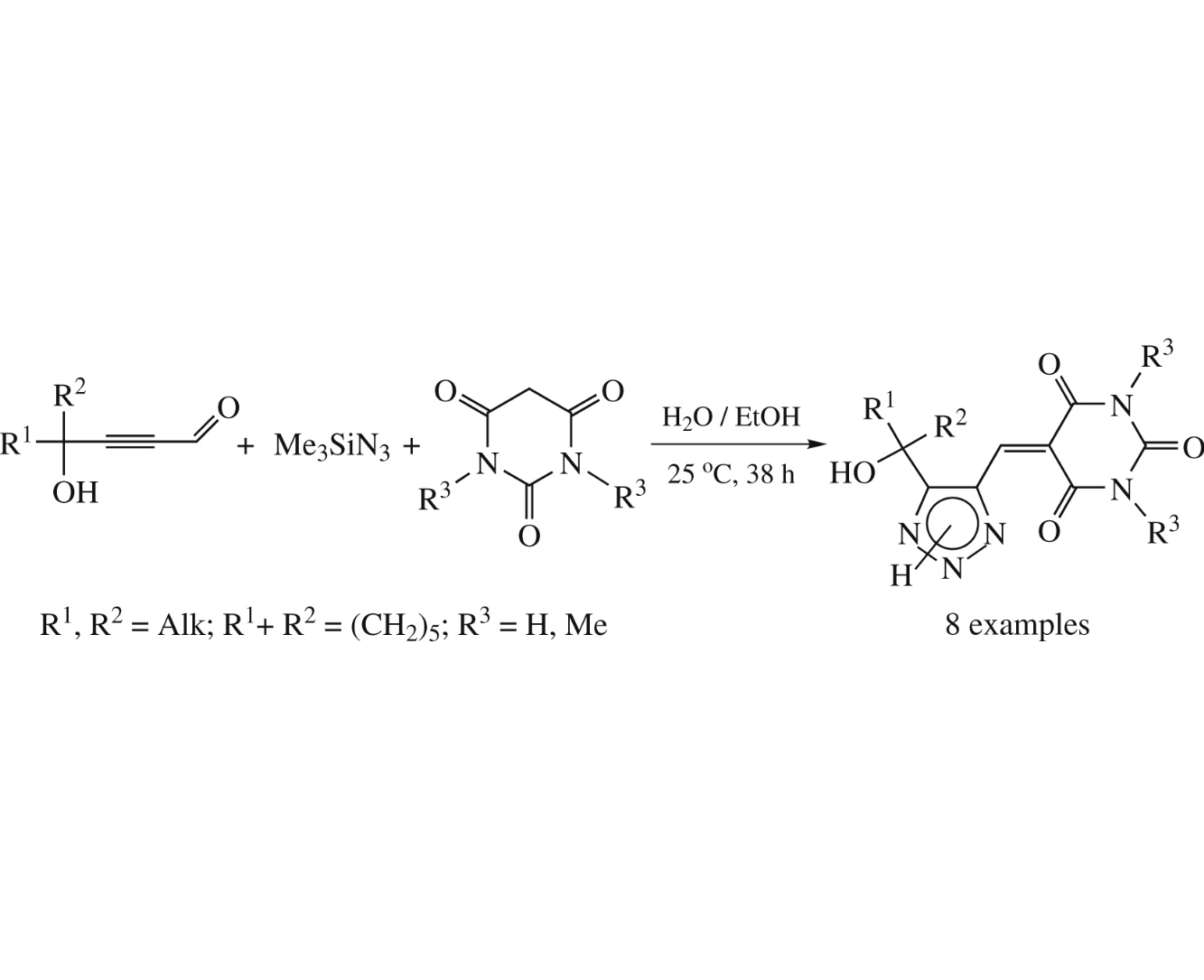
Abstract
5-[(5-Hydroxyalkyl-1H-1,2,3-triazol-4-yl)methylidene]barbiturates were eco-friendly synthesized in up to 89% yields by one-pot three-component processing (no catalyst, H2O–EtOH, room temperature, 38h) of 4-hydroxyalkynals, trimethylsilylazide and barbituric acids.
References
1.

Pirrung M.C., Sarma K.D.
Journal of the American Chemical Society,
2003
2.

Dömling A.
Chemical Reviews,
2005
3.

Ohno H., Ohta Y., Oishi S., Fujii N.
Angewandte Chemie - International Edition,
2007
4.

Yu J., Shi F., Gong L.
Accounts of Chemical Research,
2011
5.

Monfardini I., Huang J., Beck B., Cellitti J.F., Pellecchia M., Dömling A.
Journal of Medicinal Chemistry,
2011
6.

