Abstract
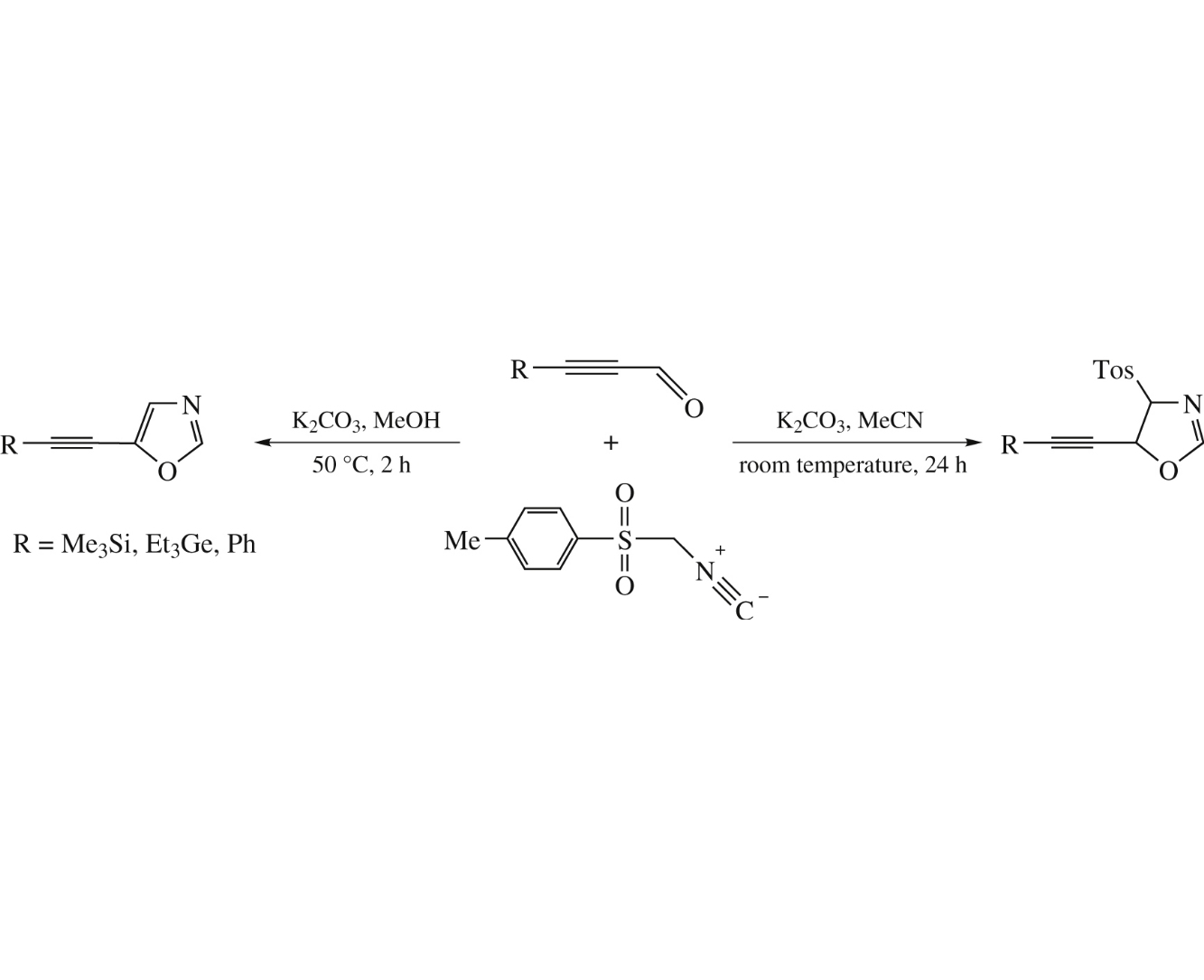
The reaction of 3-(trimethylsilyl)propynal, 3-(triethylgermyl) propynal or 3-phenylpropynal with tosylmethyl isocyanide affords new 5-alkynyl-4-tosyl-2-oxazolines and/or 5-alkynyl-1,3-oxazoles.
References
1.
S. L. Gwaltney, II, H.-S. Jae, D.M. Kalvin, G. Liu, H.L. Sham, Q. Li, A.K. Claiborne, L. Wang, K.J. Barr and K. W. Woods, WO Patent 00/06556, 2000
2.

