Abstract
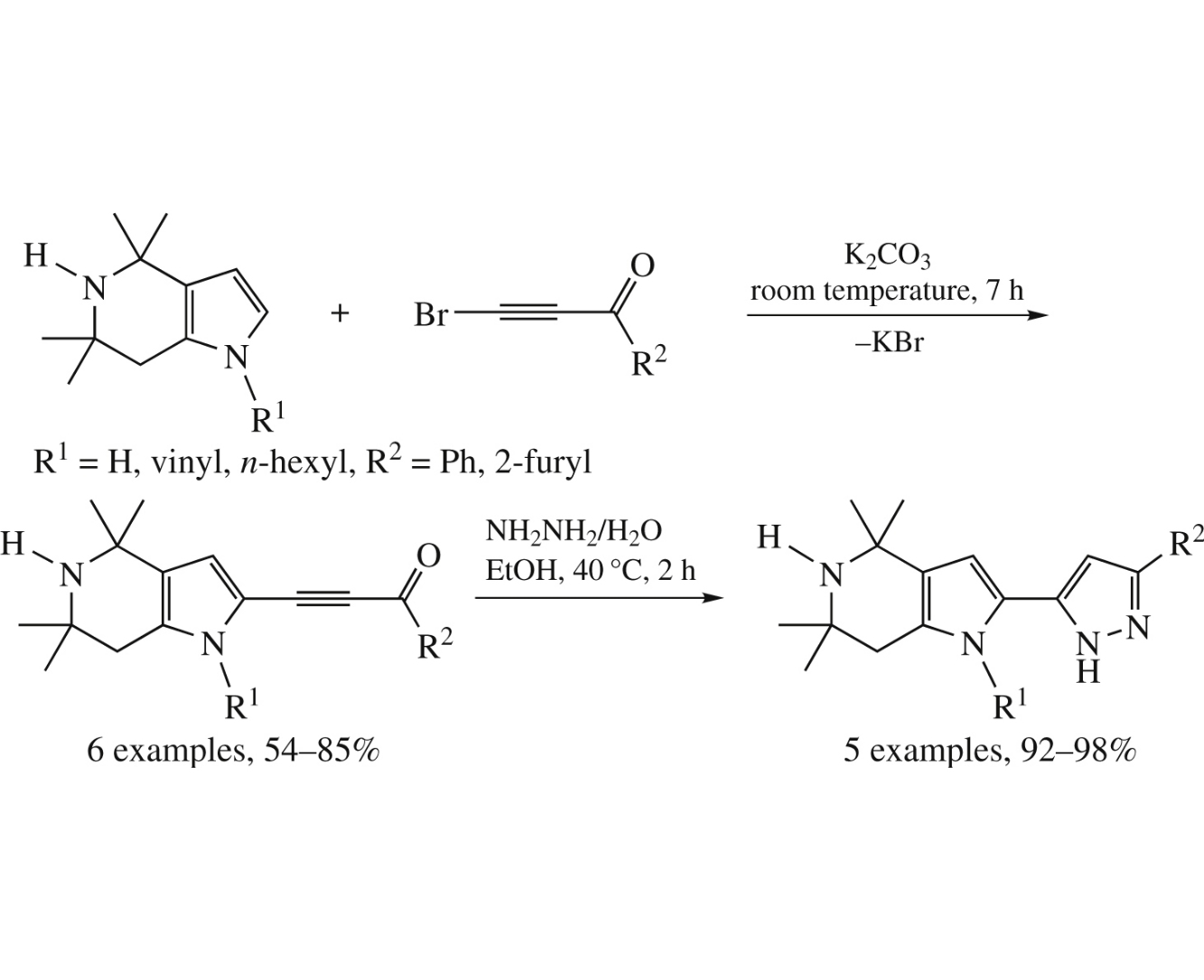
Pharmaceutically relevant substituted 2-(pyrazol-5-yl)-4,5,6,7-tetrahydropyrrolo [3,2-c]pyridines have been assembled in good to excellent yields via the reaction of 2,2,6,6-tetramethylpiperidin-4-one oxime with acetylene, cross-coupling of the resulting 4,4,6,6-tetramethyl-4,5,6,7 tetrahydropyrrolo[3,2-c]pyridines with aroylbromoacetylenes, and reaction of the formed 2-(aroylethynyl)-4,4,6,6-tetramethyl-4,5,6,7-tetrahydropyrrolo[3,2-c]pyridines with hydrazine.
References
1.

