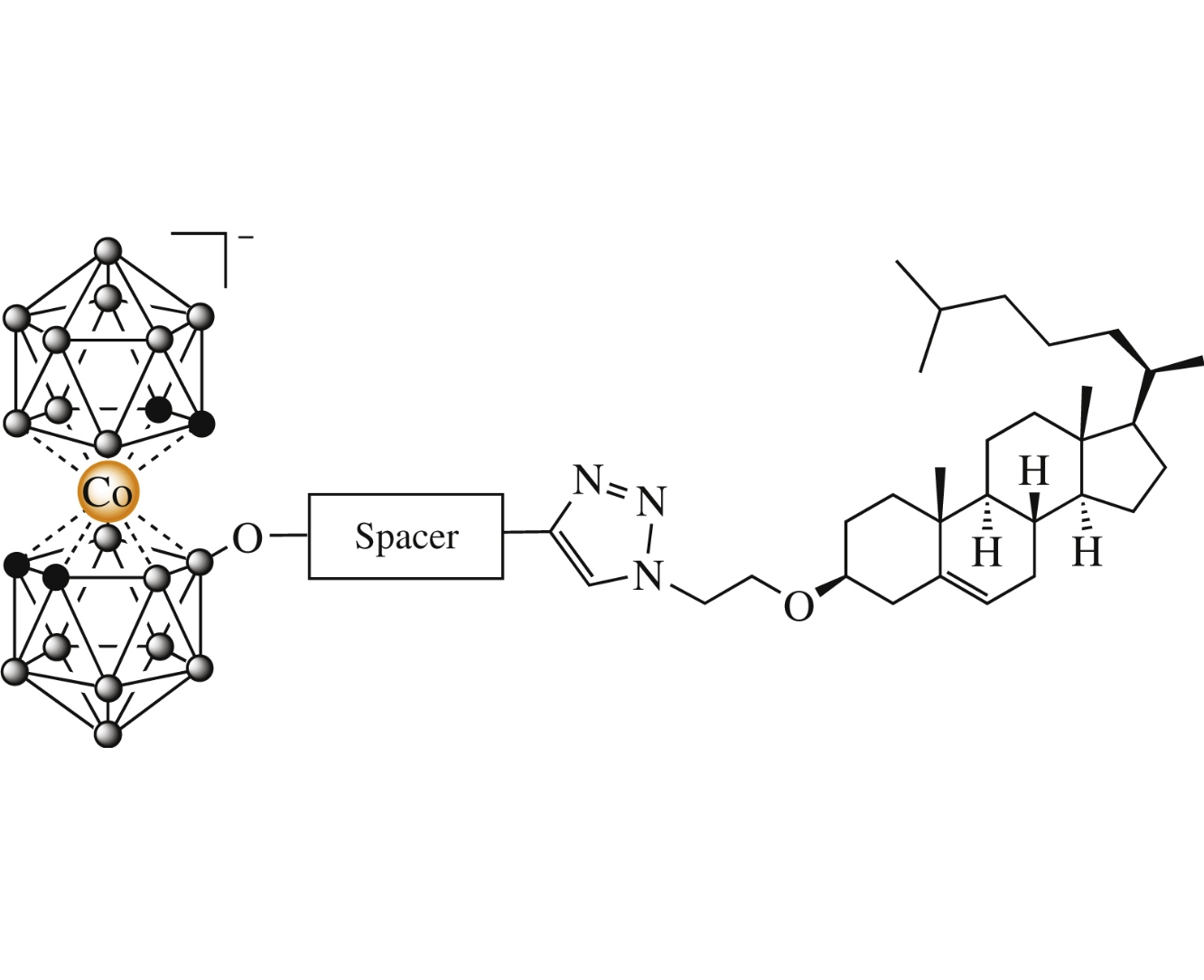
Abstract
New conjugates of cholesterol and cobalt bis(dicarbollide) were synthesized using ‘click’ cycloaddition of 3b-(2-azidoethyl) cholest-5-ene and acetylene-equipped cobalt bis(dicarbollides). The synthesized boronated cholesterols can be of interest for liposomal drug delivery for boron neutron capture therapy of cancer.
References
1.
2.

