencom.2019.11.022_bib0020
Voloshin
Cage Metal Complexes: Clathrochelates Revisited,
2017
5.

Pantani O., Naskar S., Guillot R., Millet P., Anxolabéhère-Mallart E., Aukauloo A.
Angewandte Chemie - International Edition,
2008
6.

Anxolabéhère-Mallart E., Costentin C., Fournier M., Nowak S., Robert M., Savéant J.
Journal of the American Chemical Society,
2012
7.

El Ghachtouli S., Fournier M., Cherdo S., Guillot R., Charlot M., Anxolabéhère-Mallart E., Robert M., Aukauloo A.
Journal of Physical Chemistry C,
2013
8.

Voloshin Y.Z., Dolganov A.V., Varzatskii O.A., Bubnov Y.N.
Chemical Communications,
2011
9.

Grigoriev S.A., Pushkarev A.S., Pushkareva I.V., Millet P., Belov A.S., Novikov V.V., Belaya I.G., Voloshin Y.Z.
International Journal of Hydrogen Energy,
2017
10.

Varzatskii O.A., Oranskiy D.A., Vakarov S.V., Chornenka N.V., Belov A.S., Vologzhanina A.V., Pavlov A.A., Grigoriev S.A., Pushkarev A.S., Millet P., Kalinichenko V.N., Voloshin Y.Z., Dedov A.G.
International Journal of Hydrogen Energy,
2017
11.

Voloshin Y.Z., Chornenka N.V., Varzatskii O.A., Belov A.S., Grigoriev S.A., Pushkarev A.S., Millet P., Kalinichenko V.N., Belaya I.G., Bugaenko M.G., Dedov A.G.
Electrochimica Acta,
2018
12.

Antuch M., Millet P., Iwase A., Kudo A., Grigoriev S.A., Voloshin Y.Z.
Electrochimica Acta,
2017
13.

Holmen A.
Catalysis Today,
2009
14.

Christian Enger B., Lødeng R., Holmen A.
Applied Catalysis A: General,
2008
15.

Zeng S., Zhang X., Fu X., Zhang L., Su H., Pan H.
Applied Catalysis B: Environmental,
2013
16.

Partial oxidation of methane on Ni and Pd catalysts: Influence of active phase and CeO2 modification
Rodrigues L.M., Silva R.B., Rocha M.G., Bargiela P., Noronha F.B., Brandão S.T.
Catalysis Today,
2012
17.

Chalupka K.A., Jozwiak W.K., Rynkowski J., Maniukiewicz W., Casale S., Dzwigaj S.
Applied Catalysis B: Environmental,
2014
18.

Dedov A.G., Loktev A.S., Komissarenko D.A., Parkhomenko K.V., Roger A.-., Shlyakhtin O.A., Mazo G.N., Moiseev I.I.
Fuel Processing Technology,
2016
19.
Zagaynov I.V., Loktev A.S., Mukhin I.E., Dedov A.G., Moiseev I.I.
Mendeleev Communications,
2017
20.

Nikoo M.K., Amin N.A.
Fuel Processing Technology,
2011