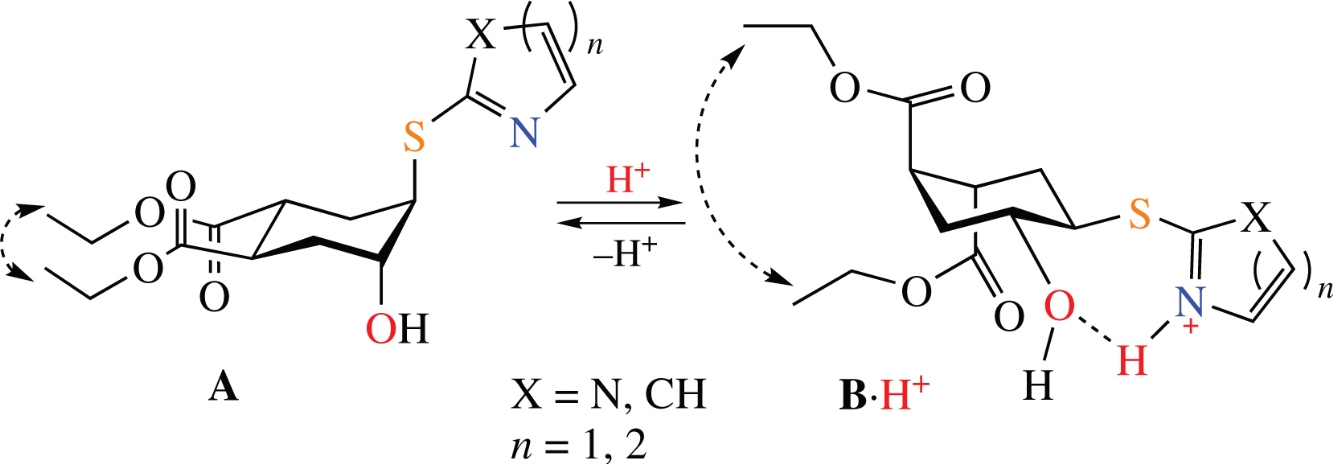
Abstract
A series of trans-2-(azaarylsulfanyl)cyclohexanol derivatives, structurally similar to previously studied trans-2-amino-cyclohexanols, were synthesized through epoxide ring opening under basic conditions with sodium tetraborate as a catalyst. 1H NMR spectroscopy was used to elucidate the conformational equilibrium in various solvents and its acid-induced change due to stabilization of the conformer with the azaarylsulfanyl and hydroxy groups in equatorial position by an intramolecular hydrogen bond and electrostatic interactions.
References
1.

Lou J., Zhang X., Best M.D.
Chemistry - A European Journal,
2018
2.

Knipe P.C., Thompson S., Hamilton A.D.
Chemical Science,
2015
3.
10.1016/j.mencom.2019.09.005_bib0015
Applications of Supramolecular Chemistry,
2012
4.

Kay E., Leigh D., Zerbetto F.
Angewandte Chemie - International Edition,
2007
5.

Samoshin V.V.
Biomolecular Concepts,
2014
6.

Zheng Y., Liu X., Samoshina N.M., Samoshin V.V., Franz A.H., Guo X.
Chemistry and Physics of Lipids,
2018
7.

Samoshin V.V., Zheng Y., Liu X.
Journal of Physical Organic Chemistry,
2017
8.

Zefirov N.S., Gurvich L.G., Shashkov A.S., Krimer M.Z., Vorob'eva E.A.
Tetrahedron,
1976
9.
10.1016/j.mencom.2019.09.005_bib0045
Samoshin
Russ. J. Org. Chem.,
1993
10.

Booth H.
Progress in Nuclear Magnetic Resonance Spectroscopy,
1969
11.
10.1016/j.mencom.2019.09.005_bib0055
Eliel
Chem. Ind.,
1959