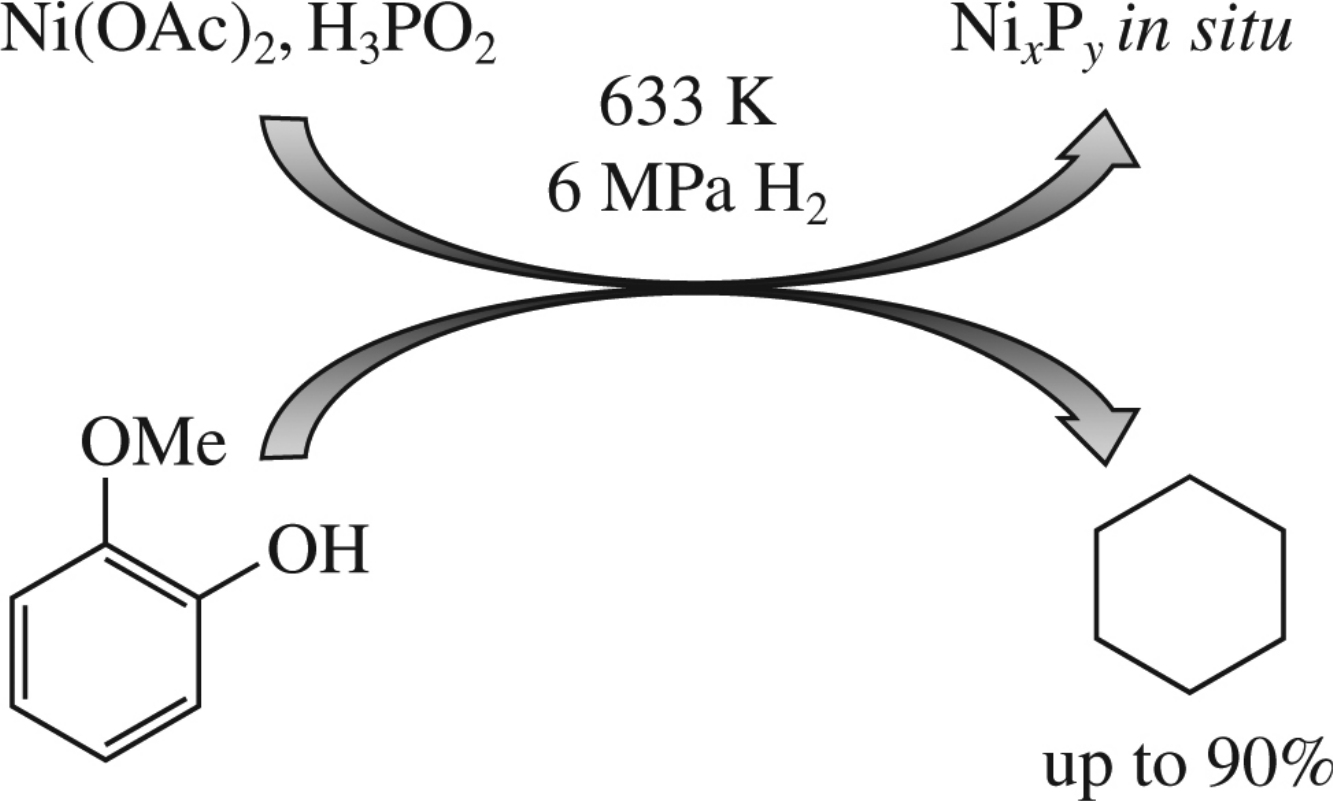
Abstract
The title catalytic system exhibited high activity in the hydrogenation–hydrodeoxygenation of guaiacol (a low-molecular-weight product of lignin degradation). The conversion of guaiacol reached 97%, and the selectivity for cyclohexane was as high as 94%. Nickel phosphide phases were obtained in the reaction medium, and their presence determined the high activity of the catalytic system in the hydroconversion of guaiacol.
References
1.

