Abstract
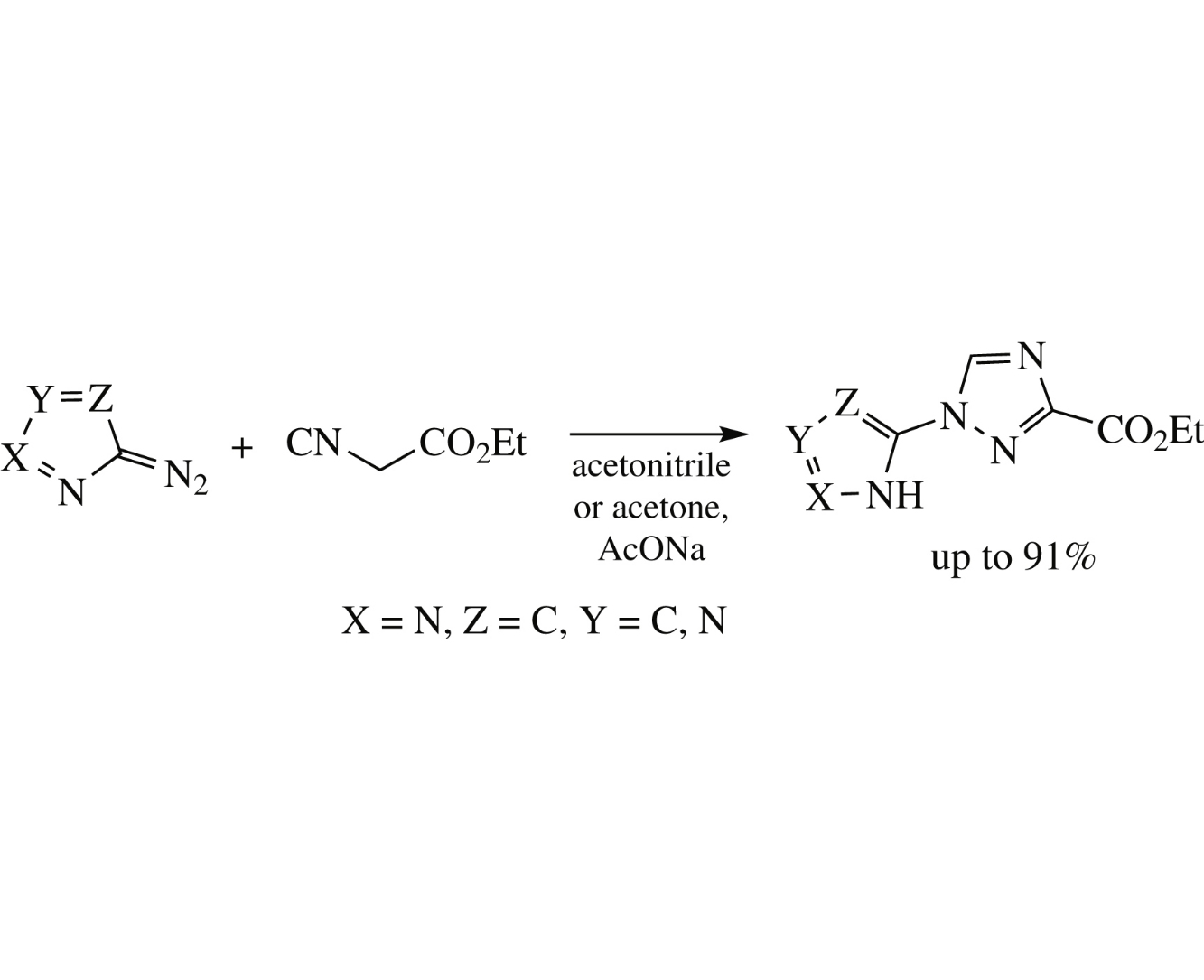
PASE (pot, atom and step economic) synthesis of 1-azolyl-1H-1,2,4-triazole derivatives in up to 91% yield has been accomplished by addition of 5-diazoazoles to ethyl isocyanoacetate.
References
1.

Ansari A., Ali A., Asif M., Shamsuzzaman S.
New Journal of Chemistry,
2017
2.
10.1016/j.mencom.2019.11.016_bib0010
Luca
Curr. Med. Chem.,
2006
3.

Al-Masoudi I.A., Al-Soud Y.A., Al-Salihi N.J., Al-Masoudi N.A.
Chemistry of Heterocyclic Compounds,
2006
4.

Sathish Kumar S., P. Kavitha H.
Mini-Reviews in Organic Chemistry,
2013
5.

Maddila S., Pagadala R., Jonnalagadda S.
Letters in Organic Chemistry,
2013
6.

Ahmadi F., Ghayahbashi M., Sharifzadeh M., Alipoiur E., Ostad S., Vosooghi M., khademi H., Amini M.
Medicinal Chemistry,
2014
7.

