Abstract
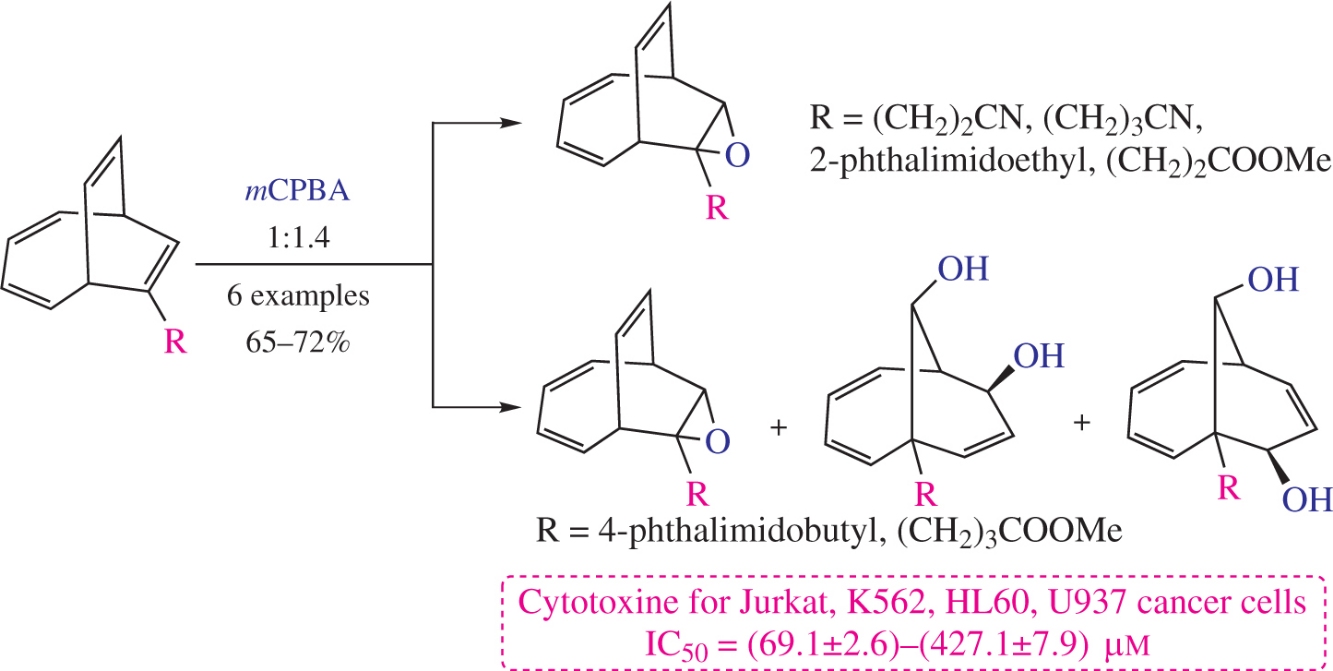
The oxidation of functionally substituted bicyclo[4.2.2]deca-2,4,7,9-tetraenes with m-chloroperbenzoic acid affords practically valuable 8-oxatricyclo[4.3.2.07,9]undeca-2,4,10-trienes and bicyclo[4.3.1]deca-2,4,8-triene-7,10-diols in yields of 65–72%. The structures of the products were established by advanced spectral methods and X-ray diffraction analysis. The new compounds were screened for in vitro cytotoxicity against Jurkat, K562, U937 and HL60 tumor cell lines.
References
1.
![Bicyclo[4.3.1]decatrienyl-kation: ein überbrücktes bishomotropyliumion.](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Schröder G., Prange U., Bowman N.S., Oth J.F.
Tetrahedron Letters,
1970
2.
![Überbrückte Bishomotropylium‐Ionen aus Bicyclo[4.3.1]decatrienen](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Schröder G., Prange U., Putze B., Thio J., Oth J.F.
Chemische Berichte,
1971
3.
![Methoxymercurierung von bullvalen und bicyclo[4.2.2]deca-2,4,7,9-tetraen](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Löffler H.-., Schröder G.
Tetrahedron Letters,
1970
4.
![1-Methylbicyclo[4.3.0]nonatrienyl cation. A 1,4-bishomotropylium ion](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Ahlberg P., Harris D.L., Winstein S.
Journal of the American Chemical Society,
1970
5.
![Reaktion von Tetracyanäthylen mit überbrückten. Homotropilidensystemen und Bicyclo[4.2.2]decatetraen](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Reaktion von Tetracyanäthylen mit überbrückten. Homotropilidensystemen und Bicyclo[4.2.2]decatetraen
Löffler H., Martini T., Musso H., Schröder G.
Chemische Berichte,
1970
6.
![Unsaturated heterocyclic systems. LXXXIX. Reactivity of bicyclo [4.2.2] deca-2,4,7,9-tetraene derivatives under conditions of uniparticulate electrophilic addition. Intramolecular capture of zwitterionic bridged 1,4-bishomotropylium (bicyclo [4.3.1] deca-2,4,7-trienyl) intermediates](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Paquette L.A., Broadhurst M.J.
Journal of Organic Chemistry,
1973
7.

