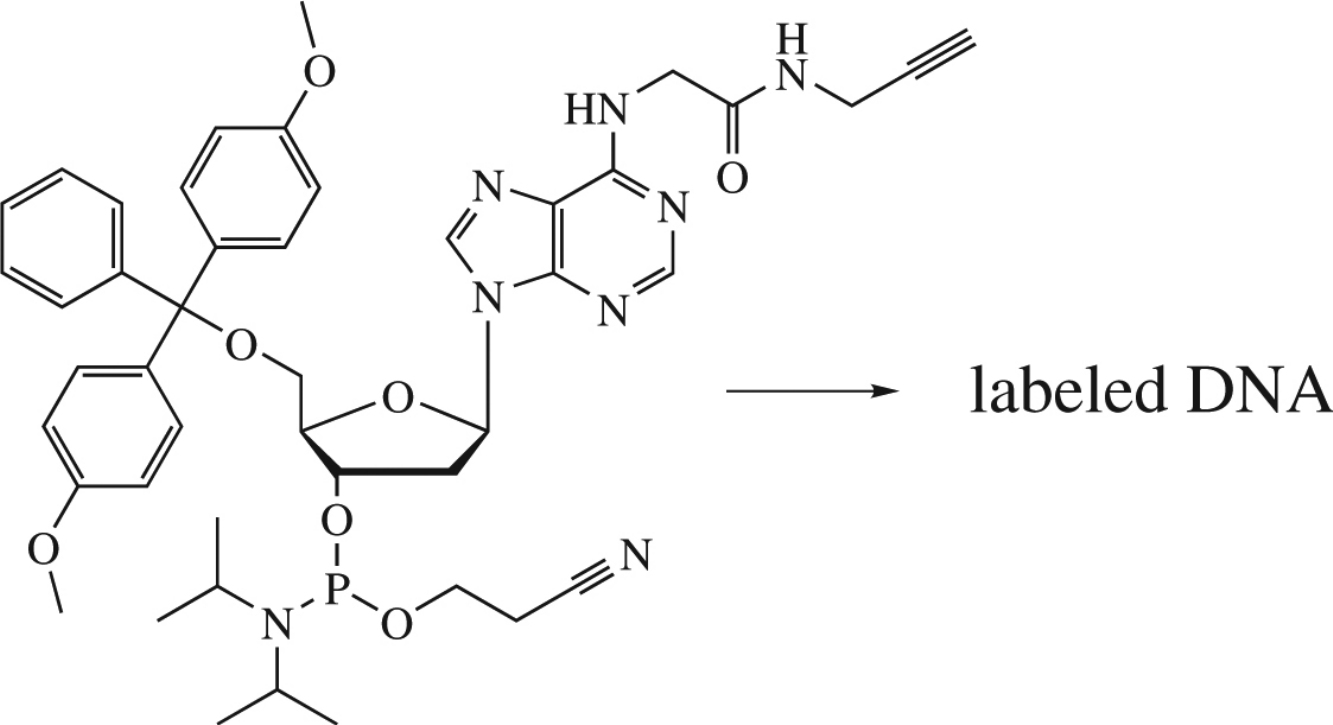
Abstract
A new 5′-dimethoxytrityl N6-alkynylated 2′-deoxyadenosine phosphoramidite has been designed and synthesized from 2′-deoxyadenosine. The applicability of this building block for the production of alkyne-modified oligodeoxynucleotide has been demonstrated. This allows one to introduce various labels into DNA using the click reaction.
References
1.

Sletten E.M., Bertozzi C.R.
Accounts of Chemical Research,
2011
2.

3.

4.

Huisgen R., Szeimies G., Möbius L.
Chemische Berichte,
1967
5.

Kolb H.C., Finn M.G., Sharpless K.B.
Angewandte Chemie - International Edition,
2001
6.

Rostovtsev V.V., Green L.G., Fokin V.V., Sharpless K.B.
Angewandte Chemie - International Edition,
2002
7.
![Peptidotriazoles on Solid Phase: [1,2,3]-Triazoles by Regiospecific Copper(I)-Catalyzed 1,3-Dipolar Cycloadditions of Terminal Alkynes to Azides](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Tornøe C.W., Christensen C., Meldal M.
Journal of Organic Chemistry,
2002
8.

Astakhova I.K., Wengel J.
Chemistry - A European Journal,
2012
9.

10.

Gramlich P. ., Warncke S., Gierlich J., Carell T.
Angewandte Chemie - International Edition,
2008
11.

Gierlich J., Burley G.A., Gramlich P.M., Hammond D.M., Carell T.
Organic Letters,
2006
12.

13.

Seela F., Sirivolu V.
Chemistry and Biodiversity,
2006
14.

Berndl S., Herzig N., Kele P., Lachmann D., Li X., Wolfbeis O.S., Wagenknecht H.
Bioconjugate Chemistry,
2009
15.

Lietard J., Meyer A., Vasseur J., Morvan F.
Journal of Organic Chemistry,
2007
16.

Okamoto A., Tainaka K., Ochi Y., Kanatani K., Saito I.
Molecular BioSystems,
2006
17.

Seela F., Pujari S.S.
Bioconjugate Chemistry,
2010
18.

Ogasawara S., Saito I., Maeda M.
Tetrahedron Letters,
2008
19.
![Synthesis of oligonucleotides containing pyrazolo[3,4-d ]pyrimidines: The influence of 7-substituted 8-aza-7-deazaadenines on the duplex structure and stability](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Seela F., Zulauf M.
Journal of the Chemical Society Perkin Transactions 1,
1999
20.

Van Berkel G.J.
European Journal of Mass Spectrometry,
2003
21.

22.

Montalbetti C.A., Falque V.
Tetrahedron,
2005
23.

Seo Y.J., Rhee H., Joo T., Kim B.H.
Journal of the American Chemical Society,
2007
24.

Jain S.S.
Nucleic Acids Research,
2003