Küçükgüzel Ş.G., Çıkla-Süzgün P.
European Journal of Medicinal Chemistry,
2015
8.

Shalini K., Kumar N., Drabu S., Sharma P.K.
Beilstein Journal of Organic Chemistry,
2011
9.
10.1016/j.mencom.2019.11.016_bib0045
Bulut
Bull. Chem. Soc. Ethiop.,
2010
10.

Diaz-Ortiz A., Prieto P., R. Carrillo J., Martin R., Torres I.
Current Organic Chemistry,
2015
11.

Aromí G., Barrios L.A., Roubeau O., Gamez P.
Coordination Chemistry Reviews,
2011
12.

F. V. Scriven E., Balasubramanian M., G. Keay J., Shobana N.
Heterocycles,
1994
13.

Moulin A., Bibian M., Blayo A., El Habnouni S., Martinez J., Fehrentz J.
Chemical Reviews,
2010
14.

Holm S.C., Straub B.F.
Organic Preparations and Procedures International,
2011
15.
Kaur P., Kaur R., Goswami M.
International Research Journal of Pharmacy,
2018
16.

MATSUMOTO K., SUZUKI M., TOMIE M., YONEDA N., MIYOSHI M.
Synthesis,
1975
17.

Gulevich A.V., Zhdanko A.G., Orru R.V., Nenajdenko V.G.
Chemical Reviews,
2010
18.

Van Leusen A.M., Hoogenboom B.E., Houwing H.A.
Journal of Organic Chemistry,
1976
19.
![Flow synthesis of ethyl isocyanoacetate enabling the telescoped synthesis of 1,2,4-triazoles and pyrrolo-[1,2-c]pyrimidines](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Baumann M., Rodriguez Garcia A.M., Baxendale I.R.
Organic and Biomolecular Chemistry,
2015
20.
![[3 + 2] Cycloaddition of Isocyanides with Aryl Diazonium Salts: Catalyst-Dependent Regioselective Synthesis of 1,3- and 1,5-Disubstituted 1,2,4-Triazoles](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Liu J., Shen X., Wang Y., Wang X., Bi X.
Organic Letters,
2018
21.
10.1016/j.mencom.2019.11.016_bib0105
Mokrushin
Khimiya geterotsiklicheskikh diazosoedinenii (Chemistry of Heterocyclic Diazo Compounds),
2013
22.
![Synthesis of 6,8-substituted derivatives of imidazo[5,1-c][1,2,4]triazines and 1,4-dihydroimidazo[5,1-c][1,2,4]triazin-4-ones](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Bezmaternykh M.A., Mokrushin V.S., Pospelova T.A., El'tsov O.S.
Chemistry of Heterocyclic Compounds,
1998
23.

Shchegol’kov E.V., Sadchikova E.V., Burgart Y.V., Saloutin V.I.
Russian Chemical Bulletin,
2008
24.
![Interaction of 3,8-Disubstituted Imidazo-[5,1-C][1,2,4]Triazines with Nucleophiles**](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Sadchikova E.V., Mokrushin* V.S.
Chemistry of Heterocyclic Compounds,
2014
25.
![Synthesis and structure of 4-hydroxy-4-fluoroalkyl-1,4-dihydroimidazo[5,1-c][1,2,4]triazines](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Shchegol’kov E.V., Sadchikova E.V., Burgart Y.V., Saloutin V.I.
Russian Journal of Organic Chemistry,
2009
26.

Obrecht R., Herrmann R., Ugi I.
Synthesis,
1985
27.
![Synthesis and properties of 5-aryl-3-diazo-3H-pyrazoles and 3-aryl-1H-pyrazole-5-diazonium salts. Preparation and cytolytic activity studies of 2-arylpyrazolo-[5,1-c][1,2,4]benzotriazines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Alekseeva D.L., Rakhimova V.Y., Minin A.S., Belousova A.V., Sadchikova E.V.
Chemistry of Heterocyclic Compounds,
2018
28.

Fulmer Shealy Y., Allen O'dell C.
Journal of Pharmaceutical Sciences,
1971
29.

Andersen K.E., Pedersen E.B.
European Journal of Organic Chemistry,
1986
30.

Sadchikova E.V., Mokrushin V.S.
Russian Chemical Bulletin,
2003
31.
![Pyrazoles I. Synthesis of 4-Hydroxpyrazolo[3,4-d]-v-triazine A New Analog of Hypoxanthine](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Cheng C.C., Robins R.K., Cheng K.C., Lin D.C.
Journal of Pharmaceutical Sciences,
1968
32.

Stevens M.F.
Progress in Medicinal Chemistry,
1976
33.

SHEALY Y.F., STRUCK R.F., HOLUM L.B., MONTGOMERY J.A.
Journal of Organic Chemistry,
1961