Abstract
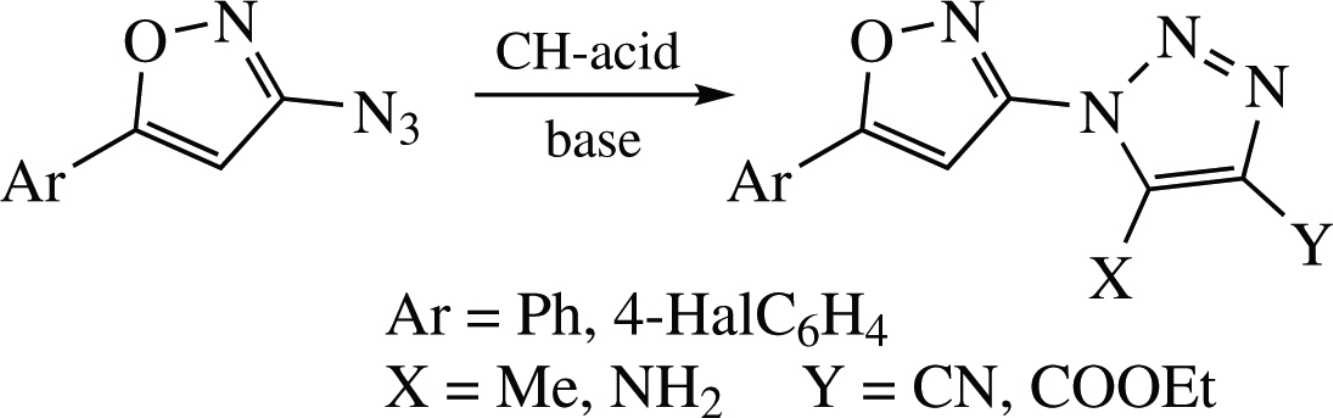
3-Azidoisoxazoles react with various active methylene compounds (malononitrile, ethyl cyanoacetate and ethyl acetoacetate) in the presence of base to give hybrid isoxazole–triazole molecules in good to nearly quantitative yields.
References
1.
Muzalevskiy V.M., Mamedzade M.N., Chertkov V.A., Bakulev V.A., Nenajdenko V.G.
Mendeleev Communications,
2018
2.

Rozin Y.A., Leban J., Dehaen W., Nenajdenko V.G., Muzalevskiy V.M., Eltsov O.S., Bakulev V.A.
Tetrahedron,
2012
3.

Chen Z., Liu Z., Cao G., Li H., Ren H.
Advanced Synthesis and Catalysis,
2017
4.
![Handling Hazards Using Continuous Flow Chemistry: Synthesis of N1-Aryl-[1,2,3]-triazoles from Anilines via Telescoped Three-Step Diazotization, Azidodediazotization, and [3 + 2] Dipolar Cycloaddition Processes](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Teci M., Tilley M., McGuire M.A., Organ M.G.
Organic Process Research and Development,
2016
5.

Brand S., Ko E.J., Viayna E., Thompson S., Spinks D., Thomas M., Sandberg L., Francisco A.F., Jayawardhana S., Smith V.C., Jansen C., De Rycker M., Thomas J., MacLean L., Osuna-Cabello M., et. al.
Journal of Medicinal Chemistry,
2017
6.
![An Organocatalytic Azide-Aldehyde [3+2] Cycloaddition: High-Yielding Regioselective Synthesis of 1,4-Disubstituted 1,2,3-Triazoles](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Ramachary D.B., Shashank A.B., Karthik S.
Angewandte Chemie - International Edition,
2014
7.

Brooke D.G., van Dam E.M., Watts C.K., Khoury A., Dziadek M.A., Brooks H., Graham L.K., Flanagan J.U., Denny W.A.
Bioorganic and Medicinal Chemistry,
2014
8.

Sokolova N.V., Nenajdenko V.G.
RSC Advances,
2013
9.

Zdzienicka A., Schols D., Andrei G., Snoeck R., Głowacka I.E.
Phosphorus, Sulfur and Silicon and the Related Elements,
2015
10.

Pokhodylo N.T., Shyyka O.Y., Savka R.D., Obushak M.D.
Russian Journal of Organic Chemistry,
2018
11.

Mamedov V.A., Valeeva V.N., Antokhina L.A., Doroshkina G.M., Chernova A.V., Nuretdinov I.A.
Chemistry of Heterocyclic Compounds,
1993
12.

Mikhailychenko S.N., Chesniuk A.A., Koniushkin L.D., Firgang S.I., Zaplishny V.N.
Chemistry of Heterocyclic Compounds,
2004
13.
![Synthesis and transformations of substituted 4,6-dimethylpyrazolo[3,4-b]pyridyl-3-azides and -sulfonyl chlorides](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Dmitrieva I.G., Dyadyuchenko L.V., Strelkov V.D., Kaigorodova E.A.
Chemistry of Heterocyclic Compounds,
2008
14.
Abbas Temerik H.H., Younes M.I., Metwally S.A.
Collection of Czechoslovak Chemical Communications,
2003
15.

Sysak A., Obmińska-Mrukowicz B.
European Journal of Medicinal Chemistry,
2017
16.

Chen Q., Liu F., Xu F., Yang C.
Journal of Heterocyclic Chemistry,
2008
17.

Rao Y.J., Srinivas A.
Journal of Heterocyclic Chemistry,
2014
18.

Najafi Z., Mahdavi M., Safavi M., Saeedi M., Alinezhad H., Pordeli M., Kabudanian Ardestani S., Shafiee A., Foroumadi A., Akbarzadeh T.
Journal of Heterocyclic Chemistry,
2014
19.

Niu T., Lv M., wang L., Yi W., Cai C.
Organic and Biomolecular Chemistry,
2013
20.

Coffman K.C., Hartley T.P., Dallas J.L., Kurth M.J.
ACS Combinatorial Science,
2012
21.

Krivopalov V.P., Shkurko O.P.
Russian Chemical Reviews,
2005
22.
10.1016/j.mencom.2019.09.017_bib0110
Banert
Organic Azides – Syntheses and Applications,
2010
23.

Lemport P.S., Smolyar I.V., Khrustalev V.N., Roznyatovsky V.A., Popov A.V., Kobelevskaya V.A., Rozentsveig I.B., Nenajdenko V.G.
Organic Chemistry Frontiers,
2019
24.

Shastin A.V., Tsyrenova B.D., Sergeev P.G., Roznyatovsky V.A., Smolyar I.V., Khrustalev V.N., Nenajdenko V.G.
Organic Letters,
2018
25.

Battye T.G., Kontogiannis L., Johnson O., Powell H.R., Leslie A.G.
Acta Crystallographica Section D Biological Crystallography,
2011
26.

Evans P.
Acta Crystallographica Section D Biological Crystallography,
2005
27.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015
28.

Dheer D., Singh V., Shankar R.
Bioorganic Chemistry,
2017