Abstract
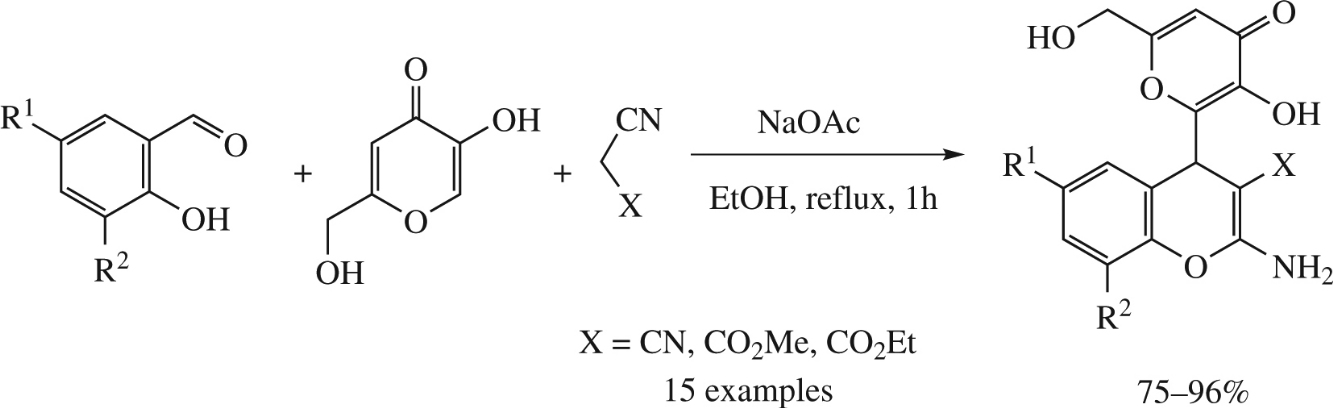
A multicomponent one-pot environmentally benign transformation of salicylaldehydes, kojic acid and malononitrile or its derivativies, catalyzed by sodium acetate, in a small amount of ethanol results in efficient formation of new substituted 2-amino-4-[3-hydroxy-6-(hydroxymethyl)-4-oxo-4H-pyran-2-yl]-4H-chromene-3-carbonitriles or -3-carboxylates in 75–96% yields.
References
1.
10.1016/j.mencom.2019.09.035_bib0005
Multicomponent Reactions,
2005
2.

Wender P.A.
Natural Product Reports,
2014
3.
ZIRAK M., EFTEKHARI-SIS B.
Turkish Journal of Chemistry,
2015
4.

Bentley R.
Natural Product Reports,
2006
5.

Chang T.
International Journal of Molecular Sciences,
2009
6.

Cabanes J., Chazarra S., Garcia-Carmona F.
Journal of Pharmacy and Pharmacology,
1994
7.

Reddy B.V., Reddy M.R., Madan C., Kumar K.P., Rao M.S.
Bioorganic and Medicinal Chemistry Letters,
2010
8.

Rho H.S., Ahn S.M., Yoo D.S., Kim M.K., Cho D.H., Cho J.Y.
Bioorganic and Medicinal Chemistry Letters,
2010
9.

Aytemir M.D., Özçelik B.
European Journal of Medicinal Chemistry,
2010
10.

Tanaka R., Tsujii H., Yamada T., Kajimoto T., Amano F., Hasegawa J., Hamashima Y., Node M., Katoh K., Takebe Y.
Bioorganic and Medicinal Chemistry,
2009
11.
10.1016/j.mencom.2019.09.035_bib0055
Murray
The Natural Coumarins: Occurrence, Chemistry and Biochemistry,
1982
12.

Baglin T.P., Keeling D.M., Watson H.G.
British Journal of Haematology,
2005
13.

Gourdeau H., Leblond L., Hamelin B., Desputeau C., Dong K., Kianicka I., Custeau D., Boudreau C., Geerts L., Cai S., Drewe J., Labrecque D., Kasibhatla S., Tseng B.
Molecular Cancer Therapeutics,
2004
14.
![Electrochemically induced multicomponent assembling of isatins, 4-hydroxyquinolin-2(1H)-one and malononitrile: a convenient and efficient way to functionalized spirocyclic [indole-3,4′-pyrano[3,2-c]quinoline] scaffold](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Elinson M.N., Merkulova V.M., Ilovaisky A.I., Demchuk D.V., Belyakov P.A., Nikishin G.I.
Molecular Diversity,
2009
15.

Vereshchagin A.N., Elinson M.N., Ryzhkov F.V., Nasybullin R.F., Bobrovsky S.I., Goloveshkin A.S., Egorov M.P.
Comptes Rendus Chimie,
2015
16.
Elinson M.N., Ryzhkov F.V., Vereshchagin A.N., Korshunov A.D., Novikov R.A., Egorov M.P.
Mendeleev Communications,
2017
17.
![SiO2–OSO3H Nanoparticles: An Efficient, Versatile and New Reagent for the One-Pot Synthesis of 2-Amino-8-Oxo-4,8-Dihydropyrano[3,2-b]Pyran-3-Carbonitrile Derivatives in Water, a Green Protocol](/storage/images/resized/ruydfaB80LDjlkYqsfOeUAZohOIODyq7bQzis5O7_small_thumb.webp)
Sadeghi B., Nezhad P.F., Hashemian S.
Journal of Chemical Research,
2014
18.
![Ferrocene-Containing Ionic Liquid Supported on Silica Nanospheres (SiO2@Imid-Cl@Fc) as a Mild and Efficient Heterogeneous Catalyst for the Synthesis of Pyrano[3,2-b]Pyran Derivatives Under Ultrasound Irradiation](/storage/images/resized/ruydfaB80LDjlkYqsfOeUAZohOIODyq7bQzis5O7_small_thumb.webp)
Teimuri-Mofrad R., Esmati S., Rabiei M., Gholamhosseini-Nazari M.
Journal of Chemical Research,
2018
19.
Vereshchagin A.N., Elinson M.N., Anisina Y.E., Ryzhkov F.V., Goloveshkin A.S., Bushmarinov I.S., Zlotin S.G., Egorov M.P.
Mendeleev Communications,
2015
20.
Elinson M.N., Vereshchagin A.N., Anisina Y.E., Goloveshkin A.S., Ushakov I.E., Egorov M.P.
Mendeleev Communications,
2018
21.

22.

Butler R.N., Coyne A.G.
Organic and Biomolecular Chemistry,
2016
23.

24.

Sarkar A., Santra S., Kundu S.K., Hajra A., Zyryanov G.V., Chupakhin O.N., Charushin V.N., Majee A.
Green Chemistry,
2016
25.
![One-Pot ‘On-solvent’ Multicomponent Protocol for the Synthesis of Medicinally Relevant 4H-Pyrano[3,2-c]quinoline Scaffold](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Vereshchagin A.N., Elinson M.N., Nasybullin R.F., Ryzhkov F.V., Bobrovsky S.I., Bushmarinov I.S., Egorov M.P.
Helvetica Chimica Acta,
2015
26.
Elinson M.N., Medvedev M.G., Ilovaisky A.I., Merkulova V.M., Zaimovskaya T.A., Nikishin G.I.
Mendeleev Communications,
2013
27.
![Chemical and electrocatalytic cascade cyclization of salicylaldehyde with three molecules of malononitrile: ‘one-pot’ simple and efficient way to the chromeno[2,3-b]pyridine scaffold](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Elinson M.N., Gorbunov S.V., Vereshchagin A.N., Nasybullin R.F., Goloveshkin A.S., Bushmarinov I.S., Egorov M.P.
Tetrahedron,
2014