Dömling A., Wang W., Wang K.
Chemical Reviews,
2012
7.

Guo X., Hu W.
Accounts of Chemical Research,
2013
8.

Cioc R.C., Ruijter E., Orru R.V.
Green Chemistry,
2014
9.

Preeti P., Singh K.N.
Organic and Biomolecular Chemistry,
2018
10.

Sunderhaus J., Martin S.
Chemistry - A European Journal,
2009
11.

Jiang B., Rajale T., Wever W., Tu S., Li G.
Chemistry - An Asian Journal,
2010
12.
Gore R.P., Rajput A.P.
Drug Invention Today,
2013
13.

Mohammadi Ziarani G., Aleali F., Lashgari N.
RSC Advances,
2016
14.

Claudio Viegas-Junior, Eliezer J. Barreiro, Carlos Alberto Manssour Fraga
Current Medicinal Chemistry,
2007
15.

B. Tsogoeva S.
Mini-Reviews in Medicinal Chemistry,
2010
16.

Nepali K., Sharma S., Sharma M., Bedi P.M., Dhar K.L.
European Journal of Medicinal Chemistry,
2014
17.
10.1016/j.mencom.2019.11.017_sbref0020a
Garcia
Pharmacology and Physiology for Anesthesia: Foundations and Clinical Application,
2019
18.

Smith M.C., Riskin B.J.
Drugs,
1991
19.
10.1016/j.mencom.2019.11.017_sbref0020c
Lopez-Munoz
Neuropsychiatr. Dis. Treat.,
2005
20.

Siddiqui N., Ahsan W.
Archiv der Pharmazie,
2009
21.
![One-pot Synthesis of 5-{[Bis(azolylmethylthio)]methylene}pyrimidine-2,4,6-(1H,3H,5H)-triones](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Padmavathi V., Reddy G.D., Venkatesh B.C., Padmaja A.
Archiv der Pharmazie,
2010
22.
![Synthesis, characterization, and antimicrobial activities of barbital-based alkaline earth metal complexes: the X-ray crystal structure of [Ba2H(Barb)5] (Barb = 5,5-diethyl barbiturate)](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Ibrahim M.M., Al-Juaid S., Mohamed M.A., Yassin M.H.
Journal of Coordination Chemistry,
2012
23.

Mahmood Qureshi A., Qadir M., Rauf A., Idrees M., Mumtaz S., Najam-ul-Haq M., Ismail M., Athar M., Khushal R., Riaz S., Bokhari H.
Letters in Drug Design and Discovery,
2011
24.

Yan Q., Cao R., Yi W., Chen Z., Wen H., Ma L., Song H.
European Journal of Medicinal Chemistry,
2009
25.
10.1016/j.mencom.2019.11.017_sbref0030a
Demopoulos
Acta Neurol. Scand.,
1977
26.

Khan K.M., Ali M., Ajaz A., Perveen S., Choudhary M.I., Atta-ur-Rahman
Letters in Drug Design and Discovery,
2008
27.

Sokmen B.B., Ugras S., Sarikaya H.Y., Ugras H.I., Yanardag R.
Applied Biochemistry and Biotechnology,
2013
28.

Almaas R., Saugstad O., Pleasure D., Rootwelt T.
Anesthesiology,
2000
29.
10.1016/j.mencom.2019.11.017_sbref0030e
Moon
Oncotarget,
2017
30.

Harriman G.C., Brewer M., Bennett R., Kuhn C., Bazin M., Larosa G., Skerker P., Cochran N., Gallant D., Baxter D., Picarella D., Jaffee B., Luly J.R., Briskin M.J.
Bioorganic and Medicinal Chemistry Letters,
2008
31.

Biradar J.S., Sasidhar B.S., Parveen R.
European Journal of Medicinal Chemistry,
2010
32.

Ivanova B.B., Spiteller M.
Crystal Growth and Design,
2010
33.
![Erratum: Nonlinear optical properties of p‐substituted benzalbarbituric acids [Appl. Phys. Lett. 56, 718 (1990)]](/storage/images/resized/ARM4e6URKRsbRZvIF0vFis9DjxGloBjnBYJXbHmZ_small_thumb.webp)
Kondo K., Ochiai S., Takemoto K., Irie M.
Applied Physics Letters,
1990
34.

Fırıncı E., Giziroglu E., Celepci D.B., Söyleyici H.C., Aygün M.
Journal of Molecular Structure,
2017
35.