D’yakonov V.A., Agliullina R.A., Makhamatkhanova A.L., Tyumkina T.V., Dzhemilev U.M.
Chemistry of Heterocyclic Compounds,
2018
8.
![Cobalt-Catalyzed [6 + 2] Cycloaddition of Alkynes with 1,3,5,7-Cyclooctatetraene as a Key Element in the Direct Construction of Substituted Bicyclo[4.3.1]decanes](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
D’yakonov V.A., Kadikova G.N., Dzhemileva L.U., Gazizullina G.F., Ramazanov I.R., Dzhemilev U.M.
Journal of Organic Chemistry,
2016
9.
![Oxidative skeletal rearrangement of bicyclo[4.2.2]deca-2,4,7,9-tetraenes to bicyclo[4.3.1]deca-2,4,8-triene-7,10-diols and study of the antitumor activity of the products in vitro](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
D'yakonov V.A., Kadikova G.N., Dzhemileva L.U., Gazizullina G.F., Yunusbaeva M.M., Dzhemilev U.M.
Tetrahedron,
2018
10.

Dyakonov V.A., Kadikova G.N., Dzhemilev U.M.
Russian Chemical Reviews,
2018
11.
![A ring-closing metathesis approach to the bicyclo[4.3.1]decane core of caryolanes](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Goldring W.P., Paden W.T.
Tetrahedron Letters,
2011
12.
![Total Synthesis of the CP-Molecules (CP-263,114 and CP-225,917, Phomoidrides B and A). 1. Racemic and Asymmetric Synthesis of Bicyclo[4.3.1] Key Building Blocks](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Nicolaou K.C., Jung J., Yoon W.H., Fong K.C., Choi H.-., He Y., Zhong Y.-., Baran P.S.
Journal of the American Chemical Society,
2002
13.

Richter J.M., Ishihara Y., Masuda T., Whitefield B.W., Llamas T., Pohjakallio A., Baran P.S.
Journal of the American Chemical Society,
2008
14.

Stratmann K., Moore R.E., Bonjouklian R., Deeter J.B., Patterson G.M., Shaffer S., Smith C.D., Smitka T.A.
Journal of the American Chemical Society,
1994
15.

Tanis S.P., Herrinton P.M.
Journal of Organic Chemistry,
1985
16.
10.1016/j.mencom.2019.09.013_sbref0015f
Scheuer
Marine Natural Products. Chemical and Biological Perspectives,
1983
17.

Drahl M.A., Akhmedov N.G., Williams L.J.
Tetrahedron Letters,
2011
18.

Choudhary M.I., Siddiqui Z.A., Nawaz S.A., Atta-ur-Rahman
Journal of Natural Products,
2006
19.

DABRAH T.T., HARWOOD H.J., HUANG L.H., JANKOVICH N.D., KANEKO T., LI J., LINDSEY S., MOSHIER P.M., SUBASHI T.A., THERRIEN M., WATTS P.C.
Journal of Antibiotics,
2012
20.
![Cobalt(I)-Catalyzed Cycloaddition of Functionally Substituted Alkynes and 1,3-Diynes to 1,3,5,7-Cyclooctatetraene in the Synthesis of Bicyclo[4.2.2]deca-2,4,7,9-tetraenes](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
D'yakonov V.A., Kadikova G.N., Gazizullina G.F., Dzhemilev U.M.
ChemistrySelect,
2018
21.
![Cobalt(I)-Catalyzed [6+2] Cycloadditions of Cyclooctatetra(tri)ene with Alkynes](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Achard M., Mosrin M., Tenaglia A., Buono G.
Journal of Organic Chemistry,
2006
22.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007