Yang X., Zhou G., Wong W.
Chemical Society Reviews,
2015
2.

Gholap S.S.
European Journal of Medicinal Chemistry,
2016
3.

Gouda A.M., Abdelazeem A.H., Omar H.A., Abdalla A.N., Abourehab M.A., Ali H.I.
Bioorganic and Medicinal Chemistry,
2017
4.

Pathania S., Rawal R.K.
European Journal of Medicinal Chemistry,
2018
5.
6.

El-Gamal M.I., Anbar H.S.
Expert Opinion on Therapeutic Patents,
2017
7.

Zhao S., Wang S.
Chemical Society Reviews,
2010
8.

Mérour J., Buron F., Plé K., Bonnet P., Routier S.
Molecules,
2014
9.
![Pyrrolo[2,3-b]pyridine derivatives as potent Bruton’s tyrosine kinase inhibitors](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Zhao X., Huang W., Wang Y., Xin M., Jin Q., Cai J., Tang F., Zhao Y., Xiang H.
Bioorganic and Medicinal Chemistry,
2015
10.

Immadi S.S., Dopart R., Wu Z., Fu B., Kendall D.A., Lu D.
Cannabis and Cannabinoid Research,
2018
11.

12.

Kaptein A., Oubrie A., Zwart E.D., Hoogenboom N., Wit J.D., Kar B.V., Hoek M.V., Vogel G., Kimpe V.D., Schultz-Fademrecht C., Borsboom J., Zeeland M.V., Versteegh J., Kazemier B., Roos J.D., et. al.
Bioorganic and Medicinal Chemistry Letters,
2011
13.
![5-(2-Amino-pyrimidin-4-yl)-1H-pyrrole and 2-(2-amino-pyrimidin-4-yl)-1,5,6,7-tetrahydro-pyrrolo[3,2-c]pyridin-4-one derivatives as new classes of selective and orally available Polo-like kinase 1 inhibitors](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Caruso M., Valsasina B., Ballinari D., Bertrand J., Brasca M.G., Caldarelli M., Cappella P., Fiorentini F., Gianellini L.M., Scolaro A., Beria I.
Bioorganic and Medicinal Chemistry Letters,
2012
14.

Vanotti E., Amici R., Bargiotti A., Berthelsen J., Bosotti R., Ciavolella A., Cirla A., Cristiani C., D’Alessio R., Forte B., Isacchi A., Martina K., Menichincheri M., Molinari A., Montagnoli A., et. al.
Journal of Medicinal Chemistry,
2008
15.
![Constrained analogs of CB-1 antagonists: 1,5,6,7-Tetrahydro-4H-pyrrolo[3,2-c]pyridine-4-one derivatives](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Smith R.A., Fathi Z., Brown S., Choi S., Fan J., Jenkins S., Kluender H.C., Konkar A., Lavoie R., Mays R., Natoli J., O’Connor S.J., Ortiz A.A., Podlogar B., Taing C., et. al.
Bioorganic and Medicinal Chemistry Letters,
2007
16.
![Identification of 2-(4,5,6,7-tetrahydro-1H-pyrrolo[3,2-c]pyridin-3-yl)-ethylamine derivatives as novel GnRH receptor antagonists](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Chen M., Guo Z., Lanier M.C., Zhao L., Betz S.F., Huang C.Q., Loweth C.J., Ashweek N.J., Liu X., Struthers R.S., Bradbury M.J., Behan J.W., Wen J., O’Brien Z., Saunders J., et. al.
Bioorganic and Medicinal Chemistry Letters,
2007
17.
10.1016/j.mencom.2019.11.018_bib0045
Masood
Rasayan J. Chem.,
2016
18.
J. L. Archibald and K. Heatherington, US Patent 3992544, 1976.
19.
![Pyrrolo[3,2- c ]pyridine derivatives as inhibitors of platelet aggregation](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Altomare C., Summo L., Cellamare S., Varlamov A.V., Voskressensky L.G., Borisova T.N., Carotti A.
Bioorganic and Medicinal Chemistry Letters,
2000
20.
![Investigation on the antiplatelet activity of pyrrolo[3,2-c]pyridine-containing compounds](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Voskressensky L.G., de Candia M., Carotti A., Borisova T.N., Kulikova L.N., Varlamov A.V., Altomare C.
Journal of Pharmacy and Pharmacology,
2003
21.
R. A. Smith, W.C. Wong, S.J. Olamovor, S. Choi, H.C. E. Kluender, Z. Zhang, R.C. Lavoie, J. Fan and B. L. Podlogar, WO Patent 03/027114A1, 2003.
22.
L. Cheng, US Patent 7576095B2, 2009.
23.

