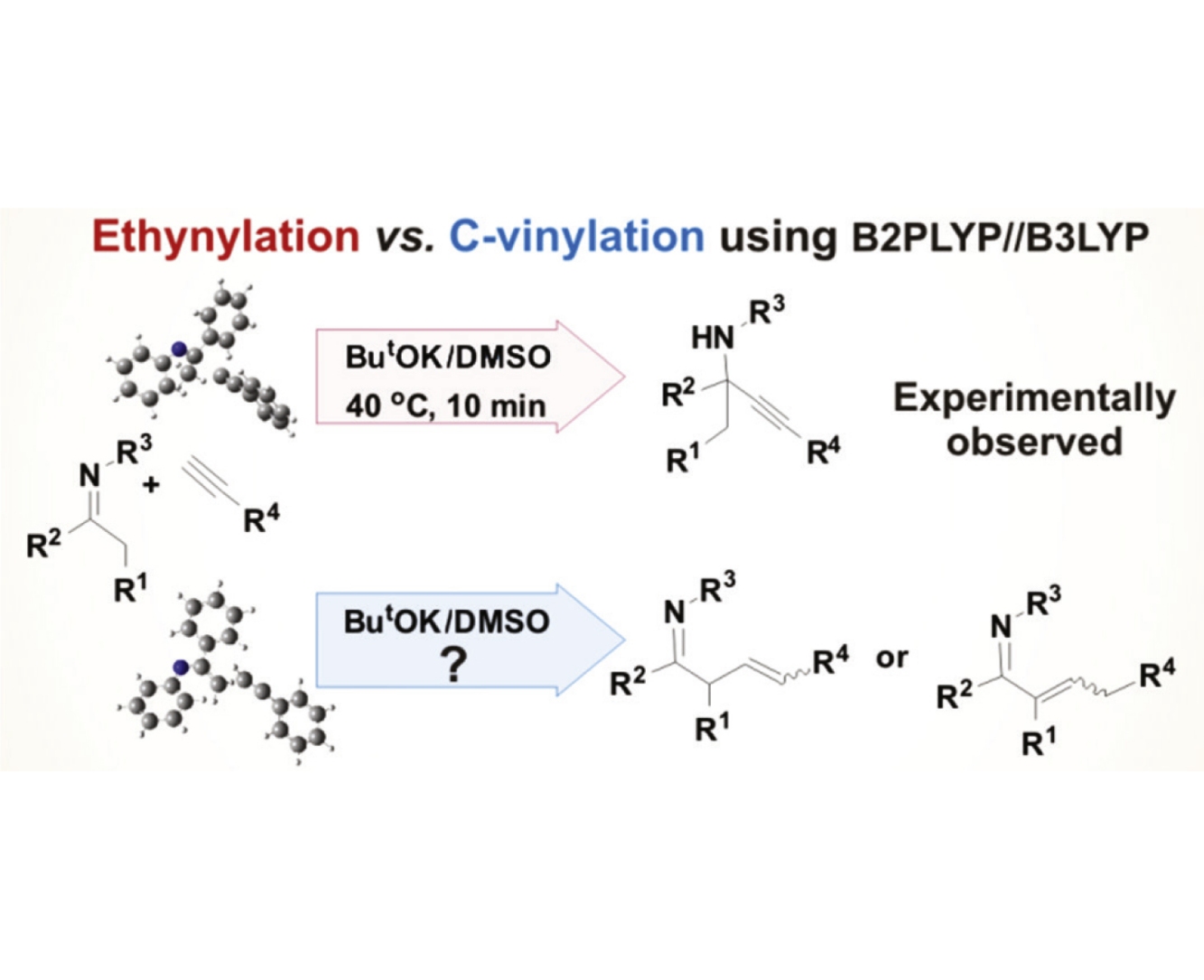
Abstract
The kinetic (free activation energies) and thermodynamic factors, which ensure mild conditions for the ethynylation of imines with acetylenes, have been established by quantum chemical calculations using B2PLYP/6-311+G**//B3LYP/6-31+G*. According to the calculations, the alternative route of superbase catalyzed C-vinylation requires harsher reaction conditions as compared to the ethynylation one.
References
1.

Lauder K., Toscani A., Scalacci N., Castagnolo D.
Chemical Reviews,
2017
2.

Bidusenko I.A., Schmidt E.Y., Ushakov I.A., Trofimov B.A.
European Journal of Organic Chemistry,
2018
3.

Trofimov B.A., Schmidt E.Y.
Accounts of Chemical Research,
2018
4.

Kobychev V.B., Orel V.B., Zankov D.V., Vitkovskaya N.M., Trofimov B.A.
Russian Chemical Bulletin,
2017
5.

Becke A.D.
Physical Review A,
1988
6.

Lee C., Yang W., Parr R.G.
Physical Review B,
1988
7.

Becke A.D.
Journal of Chemical Physics,
1993
8.

Grimme S.
Journal of Chemical Physics,
2006
9.
![Two classes of heterocycles-6,8-dioxabicyclo[3.2.1]octanes and cyclopentenols from the same reagents: A quantum-chemical comparison of mechanism](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Vitkovskaya N.M., Orel V.B., Kobychev V.B., Schmidt E.Y., Trofimov B.A.
International Journal of Quantum Chemistry,
2018
10.

Tomasi J., Mennucci B., Cancès E.
Journal of Molecular Structure THEOCHEM,
1999
11.

Wertz D.H.
Journal of the American Chemical Society,
1980
12.

Vitkovskaya N.M., Kobychev V.B., Bobkov A.S., Orel V.B., Schmidt E.Y., Trofimov B.A.
Journal of Organic Chemistry,
2017
13.
10.1016/j.mencom.2019.11.005_bib0065
Frisch
Gaussian 09, Revision C.01,
2009
14.

Bordwell F.G., Algrim D.J.
Journal of the American Chemical Society,
1988
15.

Olmstead W.N., Margolin Z., Bordwell F.G.
Journal of Organic Chemistry,
1980
16.

Bordwell F.G., Drucker G.E., Andersen N.H., Denniston A.D.
Journal of the American Chemical Society,
1986
17.

18.

Seeman J.I.
Journal of Chemical Education,
1986
19.

Kobychev V.B., Vitkovskaya N.M., Orel V.B., Schmidt E.Y., Trofimov B.A.
Russian Chemical Bulletin,
2015