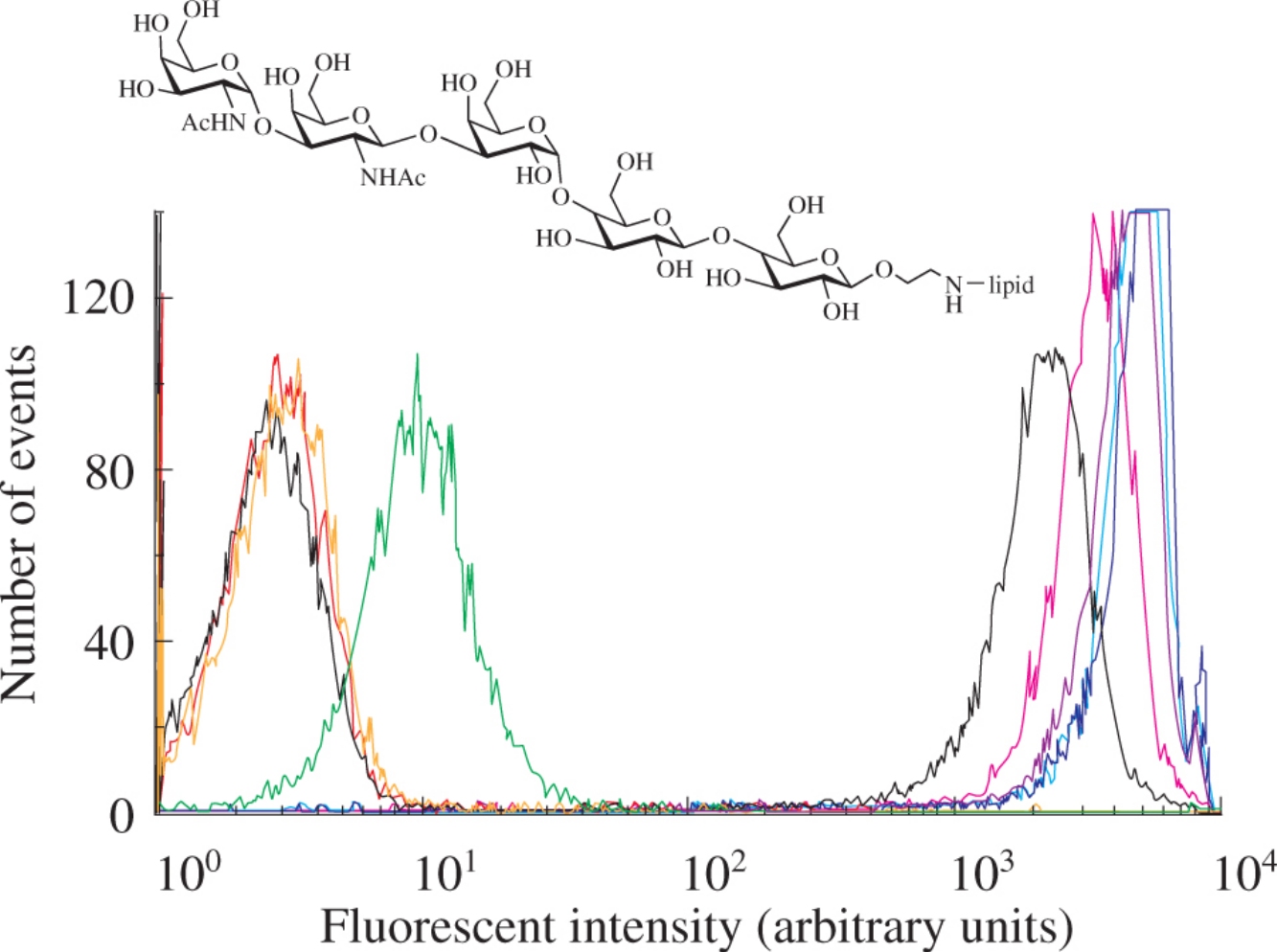
Abstract
Synthesis of the glycan part of Forssman glycolipid GalNAcα1-3GalNAcβ1-3Galα1-4Galβ1-4Glc–Cer in the form of 2-aminoethyl glycoside has been carried out. The glycoside has been converted into a lipophilic derivative capable of controlled inserting into erythrocytes. The obtained surface-modified cells, termed kodecytes, revealed a high level of the blood group system FORS serological activity.
References
1.

Svensson L., Hult A.K., Stamps R., Ångström J., Teneberg S., Storry J.R., Jørgensen R., Rydberg L., Henry S.M., Olsson M.L.
Blood,
2013
2.

Young, W.W., Hakomori S., Levine P.
Journal of Immunology,
1979
3.

Bovin N., Obukhova P., Shilova N., Rapoport E., Popova I., Navakouski M., Unverzagt C., Vuskovic M., Huflejt M.
Biochimica et Biophysica Acta - General Subjects,
2012
4.

Nilsson U.
Carbohydrate Research,
1994
5.

Sablina M.A., Tuzikov A.B., Ovchinnikova T.V., Mikhura I.V., Bovin N.V.
Russian Chemical Bulletin,
2015
6.

Zinin A.I., Malysheva N.N., Shpirt A.M., Torgov V.I., Kononov L.O.
Carbohydrate Research,
2007
7.
Schmidt R.R., Michel J.
1980
8.

King J.F., Allbutt A.D.
Canadian Journal of Chemistry,
1970
9.

Schultz M., Kunz H.
Tetrahedron Asymmetry,
1993
10.

Huang C., Wang N., Fujiki K., Otsuka Y., Akamatsu M., Fujimoto Y., Fukase K.
Journal of Carbohydrate Chemistry,
2010
11.

Korchagina E.Y., Henry S.M.
Biochemistry (Moscow),
2015