Trofimov B.A., Shapiro A.B., Nesterenko R.N., Mikhaleva A.I., Kalabin G.A., Golovanova N.I., Yakovleva I.V., Korostova S.E.
Chemistry of Heterocyclic Compounds,
1988
24.

Trofimov B.A., Vasil'tsov A.M., Mikhaleva A.I., Kalabin G.A., Shcherbakov V.V., Nesterenko R.N., Polubentsev E.A., Praliev K.D.
Chemistry of Heterocyclic Compounds,
1991
25.
10.1016/j.mencom.2019.11.018_sbref0070c
Varlamov
Synthesis,
2002
26.
![TANDEM MICHAEL ADDITION - HOFFMAN ELIMINATION SEQUENCE OF DMAD ON TETRAHYDROPYRROLO[3,2-C]PYRIDINES. NEW ROUTE TO VINYLPYRROLES .](/storage/images/resized/3SpVxcYL33bOvPq4sHxJLH2NeKNeDloahSUpNiO4_small_thumb.webp)
Varlamov A.V., Borisova T.N., Voskressensky L.G., Nsabimana B., Chernyshev A.I.
Heterocyclic Communications,
2001
27.
Borisova T.N., Voskressensky L.G., Soklakova T.A., Nsabimana B., Varlamov A.V.
Mendeleev Communications,
2002
28.
![The first synthesis and X-ray crystal structure of tetrahydropyrrolo[2,3-d]azocines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Varlamov A.V., Borisova T.N., Voskressensky L.G., Soklakova T.A., Kulikova L.N., Chernyshev A.I., Alexandrov G.G.
Tetrahedron Letters,
2002
29.

Borisova T.N., Voskressensky L.G., Soklakova T.A., Kulikova L.N., Varlamov A.V.
Molecular Diversity,
2000
30.
![Tandem transformations of tetrahydropyrrolo[3,2-c]pyridines under the action of dimethyl acetylenedicarboxylate. A novel route to pyrrolo[2,3-d]azocines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Voskressensky L.G., Borisova T.N., Vorob’eva T.A., Chernyshev A.I., Varlamov A.V.
Russian Chemical Bulletin,
2005
31.
![First Efficient One-Pot Synthesis of Tetrahydropyrrolo[2,3-d]azocines and Tetrahydroazocino[4,5-b]indoles](/storage/images/resized/doUaFUZdxUEQjLi1TwZjGHi8HXYNWWSk04dSC6Xh_small_thumb.webp)
Voskressensky L., Borisova T., Soklakova T., Kulikova L., Borisov R., Varlamov A.
Letters in Organic Chemistry,
2005
32.
![Transformations of 2-trifluoroacetyl-4,5,6,7-tetrahydro-1H-pyrrolo[3,2-c]pyridines by the action of ethyl propynoate. A novel synthesis of 2-trifluoroacetyl-4,7,8,9-tetrahydro-1H-pyrrolo[2,3-d]azocines](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Voskresenskii L.G., Borisova T.N., Vorob’eva T.A., Grishachkina O.V., Kulikova L.N., Chernyshev A.I., Varlamov A.V.
Russian Journal of Organic Chemistry,
2006
33.
10.1016/j.mencom.2019.11.018_sbref0075h
Voskressensky
2008
34.

Trofimov B.A., Stepanova Z.V., Sobenina L.N., Mikhaleva A.I., Ushakov I.A.
Tetrahedron Letters,
2004
35.
Trofimov B.A., Stepanova Z.V., Sobenina L.N., Mikhaleva A.I., Sinegovskaya L.M., Potekhin K.A., Fedyanin I.V.
Mendeleev Communications,
2005
36.
10.1016/j.mencom.2019.11.018_sbref0080c
Trofimov
2009
37.
Tomilin D.N., Soshnikov D.Y., Trofimov A.B., Gotsko M.D., Sobenina L.N., Ushakov I.A., Afonin A.V., Koldobsky A.B., Vitkovskaya N.M., Trofimov B.A.
Mendeleev Communications,
2016
38.

Yang Z., Zhang K., Gong F., Li S., Chen J., Ma J.S., Sobenina L.N., Mikhaleva A.I., Trofimov B.A., Yang G.
Journal of Photochemistry and Photobiology A: Chemistry,
2011