Goyal H.B., Seal D., Saxena R.C.
Renewable and Sustainable Energy Reviews,
2008
2.

Oyama S.T., Gott T., Zhao H., Lee Y.
Catalysis Today,
2009
3.

Oyama S.T.
Journal of Catalysis,
2003
4.

Liu D., Wang A., Liu C., Prins R.
Catalysis Today,
2017
5.

De Jesus J.C., González I., Quevedo A., Puerta T.
Journal of Molecular Catalysis A Chemical,
2005
6.

YANG S., LIANG C., PRINS R.
Journal of Catalysis,
2006
7.

Berhault G., Afanasiev P., Loboué H., Geantet C., Cseri T., Pichon C., Guillot-Deudon C., Lafond A.
Inorganic Chemistry,
2009
8.

Rodriguez J.A., Kim J., Hanson J.C., Sawhill S.J., Bussell M.E.
Journal of Physical Chemistry B,
2003
9.

Whiffen V.M., Smith K.J.
Energy & Fuels,
2010
10.

Stinner C., Prins R., Weber T.
Journal of Catalysis,
2000
11.

Clark P., Li W., Oyama S.T.
Journal of Catalysis,
2001
12.

Prins R., Bussell M.E.
Catalysis Letters,
2012
13.

Yang S., Prins R.
Chemical Communications,
2005
14.

Zhao H.Y., Li D., Bui P., Oyama S.T.
Applied Catalysis A: General,
2011
15.

Wu S., Lai P., Lin Y., Wan H., Lee H., Chang Y.
ACS Sustainable Chemistry and Engineering,
2013
16.

Wu S., Lai P., Lin Y.
Catalysis Letters,
2014
17.

Moon J., Kim E., Lee Y.
Journal of Catalysis,
2014
18.

Moon J., Lee Y.
Topics in Catalysis,
2015
19.

Ted Oyama S., Onkawa T., Takagaki A., Kikuchi R., Hosokai S., Suzuki Y., Bando K.K.
Topics in Catalysis,
2015
20.

Feitosa L.F., Berhault G., Laurenti D., Davies T.E., Teixeira da Silva V.
Journal of Catalysis,
2016
21.

Highly selective hydrodeoxygenation of anisole, phenol and guaiacol to benzene over nickel phosphide
Li Y., Fu J., Chen B.
RSC Advances,
2017
22.

Lan X., Hensen E.J., Weber T.
Applied Catalysis A: General,
2018
23.

SAWHILL S., LAYMAN K., VANWYK D., ENGELHARD M., WANG C., BUSSELL M.
Journal of Catalysis,
2005
24.

Guan Q., Li W., Zhang M., Tao K.
Journal of Catalysis,
2009
25.

Motos-Pérez B., Uzio D., Aymonier C.
ChemCatChem,
2015
26.

Stinner C., Tang Z., Haouas M., Weber T., Prins R.
Journal of Catalysis,
2002
27.

Costa D.C., Soldati A.L., Pecchi G., Bengoa J.F., Marchetti S.G., Vetere V.
Nanotechnology,
2018
28.

Pan Y., Liu Y., Zhao J., Yang K., Liang J., Liu D., Hu W., Liu D., Liu Y., Liu C.
Journal of Materials Chemistry A,
2015
29.

Li K., Wang R., Chen J.
Energy & Fuels,
2011
30.

LEE Y., OYAMA S.
Journal of Catalysis,
2006
31.

Abu I.I., Smith K.J.
Applied Catalysis A: General,
2007
32.

Cecilia J.A., Infantes-Molina A., Rodríguez-Castellón E., Jiménez-López A., Oyama S.T.
Applied Catalysis B: Environmental,
2013
33.

Bussell M.E.
Reaction Chemistry and Engineering,
2017
34.

Deliy I., Shamanaev I., Gerasimov E., Pakharukova V., Yakovlev I., Lapina O., Aleksandrov P., Bukhtiyarova G.
Catalysts,
2017