Abstract
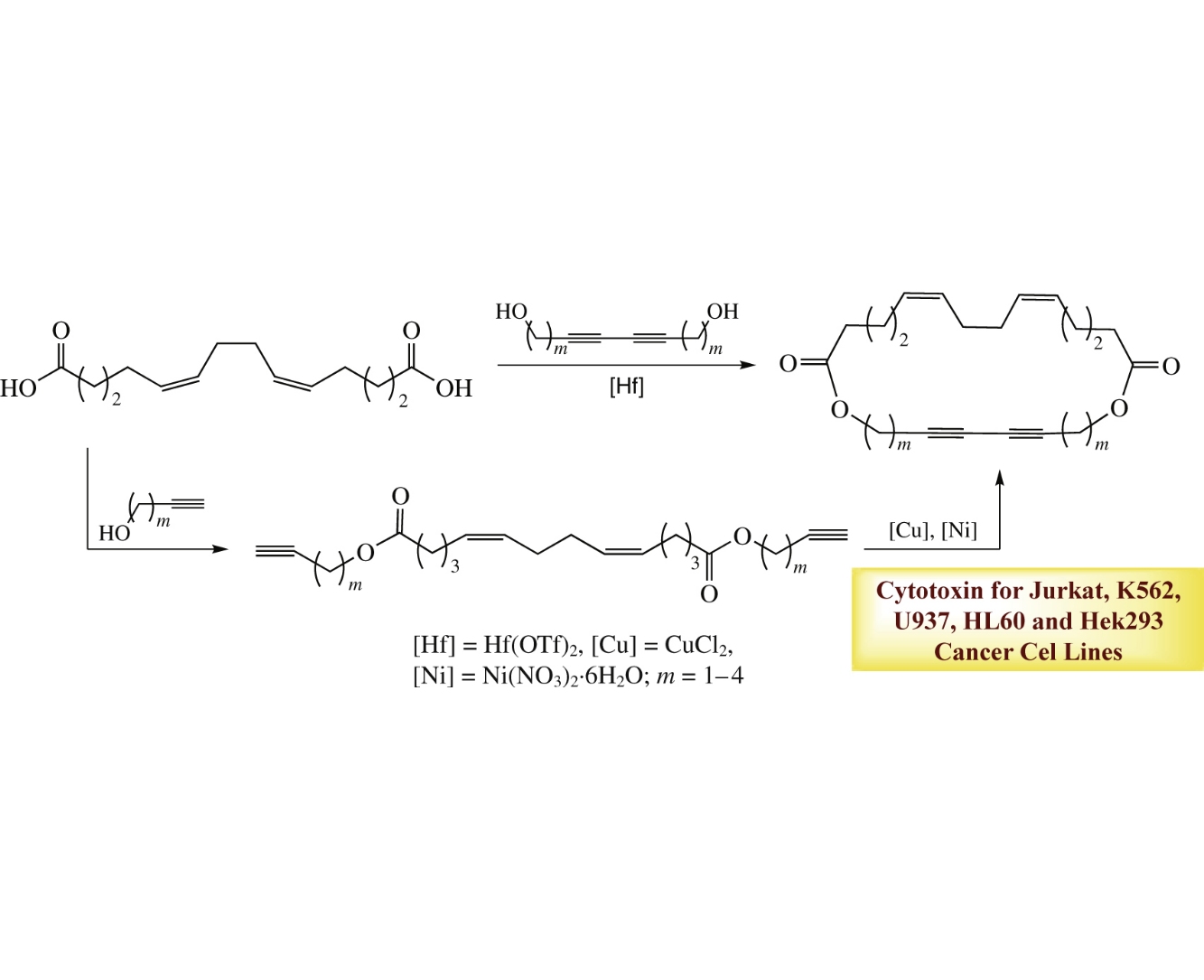
New macrodiolides incorporating 1Z,5Z-diene and 1,3-diyne moieties were obtained in 55–79% yields and with >98% stereoselectivity by Hf(OTf)4-catalyzed intermolecular cyclocondesation of (5Z,9Z)-tetradeca-5,9-diene-1,14-dioic acid with a,ω-diols. Alternative route included intramolecular oxidative coupling of a,ω-diynes, esterification products of (5Z,9Z)-tetradeca-5,9-diene-1,14-dioic acid with alkynols. The macrodiolides synthesized exhibit in vitro cytotoxic activity toward Jurkat, K562, U937, HL-60, HeLa and Hek293 cell lines.
References
1.

Driggers E.M., Hale S.P., Lee J., Terrett N.K.
Nature Reviews Drug Discovery,
2008
2.

Yudin A.K.
Chemical Science,
2015
3.

Parenty A., Moreau X., Campagne J.-.
Chemical Reviews,
2006
4.

Li Y., Yin X., Dai M.
Natural Product Reports,
2017
5.

Shi W., Lei A.
Tetrahedron Letters,
2014
6.

Verlinden S., Ballet S., Verniest G.
European Journal of Organic Chemistry,
2016
7.

Zhang B., Wang Y., Yang S., Zhou Y., Wu W., Tang W., Zuo J., Li Y., Yue J.
Journal of the American Chemical Society,
2012
8.

Wang Y., Liu Q., Xue J., Zhou Y., Yu H., Yang S., Zhang B., Zuo J., Li Y., Yue J.
Organic Letters,
2014
9.

Schaubach S., Gebauer K., Ungeheuer F., Hoffmeister L., Ilg M.K., Wirtz C., Fürstner A.
Chemistry - A European Journal,
2016
10.

Mohapatra D.K., Umamaheshwar G., Rao R.N., Rao T.S., R S.K., Yadav J.S.
Organic Letters,
2015
11.

Agbedahunsi J.M., Fakoya F.A., Adesanya S.A.
Phytomedicine,
2004
12.
10.1016/j.mencom.2019.11.002_bib0060
Corey
Molecules and Medicine,
2007
13.

D'yakonov V.A., Islamov I.I., Dzhemileva L.U., Khusainova E.M., Yunusbaeva M.M., Dzhemilev U.M.
Tetrahedron,
2018
14.
D’yakonov V.A., Islamov I.I., Khusainova E.M., Dzhemilev U.M.
Mendeleev Communications,
2018
15.

Dzhemilev U.M., Ibragimov A.G., D’yakonov V.A., Pudas M., Bergmann U., Khafizova L.O., Tyumkina T.V.
Russian Journal of Organic Chemistry,
2007
16.

Abakumov G.A., Piskunov A.V., Cherkasov V.K., Fedushkin I.L., Ananikov V.P., Eremin D.B., Gordeev E.G., Beletskaya I.P., Averin A.D., Bochkarev M.N., Trifonov A.A., Dzhemilev U.M., D'yakonov V.A., Egorov M.P., Vereshchagin A.N., et. al.
Russian Chemical Reviews,
2018
17.

D’yakonov V.A., Dzhemileva L.U., Tuktarova R.A., Makarov A.A., Islamov I.I., Mulyukova A.R., Dzhemilev U.M.
Steroids,
2015
18.

de Léséleuc M., Collins S.K.
Chemical Communications,
2015
19.

Bédard A., Collins S.K.
Journal of the American Chemical Society,
2011
20.
10.1016/j.mencom.2019.11.002_bib0100
Levy
Carbon-13 Nuclear Magnetic Resonance for Organic Chemists,
1972