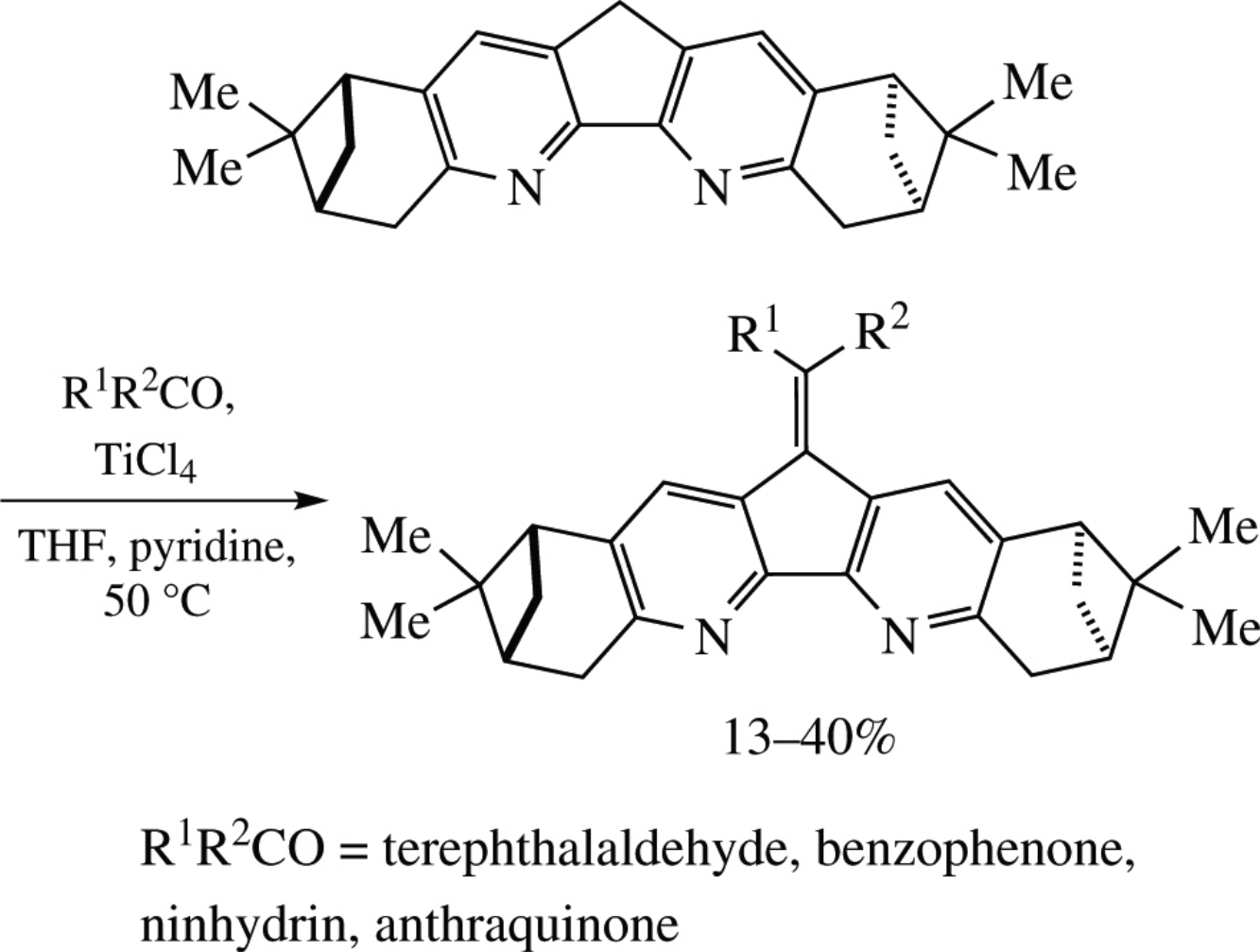
Abstract
Reaction of fused 4,5-diazafluorene–bis(nopinane) with tere-phthalaldehyde, benzophenone, ninhydrin or anthraquinone in THF in the presence of TiCl4 and pyridine results in a formation of new chiral condensation products, which exhibit solvatochromism and pleochroism. Crystal structure of the key products has been determined by X-ray crystallography, and unusual colour behavior has been explained by DFT calculations.
References
1.

Annibale V.T., Song D.
Dalton Transactions,
2016
2.

Li H., Wang J., Zhang S., Gong C., Wang F.
RSC Advances,
2018
3.

Sabithakala T., Chittireddy V.R.
Applied Organometallic Chemistry,
2018
4.

Jäger S., Gude L., Arias-Pérez M.
Bioorganic Chemistry,
2018
5.

Wezenberg S.J., Chen K., Feringa B.L.
Angewandte Chemie - International Edition,
2015
6.

Faulkner A., van Leeuwen T., Feringa B.L., Wezenberg S.J.
Journal of the American Chemical Society,
2016
7.

Peng Y., Hu B., Huang W.
Tetrahedron,
2018
8.

Levy A., Pogodin S., Cohen S., Agranat I.
European Journal of Organic Chemistry,
2007
9.

Sako K., Kakehi T., Nakano S., Oku H., Shen X.F., Iwanaga T., Yoshikawa M., Sugahara K., Toyota S., Takemura H., Shinmyozu T., Shiotsuka M., Tatemitsu H.
Tetrahedron Letters,
2014
10.

Sako K., Mugishima Y., Iwanaga T., Toyota S., Takemura H., Watanabe M., Shinmyozu T., Shiotsuka M., Tatemitsu H.
Tetrahedron Letters,
2011
11.
Vasilyev E.S., Bagryanskaya I.Y., Tkachev A.V.
Mendeleev Communications,
2017
12.

Vasilyev E.S., Agafontsev A.M., Tkachev A.V.
Synthetic Communications,
2014
13.

Mamula O., Bark T., Quinodoz B., Stoeckli-Evans H., von Zelewsky A.
Inorganica Chimica Acta,
2018
14.

Solea A.B., Wohlhauser T., Abbasi P., Mongbanziama Y., Crochet A., Fromm K.M., Novitchi G., Train C., Pilkington M., Mamula O.
Dalton Transactions,
2018
15.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
16.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
17.

Hirao Y., Nagamachi N., Hosoi K., Kubo T.
Chemistry - An Asian Journal,
2018
18.

Biedermann P.U., Stezowski J.J., Agranat I.
Chemistry - A European Journal,
2006
19.
10.1016/j.mencom.2019.09.036_bib0095
Neese
Wiley Interdisp. Rev.: Comput. Mol. Sci.,
2012
20.

Weigend F., Ahlrichs R.
Physical Chemistry Chemical Physics,
2005