Bergeron R.J., Xin M.G., Weimar W.R., Smith R.E., Wiegand J.
Journal of Medicinal Chemistry,
2001
3.

Beloglazkina A.А., Skvortsov D.А., Tafeenko V.A., Majouga А.G., Zyk N.V., Beloglazkina Е.К.
Russian Chemical Bulletin,
2018
4.

Tsuda M., Yamakawa M., Oka S., Tanaka Y., Hoshino Y., Mikami Y., Sato A., Fujiwara H., Ohizumi Y., Kobayashi J.
Journal of Natural Products,
2005
5.

Thiede S., Wosniok P.R., Herkommer D., Debnar T., Tian M., Wang T., Schrempp M., Menche D.
Chemistry - A European Journal,
2016
6.

Yeh V.S.
Tetrahedron,
2004
7.

Nicolaou K.C., Lizos D.E., Kim D.W., Schlawe D., de Noronha R.G., Longbottom D.A., Rodriquez M., Bucci M., Cirino G.
Journal of the American Chemical Society,
2006
8.

Wipf P.
Chemical Reviews,
1995
9.

Chandrasekhar S., Sudhakar A.
Organic Letters,
2009
10.

Kakkar S., Narasimhan B.
BMC Chemistry,
2019
11.
10.1016/j.mencom.2019.11.015_sbref0005k
Bansal
Int. J. Pharm. Sci. Res.,
2014
12.

Gong Z., Leu C., Wu F., Shu C.
Macromolecules,
2000
13.
Tarasenko E.A., Beletskaya I.P.
Mendeleev Communications,
2016
14.

Wang Y., Yang G., Xie F., Zhang W.
Organic Letters,
2018
15.

Hargaden G.C., Guiry P.J.
Chemical Reviews,
2009
16.

Mazuela J., Tolstoy P., Pàmies O., Andersson P.G., Diéguez M.
Organic and Biomolecular Chemistry,
2011
17.
Baranov D.S., Ryabichev A.A., Mamatyuk V.I., Gatilov Y.V., Kartsev V.G., Vasilevsky S.F.
Mendeleev Communications,
2012
18.

Liu G., Yang C., Li Y.
Journal of Organic Chemistry,
2015
19.

Mollo M.C., Orelli L.R.
Organic Letters,
2016
20.

Carlucci C., Tota A., Colella M., Ronamazzi G., Clarkson G.J., Luisi R., Degennaro L.
Chemistry of Heterocyclic Compounds,
2018
21.
10.1016/j.mencom.2019.11.015_sbref0020e
Abraham
Arabian J. Chem.,
2018
22.

Kulkarni B.A., Ganesan A.
Tetrahedron Letters,
1999
23.

Wan C., Gao L., Wang Q., Zhang J., Wang Z.
Organic Letters,
2010
24.

Keiko N.A., Vchislo N.V.
Asian Journal of Organic Chemistry,
2016
25.

van Leusen A.M., Hoogenboom B.E., Siderius H.
Tetrahedron Letters,
1972
26.
10.1016/j.mencom.2019.11.015_bib0030
Wu
Synlett,
2009
27.
![One-pot three-component green synthesis of [1H-(1,2,3-triazol-5-yl)methylidene] heterocycles based on element-substituted propynals](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Demina M.M., Medvedeva A.S., Nguyen T.L., Vu T.D., Larina L.I.
Russian Chemical Bulletin,
2017
28.

Bulanov D.A., Novokshonova I.A., Safronova L.P., Ushakov I.A., Medvedeva A.S.
Tetrahedron Letters,
2016