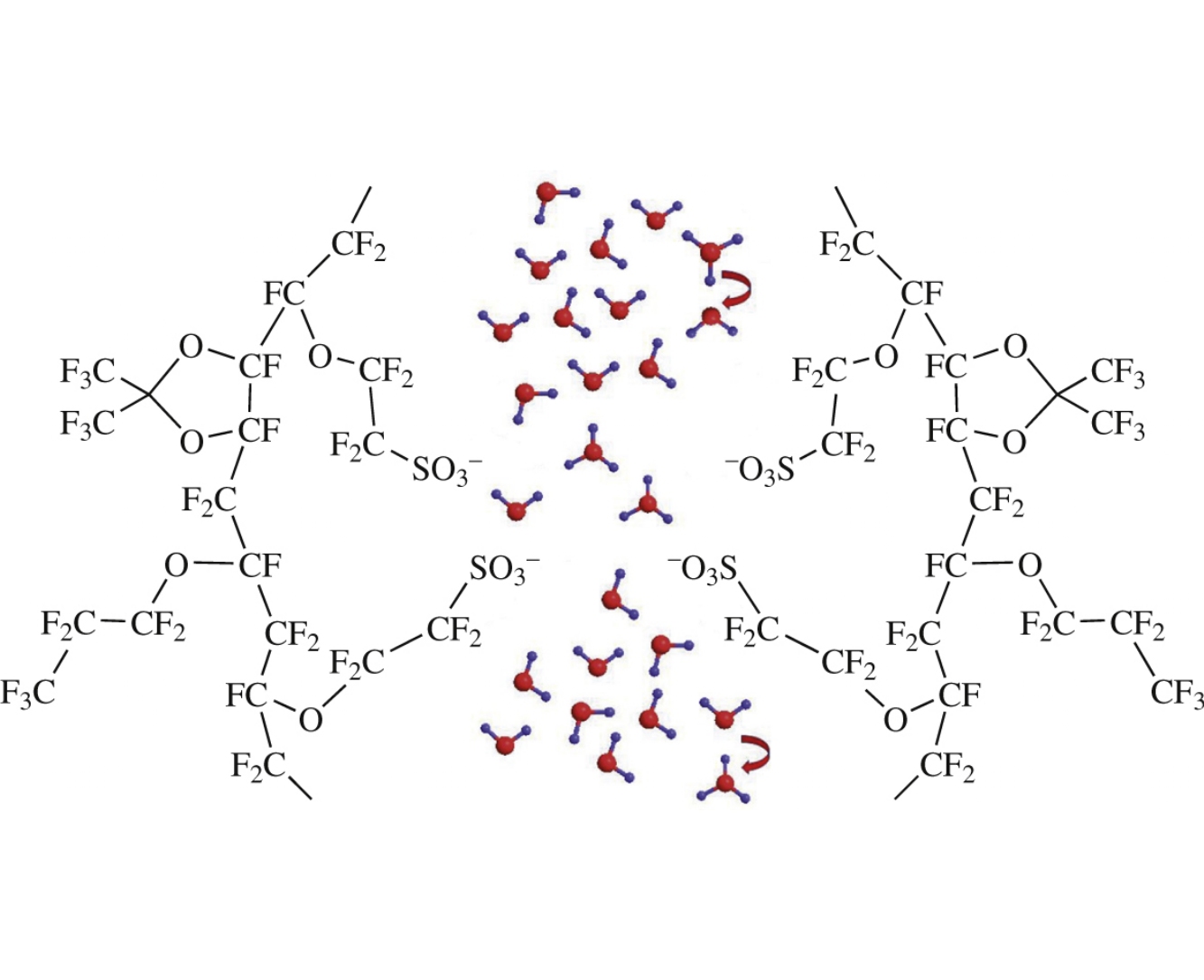
Abstract
A new perfluorinated sulfocationic polymer and a membrane based thereon have been produced using the thermally initiated high-pressure polymerization. The proton conductivity of obtained material is higher than that of commercial Nafion membranes and reaches 57mScm−1 at 21C and 114mScm−1 at 79 C.
References
1.

Brunetti A., Sun Y., Caravella A., Drioli E., Barbieri G.
International Journal of Greenhouse Gas Control,
2015
2.

Tsodikov M.V., Fedotov A.S., Antonov D.O., Uvarov V.I., Bychkov V.Y., Luck F.C.
International Journal of Hydrogen Energy,
2016
3.

Weitemeyer S., Kleinhans D., Vogt T., Agert C.
Renewable Energy,
2015
4.

5.
Yaroslavtsev A.B., Yampolskii Y.P.
Mendeleev Communications,
2014
6.

Golubenko D.V., Pourcelly G., Yaroslavtsev A.B.
Separation and Purification Technology,
2018
7.
Prikhno I.A., Ivanova K.A., Don G.M., Yaroslavtsev A.B.
Mendeleev Communications,
2018
8.

Bai H., Wang H., Zhang J., Zhang J., Lu S., Xiang Y.
Journal of Membrane Science,
2019
9.

Treekamol Y., Schieda M., Schulte K.
Materials Today: Proceedings,
2018
10.
Lee C., Na H., Jeon Y., Jung Hwang H., Kim H., Mochida I., Yoon S., Park J., Shul Y.
Journal of Industrial and Engineering Chemistry,
2019
11.

Amirinejad M., Madaeni S.S., Rafiee E., Amirinejad S.
Journal of Membrane Science,
2011
12.

Ivanchev S.S., Likhomanov V.S., Primachenko O.N., Khaikin S.Y., Barabanov V.G., Kornilov V.V., Odinokov A.S., Kulvelis Y.V., Lebedev V.T., Trunov V.A.
Petroleum Chemistry,
2012
13.

Zharov A.A., Guzyaeva I.A.
Russian Chemical Bulletin,
2010
14.

Sokolov V.I., Boyko V.E., Goriachuk I.O., Igumnov S.M., Molchanova S.I., Pogodina Y.E., Polunin E.V.
Russian Chemical Bulletin,
2017
15.

Greso A.J., Moore R.B., Cable K.M., Jarrett W.L., Mauritz K.A.
Polymer,
1997
16.

Stenina I.A., Yaroslavtsev A.B.
Inorganic Materials,
2017