Abstract
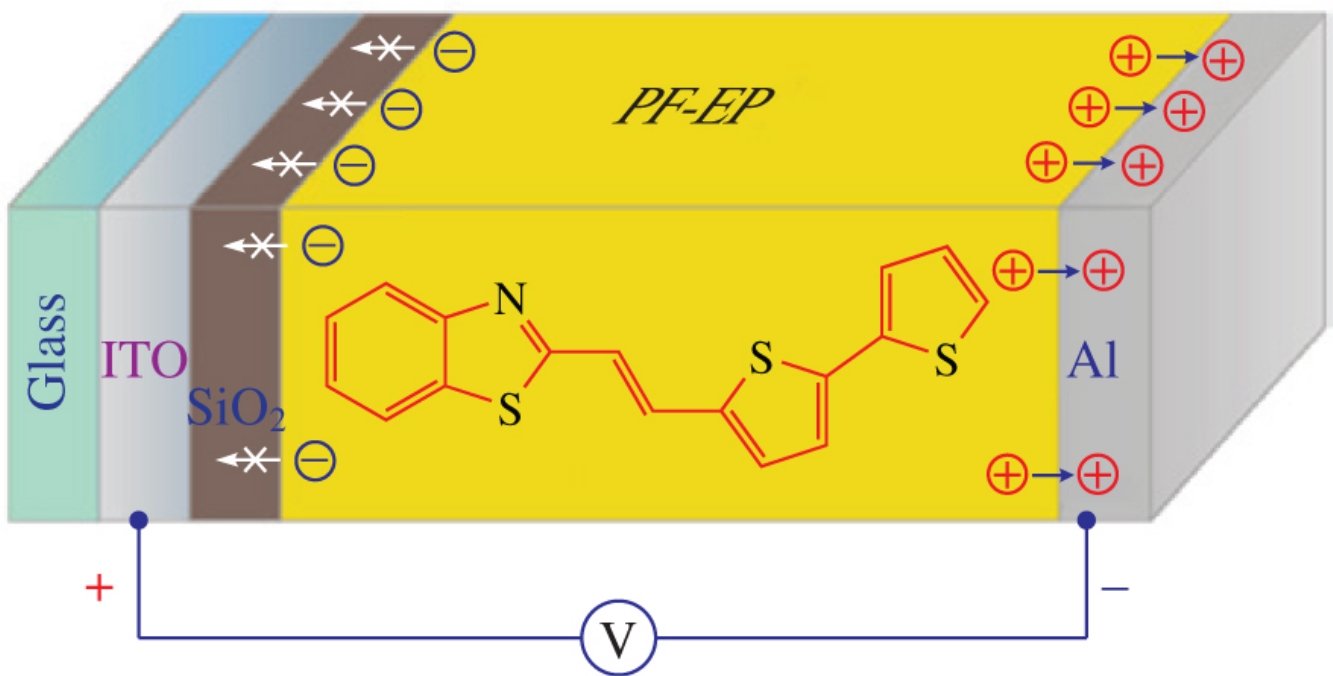
Condensation of 2-methylbenzothiazole with 2,2′-bithiophene-5-carbaldehyde affords the title compound, a new chromophore. This compound was used for the doping of poly[9,9-bis(6-diethoxylphosphorylhexyl)fluorene], and the obtained composites demonstrated high electron mobility (1.50×10−4cm2V−1s−1) in contrast to dopant-free matrixes.
References
1.

Walker B., Kim C., Nguyen T.
Chemistry of Materials,
2010
2.

Li Y., Guo Q., Li Z., Pei J., Tian W.
Energy and Environmental Science,
2010
3.

Roncali J.
Accounts of Chemical Research,
2009
4.

Lloyd M.T., Anthony J.E., Malliaras G.G.
Materials Today,
2007
5.
Solodukhin A.N., Luponosov Y.N., Buzin M.I., Peregudova S.M., Svidchenko E.A., Ponomarenko S.A.
Mendeleev Communications,
2018
6.
Lukovskaya E.V., Dyachenko N.V., Khoroshutin A.V., Bobyleva A.A., Anisimov A.V., Karnoukhova V.A., Jonusauskas G., Fedorov Y.V., Fedorova O.A.
Mendeleev Communications,
2018
7.

Walker B., Tamayo A.B., Dang X., Zalar P., Seo J.H., Garcia A., Tantiwiwat M., Nguyen T.
Advanced Functional Materials,
2009
8.

Shang H., Fan H., Liu Y., Hu W., Li Y., Zhan X.
Advanced Materials,
2011
9.

Loser S., Bruns C.J., Miyauchi H., Ortiz R.P., Facchetti A., Stupp S.I., Marks T.J.
Journal of the American Chemical Society,
2011
10.

Yin B., Yang L., Liu Y., Chen Y., Qi Q., Zhang F., Yin S.
Applied Physics Letters,
2010
11.

Cheng Y., Yang S., Hsu C.
Chemical Reviews,
2009
12.

Kanaparthi R.K., Kandhadi J., Giribabu L.
Tetrahedron,
2012
13.

Sizov A.S., Agina E.V., Ponomarenko S.A.
Russian Chemical Reviews,
2018
14.

Wang C., Dong H., Hu W., Liu Y., Zhu D.
Chemical Reviews,
2011
15.

Di C., Zhang F., Zhu D.
Advanced Materials,
2012
16.

Zaumseil J., Sirringhaus H.
Chemical Reviews,
2007
17.

Yan H., Chen Z., Zheng Y., Newman C., Quinn J.R., Dötz F., Kastler M., Facchetti A.
Nature,
2009
18.

Klauk H., Zschieschang U., Pflaum J., Halik M.
Nature,
2007
19.

Kim F.S., Guo X., Watson M.D., Jenekhe S.A.
Advanced Materials,
2010
20.

Muccini M.
Nature Materials,
2006
21.

Capelli R., Toffanin S., Generali G., Usta H., Facchetti A., Muccini M.
Nature Materials,
2010
22.

Muccini M., Koopman W., Toffanin S.
Laser and Photonics Reviews,
2011
23.

Kim Y., Son Y.
Molecular Crystals and Liquid Crystals,
2011
24.

Ginocchietti G., Cecchetto E., De Cola L., Mazzucato U., Spalletti A.
Chemical Physics,
2008
25.

Fischer E.
The Journal of Physical Chemistry,
1967
26.

Zhang B., Qin C., Ding J., Chen L., Xie Z., Cheng Y., Wang L.
Advanced Functional Materials,
2010
27.

Armin A., Juska G., Ullah M., Velusamy M., Burn P.L., Meredith P., Pivrikas A.
Advanced Energy Materials,
2013
28.
Malov V.V., Ghosh T., Nair V.C., Maslov M.M., Katin K.P., Unni K.N., Tameev A.R.
Mendeleev Communications,
2019
29.

Kim Y., Hellmuth T.J., Sysoiev D., Pauly F., Pietsch T., Wolf J., Erbe A., Huhn T., Groth U., Steiner U.E., Scheer E.
Nano Letters,
2012