Abstract
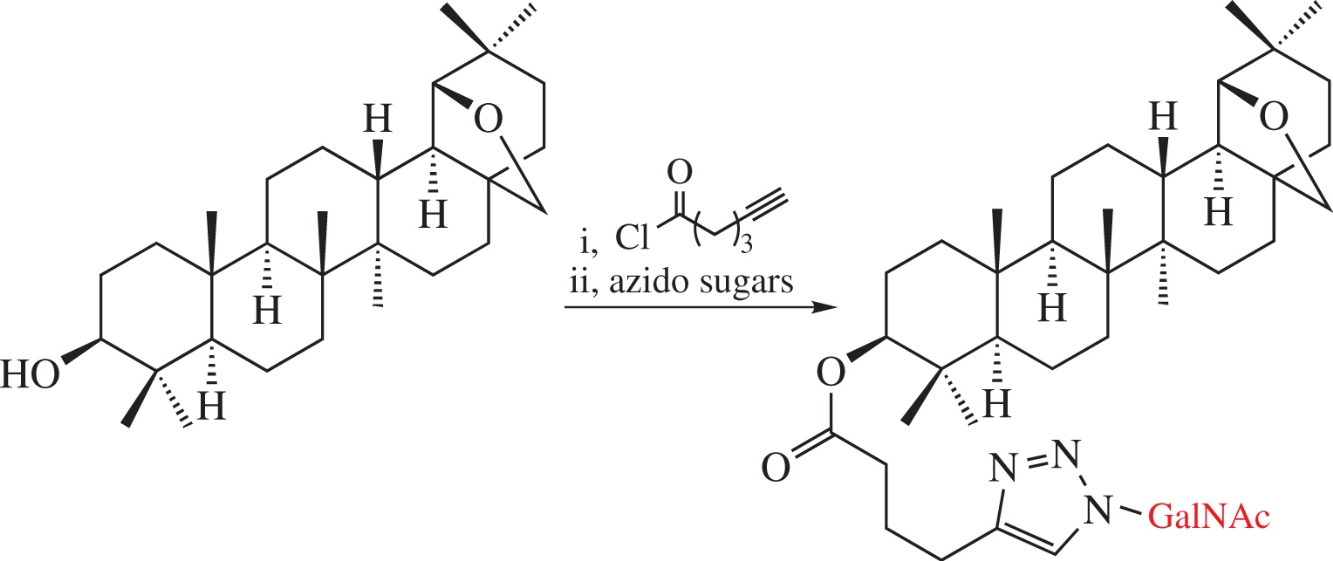
New allobetulin conjugates were obtained through its O-esterification with hex-5-ynoic acid followed by [3+2]-cycloaddition with three azido derivatives of N-acetyl-d-galactosamine. The conjugates are non-toxic in micromolar range against hepatocellular carcinoma cell lines and have a high affinity towards the HO asialoglycoprotein receptor of hepatocytes based on molecular docking and surface plasmon resonance data.
References
1.

Govindarajan M.
European Journal of Medicinal Chemistry,
2018
2.
10.1016/j.mencom.2019.09.016_sbref0005b
Schwarz
2011
3.

La Ferla B., Airoldi C., Zona C., Orsato A., Cardona F., Merlo S., Sironi E., D'Orazio G., Nicotra F.
Natural Product Reports,
2011
4.

Latxague L., Gaubert A., Barthélémy P.
Molecules,
2018
5.
Koloskova O.O., Nosova A.S., Shchelik I.S., Shilovskiy I.P., Sebyakin Y.L., Khaitov M.R.
Mendeleev Communications,
2017
6.

Dehaen W., Mashentseva A.A., Seitembetov T.S.
Molecules,
2011
7.

Kazakova O.B., Smirnova I.E., Khusnutdinova E.F., Zhukova O.S., Fetisova L.V., Apryshko G.N., Medvedeva N.I., Yamansarov E.Y., Baikova I.P., Nguyen T.T., Thu H.D.
Russian Journal of Bioorganic Chemistry,
2014
8.

Paduch R., Kandefer-Szerszen M.
Mini-Reviews in Organic Chemistry,
2014
9.

Rufino-Palomares E.E., Perez-Jimenez A., J. Reyes-Zurita F., Garcia- Salguero L., Mokhtari K., Herrera-Merchan A., P. Medina P., Peragon J., A. Lupianez J.
Current Organic Chemistry,
2015
10.
10.1016/j.mencom.2019.09.016_bib0030
Mierina
Curr. Med. Chem.,
2018
11.

Huang X., Leroux J., Castagner B.
Bioconjugate Chemistry,
2016
12.

Ivanenkov Y.A., Majouga A.G., Petrov R.A., Petrov S.A., Kovalev S.V., Maklakova S.Y., Yamansarov E.Y., Saltykova I.V., Deyneka E.V., Filkov G.I., Kotelianski V.E., Zatsepin T.S., Beloglazkina E.K.
Bioorganic and Medicinal Chemistry Letters,
2018
13.

Petrov R.A., Maklakova S.Y., Ivanenkov Y.A., Petrov S.A., Sergeeva O.V., Yamansarov E.Y., Saltykova I.V., Kireev I.I., Alieva I.B., Deyneka E.V., Sofronova A.A., Aladinskaia A.V., Trofimenko A.V., Yamidanov R.S., Kovalev S.V., et. al.
Bioorganic and Medicinal Chemistry Letters,
2018
14.

Dheer D., Singh V., Shankar R.
Bioorganic Chemistry,
2017
15.

Hou J., Liu X., Shen J., Zhao G., Wang P.G.
Expert Opinion on Drug Discovery,
2012
16.

Zhou M., Zhang R., Wang M., Xu G., Liao S.
European Journal of Medicinal Chemistry,
2017
17.

Yamansarov E.Y., Saltykova I.V., Kovalev S.V., Petrov R.A., Shkil’ D.O., Seleznev E.I., Beloglazkina E.K., Majouga A.G.
Russian Chemical Bulletin,
2019
18.

Nair J.K., Willoughby J.L., Chan A., Charisse K., Alam M.R., Wang Q., Hoekstra M., Kandasamy P., Kel’in A.V., Milstein S., Taneja N., O’Shea J., Shaikh S., Zhang L., van der Sluis R.J., et. al.
Journal of the American Chemical Society,
2014
19.

Sanhueza C.A., Baksh M.M., Thuma B., Roy M.D., Dutta S., Préville C., Chrunyk B.A., Beaumont K., Dullea R., Ammirati M., Liu S., Gebhard D., Finley J.E., Salatto C.T., King-Ahmad A., et. al.
Journal of the American Chemical Society,
2017
20.

Stokmaier D., Khorev O., Cutting B., Born R., Ricklin D., Ernst T.O., Böni F., Schwingruber K., Gentner M., Wittwer M., Spreafico M., Vedani A., Rabbani S., Schwardt O., Ernst B., et. al.
Bioorganic and Medicinal Chemistry,
2009
21.

Majouga A., Ivanenkov Y., Veselov M., Lopuhov A., Maklakova S., Beloglazkina E., Binevski P., Klyachko N., Sandulenko Y., Galkina N., Koteliansky V.
Current Drug Delivery,
2016
22.

Mosmann T.
Journal of Immunological Methods,
1983