Abstract
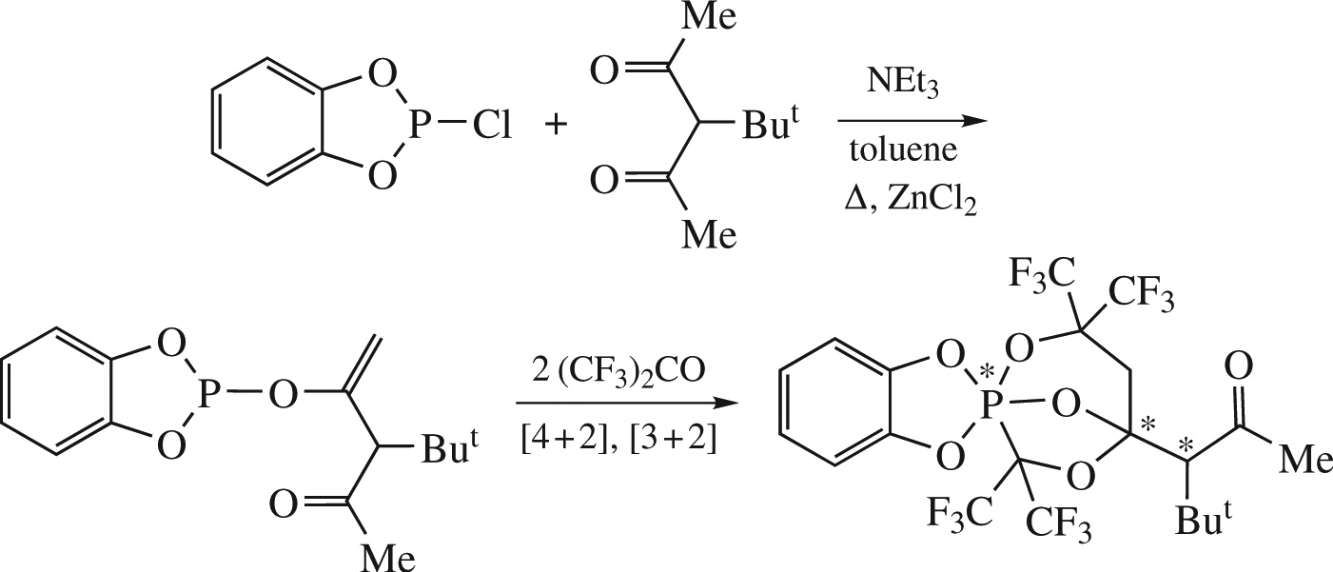
Catalytic (ZnCl2, Et3N) phosphorylation of tert-butylacetylacetone with 2-chloro-1,3,2-benzodioxaphosphole unexpectedly occurs with elimination of proton from the methyl group and leads to vinyloxyphosphole derivative, viz., 4-(1,3,2-benzodioxaphosphol-2-yloxy)-3-tert-butylpent-4-en-2-one. Its reaction with hexafluoroacetone gives a cage phosphorane as a result of subsequent chemoselective [4+2]- and [3+2]-cycloadditions with stereoselectivity above 95%.
References
1.

Kolodiazhnyi O.I.
Pure and Applied Chemistry,
2018
2.

Procházková E., Navrátil R., Janeba Z., Roithová J., Baszczyňski O.
Organic and Biomolecular Chemistry,
2019
3.

Wang T., Zhang P., Hu G., Gao Y., Wu Y., Xu P., Liu Y., Zhao Y.
ChemistrySelect,
2018
4.
Grigorenko B.L., Knyazeva M.A., Polyakov I.V., Nemukhin A.V.
Moscow University Chemistry Bulletin,
2016
5.

Zhang H., Yang L., Yan L., Liao R., Tian W.
Journal of Inorganic Biochemistry,
2018
6.

Duy Vu M., Leng W., Hsu H., Liu X.
Asian Journal of Organic Chemistry,
2018
7.

Jarwal N., Meena J.S., Thankachan P.P.
Computational and Theoretical Chemistry,
2016
8.

Hilton M.C., Zhang X., Boyle B.T., Alegre-Requena J.V., Paton R.S., McNally A.
Science,
2018
9.

Kostenko A., Dobrovetsky R.
European Journal of Organic Chemistry,
2018
10.
Pajkert R., Röschenthaler G.
Organophosphorus Chemistry,
2018
11.

Krasowska D., Pokora-Sobczak P., Jasiak A., Drabowicz J.
Advances in Heterocyclic Chemistry,
2018
12.
10.1016/j.mencom.2019.09.009_sbref0030c
Yoshifuji
Organophosphorus Chemistry: From Molecules to Applications,
2019
13.

Hazin K., Patrick B.O., Gates D.P.
Inorganic Chemistry,
2018
14.
![A twist on Hellwinkel’s salt, [P(2,2′-biphenyl)2]+[P(2,2′-biphenyl)3]–](/storage/images/resized/qZfu8vVyaDIo81Au73p6SNE8AGGzf73yfiTC33bu_small_thumb.webp)
Hazin K., Gates D.P.
Canadian Journal of Chemistry,
2018
15.

Sun C., Cao S., Zhao P., Ma W., Guo Y., Zhao Y.
Tetrahedron Letters,
2018
16.

Zhao X., Gan Z., Hu C., Duan Z., Mathey F.
Organic Letters,
2017
17.