Mahmudov K.T., Kopylovich M.N., Maharramov A.M., Kurbanova M.M., Gurbanov A.V., Pombeiro A.J.
Coordination Chemistry Reviews,
2014
36.

Agalave S.G., Maujan S.R., Pore V.S.
Chemistry - An Asian Journal,
2011
37.
10.1016/j.mencom.2019.11.017_sbref0055b
Haider
Inflammation Cell Signaling,
2014
38.

Dheer D., Singh V., Shankar R.
Bioorganic Chemistry,
2017
39.

de Carvalho da Silva F., Cardoso M.F., Ferreira P.G., Ferreira V.F.
Topics in Heterocyclic Chemistry,
2014
40.
M. J. Orocycl. Chem. FerreiranUS Patent 5064844, 1991.
41.
![Synthesis, Crystal Structure and Biological Activities ofO,O-Dialkyl α-[1-(2-Chlorothiazol-5-ylmethyl)-5-methyl- 1H-1,2,3-triazol-4-ylcarbonyloxy]alkylphosphonates](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
CHEN X., SHI D., ZHU X.
Chinese Journal of Chemistry,
2007
42.

Dai Z., Chen Y., Zhang M., Li S., Yang T., Shen L., Wang J., Qian S., Zhu H., Ye Y.
Organic and Biomolecular Chemistry,
2015
43.

Costa A.V., Oliveira M.V., Pinto R.T., Moreira L.C., Gomes E.M., Alves T.D., Pinheiro P.F., Queiroz V.T., Vieira L.F., Teixeira R.R., Júnior W.C.
Molecules,
2017
44.
Quraishi M.A., Sardar R.
Corrosion,
2002
45.

Quraishi M.A., Sudheer, Ansari K.R., Ebenso E.E.
International Journal of Electrochemical Science,
2012
46.
10.1016/j.mencom.2019.11.017_sbref0065c
Desai
Int. J. Curr. Microbiol. Appl. Sci.,
2015
47.

Ofoegbu S.U., Galvão T.L., Gomes J.R., Tedim J., Nogueira H.I., Ferreira M.G., Zheludkevich M.L.
Physical Chemistry Chemical Physics,
2017
48.
10.1016/j.mencom.2019.11.017_sbref0065e
Resende
Int. J. Electrochem.,
2019
49.

Duan T., Fan K., Fu Y., Zhong C., Chen X., Peng T., Qin J.
Dyes and Pigments,
2012
50.

Khanmohammadi H., Erfantalab M., Bayat A., Babaei A., Sohrabi M.
Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy,
2012
51.
![Thiacalix[4]arene 1,2,3-triazole-polyethylene glycol polymers: Synthesis and dye adsorption properties](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Zhu S., Guo H., Yang F., Wang Z.
Chinese Chemical Letters,
2015
52.
10.1016/j.mencom.2019.11.017_bib0075
Willis
Eur. Pat. Appl.,
1990
53.
10.1016/j.mencom.2019.11.017_sbref0080a
Chandrasekaran
Click Reactions in Organic Synthesis,
2016
54.

M. Heravi M., Tamimi M., Yahyavi H., Hosseinnejad T.
Current Organic Chemistry,
2015
55.

Lin S., Sharma A.
Chemistry of Heterocyclic Compounds,
2018
56.
Muzalevskiy V.M., Mamedzade M.N., Chertkov V.A., Bakulev V.A., Nenajdenko V.G.
Mendeleev Communications,
2018
57.

Karsakova I.V., Smirnov A.Y., Baranov M.S.
Chemistry of Heterocyclic Compounds,
2018
58.
10.1016/j.mencom.2019.11.017_bib0085
Novokshonova
Synthesis,
2008
59.

Medvedeva A.S., Demina M.M., Nguyen T.L., Vu T.D., Bulanov D.A., Novokshonov V.V.
Russian Journal of Organic Chemistry,
2013
60.
Medvedeva A.S., Demina M.M., Vu T.D., Andreev M.V., Shaglaeva N.S., Larina L.I.
Mendeleev Communications,
2016
61.
![One-pot three-component green synthesis of [1H-(1,2,3-triazol-5-yl)methylidene] heterocycles based on element-substituted propynals](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Demina M.M., Medvedeva A.S., Nguyen T.L., Vu T.D., Larina L.I.
Russian Chemical Bulletin,
2017
62.

Medvedeva A.S., Mitroshina I.V., Afonin A.V., Chernyshev K.A., Bulanov D.A., Mareev A.V.
Russian Journal of Organic Chemistry,
2013
63.

Medvedeva A.S., Safronova L.P., Chichkareva G.G., Voronkov M.G.
Russian Chemical Bulletin,
1976