Abstract
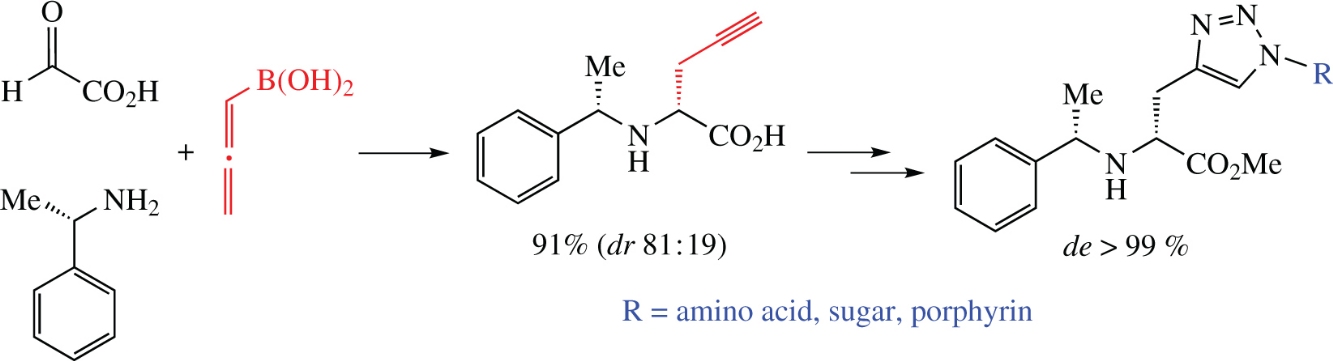
The Petasis–Mannich reaction of allenylboronic acid with glyoxylic acid and (S)-1-phenylethylamine diastereoselectively leads to N-substituted propargylglycine of high optical purity in good yield. This product can be further subjected to esterification followed by bioconjugate synthesis using CuI-catalyzed alkyne–azide 1,3-dipolar cycloaddition reaction (‘click reaction’).
References
1.

Candeias N.R., Montalbano F., Cal P.M., Gois P.M.
Chemical Reviews,
2010
2.

Petasis N.A., Akritopoulou I.
Tetrahedron Letters,
1993
3.
10.1016/j.mencom.2019.09.006_bib0015
Petasis
Multicomponent Reactions,
2005
4.

Kaiser P.F., Churches Q.I., Hutton C.A.
Australian Journal of Chemistry,
2007
5.

Pye P.J., Rossen K., Weissman S.A., Maliakal A., Reamer R.A., Ball R., Tsou N.N., Volante R.P., Reider P.J.
Chemistry - A European Journal,
2002
6.

Hong Z., Liu L., Hsu C., Wong C.
Angewandte Chemie - International Edition,
2006
7.

Davis A.S., Pyne S.G., Skelton B.W., White A.H.
Journal of Organic Chemistry,
2004
8.

Sugiyama S., Arai S., Ishii K.
Tetrahedron Asymmetry,
2004
9.
10.1016/j.mencom.2019.09.006_bib0045
Pyne
Org. React.,
2014
10.
10.1016/j.mencom.2019.09.006_bib0050
Ramadhar
Boronic Acids,
2011
11.

Flagstad T., Hansen M.R., Le Quement S.T., Givskov M., Nielsen T.E.
ACS Combinatorial Science,
2014
12.

Matthies S., Stallforth P., Seeberger P.H.
Journal of the American Chemical Society,
2015
13.

Zhang J., Yun F., Xie R., Cheng C., Chen G., Li J., Tang P., Yuan Q.
Tetrahedron Letters,
2016
14.

Manolikakes G., Diehl A., Ouadoudi O., Andreadou E.
Synthesis,
2018
15.
10.1016/j.mencom.2019.09.006_bib0075
Reddy
RCS Adv.,
2016
16.

Candeias N.R., Veiros L.F., Afonso C.A., Gois P.M.
European Journal of Organic Chemistry,
2009
17.

Thaima T., Pyne S.G.
Organic Letters,
2015
18.

Liepouri F., Bernasconi G., Petasis N.A.
Organic Letters,
2015
19.

Thirumurugan P., Matosiuk D., Jozwiak K.
Chemical Reviews,
2013
20.

Kuijpers B.H., Groothuys S., Soede A.C., Laverman P., Boerman O.C., van Delft F.L., Rutjes F.P.
Bioconjugate Chemistry,
2007
21.

Tiwari V.K., Mishra B.B., Mishra K.B., Mishra N., Singh A.S., Chen X.
Chemical Reviews,
2016
22.

Ikeda N., Arai I., Yamamoto H.
Journal of the American Chemical Society,
1986
23.

Morozova V.A., Beletskaya I.P., Titanyuk I.D.
Tetrahedron Asymmetry,
2017