Sharma R., Ravikanth M.
Journal of Porphyrins and Phthalocyanines,
2016
18.

Kano N., O'Brien N.J., Uematsu R., Ramozzi R., Morokuma K.
Angewandte Chemie,
2017
19.
![Features of Reaction of 2-(5-Methyl-2-phenyl-2H-1,2,3-diazaphosphol-4-yl)-4H-benzo[e]-1,3,2-dioxaphosphorin-4-one with 1,2-Dicarbonyl Compounds](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Mironov V.F., Ivkova G.A., Abdrakhmanova L.M., Mironova E.V., Musin R.Z., Cherkasov V.K.
Russian Journal of General Chemistry,
2018
20.
Nasibullin I.O., Nemtarev A.V., Mironov V.F.
Mendeleev Communications,
2017
21.

Jiang X., Toya Y., Matsukawa S., Kojima S., Jimenez-Halla J.O., Shang R., Nakamoto M., Yamamoto Y.
Chemical Science,
2019
22.

Mattioli E.J., Bottoni A., Calvaresi M.
Journal of Chemical Information and Modeling,
2019
23.

Chagas M.A., Pereira E.S., Da Silva J.C., Rocha W.R.
Journal of Molecular Modeling,
2018
24.

van Beek B., van Bochove M.A., Hamlin T.A., Bickelhaupt F.M.
Electronic Structure,
2019
25.
Abdrakhmanova L.M., Mironov V.F., Baronova T.A., Dimukhametov M.N., Krivolapov D.B., Litvinov I.A., Balandina A.A., Latypov S.K., Konovalov A.I.
Mendeleev Communications,
2006
26.
Abdrakhmanova L.M., Mironov V.F., Baronova T.A., Dimukhametov M.N., Krivolapov D.B., Litvinov I.A., Musin R.Z., Konovalov A.I.
Mendeleev Communications,
2007
27.
Mironov V.F., Kotorova Y.Y., Burnaeva L.M., Balandina A.A., Latypov S.K., Dobrynin A.B., Gubaidullin A.T., Litvinov I.A., Musin R.Z., Konovalova I.V.
Mendeleev Communications,
2009
28.
Abdrakhmanova L.M., Mironov V.F., Dimukhametov M.N., Krivolapov D.B., Litvinov I.A.
Mendeleev Communications,
2010
29.
![Caged C-P phosphoranes based on 2-(2-methyl-4-oxopent-2-yloxy)- and 2-[2-(methylcarbonyl)-1-phenoxy]-1,3,2-benzodioxaphospholes and diethyl mesoxalate](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Mironov V.F., Dimukhametov M.N., Mironova E.V., Krivolapov D.B., Abdrakhmanova L.M.
Russian Journal of General Chemistry,
2015
30.

Mironov V.F., Dimukhametov M.N., Mironova E.V., Krivolapov D.B., Ivkova G.A., Abdrakhmanova L.M.
Russian Journal of General Chemistry,
2015
31.
![Preparation and crystal structure of 1,1-tetramethylethylenedioxy-3,4-diphenyl-6-trichloromethyl-2,5,7,1-trioxaphosphabicyclo[2.2.11,4]heptane and 2-(1-hydroxy-2,2,2-trichloroethoxy)-4,4,5,5-tetramethyl- 2-oxo-1,3,2-dioxaphospholane](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Khasiyatullina N.R., Mironov V.F., Mironova E.V., Krivolapov D.B., Litvinov I.A.
Russian Journal of General Chemistry,
2016
32.

Mironov V.F., Dimukhametov M.N., Efimov S.V., Aminova R.M., Karataeva F.K., Krivolapov D.B., Mironova E.V., Klochkov V.V.
Journal of Organic Chemistry,
2016
33.

Khasiyatullina N.R., Mironov V.F., Krivolapov D.B., Mironova E.V., Gnezdilov O.I.
RSC Advances,
2016
34.
![Synthesis and steric structure of 3′,3′,5′-trimethyl-7′,7′-bis(trifluoromethyl)spiro[1,3,2-benzodioxaphosphole-2,1′-[2,6,8]trioxa[1λ5]phosphabicyclo[3.2.1]octane] and 3′,5′-dimethyl-7′,7′-bis(trifluoromethyl)spiro[1,3,2-benzodioxaphosphole-2,1′-[2,6,8]trioxa[1λ5]phosphabicyclo[3.2.1]oct[3]ene]](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Mironov V.F., Baronova T.A., Mironova E.V., Dimukhametov M.N., Krivolapov D.B., Abdrakhmanova L.M.
Russian Journal of Organic Chemistry,
2015
35.
A. R. M. Bazouin, J. P. Dunogues and M. J. C. Lefort, Patent US 3472888, 1969.
36.

Pudovik M.A., Ovchinnikov V.V., Cherkasov R.A., Pudovik A.N.
Russian Chemical Reviews,
1983
37.

38.

Khasiyatullina N.R., Baronova T.A., Mironova E.V., Fayzullin R.R., Litvinov I.A., Efimov S.V., Musin R.Z., Klochkov V.V., Mironov V.F.
Organic Chemistry Frontiers,
2018
39.

Spek A.L.
Acta Crystallographica Section D Biological Crystallography,
2009
40.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
41.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015
42.

Farrugia L.J.
Journal of Applied Crystallography,
2012
43.

Flack H.D., Bernardinelli G.
Journal of Applied Crystallography,
2000