Abstract
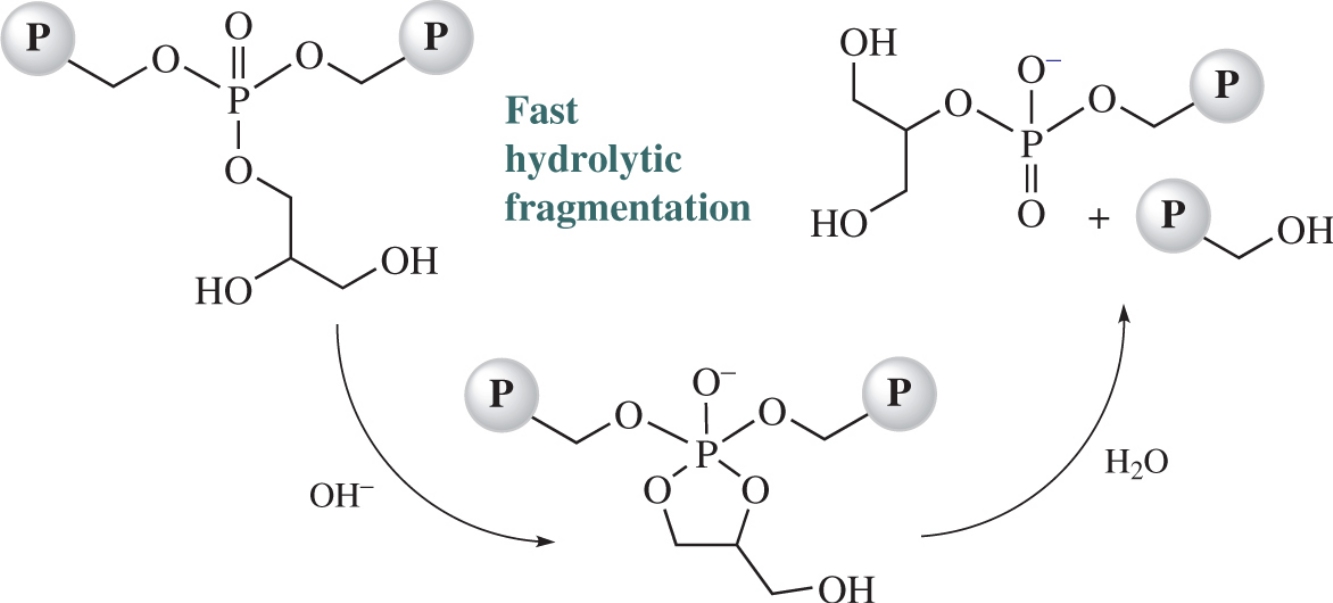
2,3-Dihydroxypropyl functionalized polyphosphates obtained via controlled acid-catalyzed hydrolysis of 2-[(2,2-dimethyl-1,3-dioxolan-4-yl)methoxy]- or 2-(oxiran-2-ylmethoxy)-1,3,2-dioxaphospholane 2-oxide homopolymers are highly sensitive to basic hydrolysis along the polyphosphate chain.
References
1.

Wang Y., Yuan Y., Du J., Yang X., Wang J.
Macromolecular Bioscience,
2009
2.
10.1016/j.mencom.2019.09.010_bib0010
Lapienis
2012
3.

Nifant'ev I.E., Shlyakhtin A.V., Bagrov V.V., Komarov P.D., Kosarev M.A., Tavtorkin A.N., Minyaev M.E., Roznyatovsky V.A., Ivchenko P.V.
Polymer Chemistry,
2017
4.

Nifant’ev I., Shlyakhtin A., Kosarev M., Karchevsky S., Ivchenko P.
Polymers,
2018
5.

Clément B., Grignard B., Koole L., Jérôme C., Lecomte P.
Macromolecules,
2012
6.
10.1016/j.mencom.2019.09.010_bib0030
Wang
2012
7.

Wang H., Su L., Li R., Zhang S., Fan J., Zhang F., Nguyen T.P., Wooley K.L.
ACS Macro Letters,
2017
8.