Soloway A.H., Tjarks W., Barnum B.A., Rong F., Barth R.F., Codogni I.M., Wilson J.G.
Chemical Reviews,
1998
3.
10.1016/j.mencom.2019.11.007_bib0015
Florence
Liposomes in Drug Delivery,
1993
4.

Pattni B.S., Chupin V.V., Torchilin V.P.
Chemical Reviews,
2015
5.

Olusanya T., Haj Ahmad R., Ibegbu D., Smith J., Elkordy A.
Molecules,
2018
6.

Noble C.O., Kirpotin D.B., Hayes M.E., Mamot C., Hong K., Park J.W., Benz C.C., Marks J.D., Drummond D.C.
Expert Opinion on Therapeutic Targets,
2004
7.

Noble G.T., Stefanick J.F., Ashley J.D., Kiziltepe T., Bilgicer B.
Trends in Biotechnology,
2014
8.
Koloskova O.O., Nosova A.S., Shchelik I.S., Shilovskiy I.P., Sebyakin Y.L., Khaitov M.R.
Mendeleev Communications,
2017
9.
10.1016/j.mencom.2019.11.007_bib0045
Nakamura
Boron Science: New Technologies and Applications,
2012
10.

Nakamura H.
Future Medicinal Chemistry,
2013
11.
V. I. Bregadze, I.B. Sivaev, A. Semioshkin, A.V. Shmal’ko, I.D. Kosenko, K.V. Lebedeva, S. Mandal, P. Sreejyothi, R.D. Dubey, A. Sarkar, Z. Li, Z. Shen, A. Wu and N. S. Hosmane, Chem. Eur. J., submitted.
12.
![Metallacarboranes on the Road to Anticancer Therapies: Cellular Uptake, DNA Interaction, and Biological Evaluation of Cobaltabisdicarbollide [COSAN]−](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Fuentes I., García-Mendiola T., Sato S., Pita M., Nakamura H., Lorenzo E., Teixidor F., Marques F., Viñas C.
Chemistry - A European Journal,
2018
13.
Sivaev I.B., Bregadze V.I.
Collection of Czechoslovak Chemical Communications,
1999
14.

Dash B.P., Satapathy R., Swain B.R., Mahanta C.S., Jena B.B., Hosmane N.S.
Journal of Organometallic Chemistry,
2017
15.

Hawthorne M.F., Young D.C., Andrews T.D., Howe D.V., Pilling R.L., Pitts A.D., Reintjes M., Warren L.F., Wegner P.A.
Journal of the American Chemical Society,
1968
16.

Kolb H.C., Sharpless K.B.
Drug Discovery Today,
2003
17.

Moses J.E., Moorhouse A.D.
Chemical Society Reviews,
2007
18.

Tron G.C., Pirali T., Billington R.A., Canonico P.L., Sorba G., Genazzani A.A.
Medicinal Research Reviews,
2008
19.

Haldón E., Nicasio M.C., Pérez P.J.
Organic and Biomolecular Chemistry,
2015
20.

Zhang X., Yang X., Zhang S.
Synthetic Communications,
2009
21.

Wojtczak B., Andrysiak A., Grüner B., Lesnikowski Z.
Chemistry - A European Journal,
2008
22.

Kosenko I.D., Lobanova I.A., Chekulaeva L.A., Godovikov I.A., Bregadze V.I.
Russian Chemical Bulletin,
2013
23.
10.1016/j.mencom.2019.11.007_bib0095
Shmal’ko
Inorg. Lett.,
2015
24.

Kosenko I.D., Lobanova I.A., Ananyev I.V., Godovikov I.A., Chekulaeva L.A., Starikova Z.A., Qi S., Bregadze V.I.
Journal of Organometallic Chemistry,
2014