Bauer K.N., Tee H.T., Velencoso M.M., Wurm F.R.
Progress in Polymer Science,
2017
9.

Penczek S., Pretula J.B., Kaluzynski K., Lapienis G.
Israel Journal of Chemistry,
2012
10.

Zhang S., Wang H., Shen Y., Zhang F., Seetho K., Zou J., Taylor J.A., Dove A.P., Wooley K.L.
Macromolecules,
2013
11.

Wang D., Williams C.G., Yang F., Cher N., Lee H., Elisseeff J.H.
Tissue Engineering,
2005
12.

Zhao Z., Wang J., Mao H., Leong K.W.
Advanced Drug Delivery Reviews,
2003
13.

Lim Y.H., Tiemann K.M., Heo G.S., Wagers P.O., Rezenom Y.H., Zhang S., Zhang F., Youngs W.J., Hunstad D.A., Wooley K.L.
ACS Nano,
2015
14.

Zhai X., Huang W., Liu J., Pang Y., Zhu X., Zhou Y., Yan D.
Macromolecular Bioscience,
2011
15.

McKinlay C.J., Waymouth R.M., Wender P.A.
Journal of the American Chemical Society,
2016
16.

Zou J., Zhang F., Zhang S., Pollack S.F., Elsabahy M., Fan J., Wooley K.L.
Advanced healthcare materials,
2013
17.

Du J., Du X., Mao C., Wang J.
Journal of the American Chemical Society,
2011
18.

Steinbach T., Wurm F.R.
Angewandte Chemie - International Edition,
2015
19.

Yilmaz Z.E., Jérôme C.
Macromolecular Bioscience,
2016
20.

Zhang F., Zhang S., Pollack S.F., Li R., Gonzalez A.M., Fan J., Zou J., Leininger S.E., Pavía-Sanders A., Johnson R., Nelson L.D., Raymond J.E., Elsabahy M., Hughes D.M., Lenox M.W., et. al.
Journal of the American Chemical Society,
2015
21.

Simon J., Wolf T., Klein K., Landfester K., Wurm F.R., Mailänder V.
Angewandte Chemie - International Edition,
2018
22.

Wang H., Dong M., Khan S., Su L., Li R., Song Y., Lin Y., Kang N., Komatsu C.H., Elsabahy M., Wooley K.L.
ACS Macro Letters,
2018
23.

Hirano Y., Iwasaki Y.
Colloids and Surfaces B: Biointerfaces,
2017
24.

Baran J., Penczek S.
Macromolecules,
1995
25.

Nifant'ev I.E., Shlyakhtin A.V., Bagrov V.V., Komarov P.D., Tavtorkin A.N., Minyaev M.E., Kosarev M.A., Ivchenko P.V.
European Polymer Journal,
2018
26.

Bauer K.N., Liu L., Wagner M., Andrienko D., Wurm F.R.
European Polymer Journal,
2018
27.

Müller L.K., Steinbach T., Wurm F.R.
RSC Advances,
2015
28.

Song W., Du J., Liu N., Dou S., Cheng J., Wang J.
Macromolecules,
2008
29.
Nifant’ev I.E., Shlyakhtin A.V., Bagrov V.V., Komarov P.D., Kosarev M.A., Tavtorkin A.N., Minyaev M.E., Roznyatovsky V.A., Ivchenko P.V.
Mendeleev Communications,
2018
30.

31.

Nifant'ev I.E., Shlyakhtin A.V., Tavtorkin A.N., Ivchenko P.V., Borisov R.S., Churakov A.V.
Catalysis Communications,
2016
32.

Nifant'ev I.E., Shlyakhtin A.V., Bagrov V.V., Minyaev M.E., Churakov A.V., Karchevsky S.G., Birin K.P., Ivchenko P.V.
Dalton Transactions,
2017
33.

Nifant'ev I.E., Shlyakhtin A.V., Tavtorkin A.N., Kosarev M.A., Gavrilov D.E., Komarov P.D., Ilyin S.O., Karchevsky S.G., Ivchenko P.V.
European Polymer Journal,
2019
34.

Mosbo J.A., Verkade J.G.
Journal of Organic Chemistry,
1977