Abstract
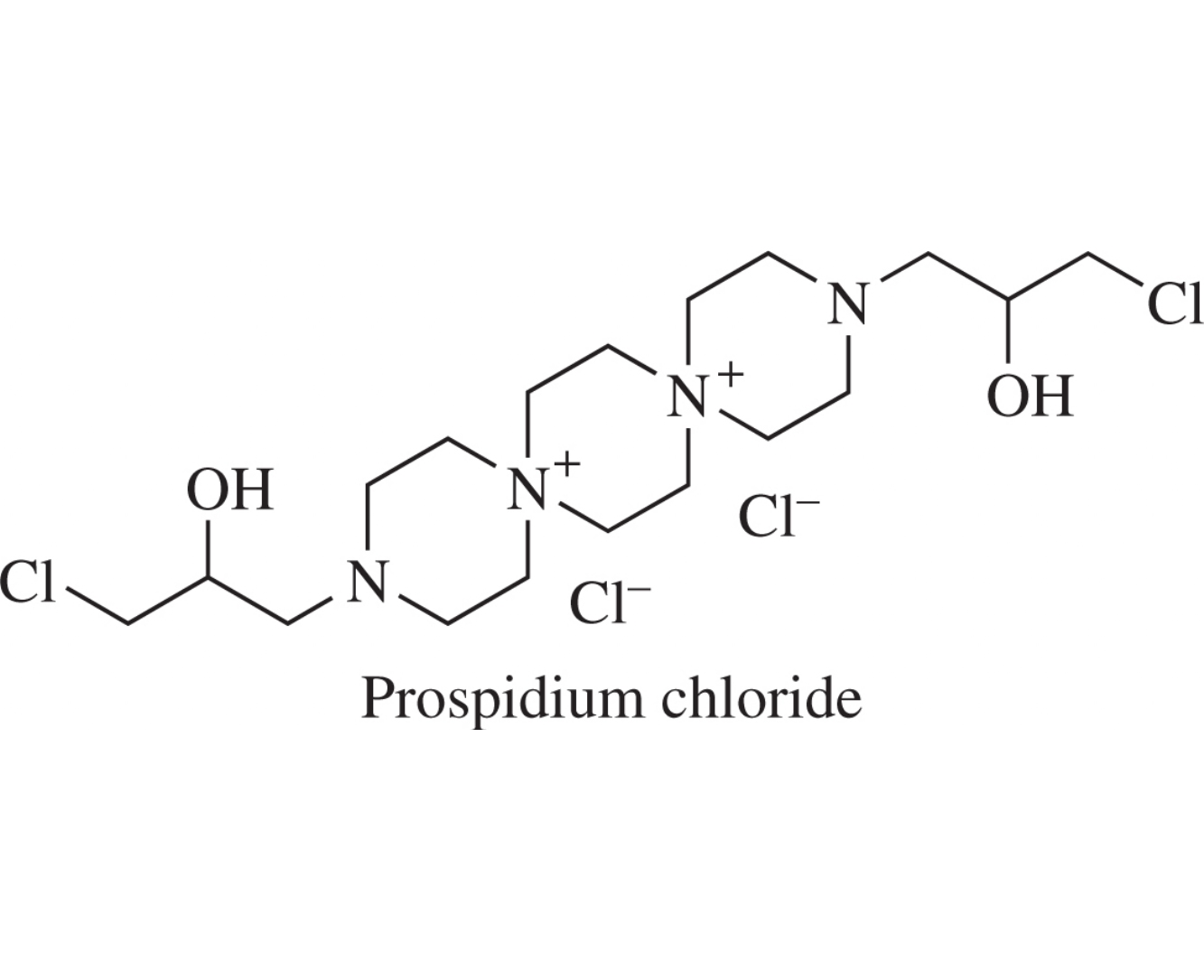
The thermochemical behavior of well-established heterocyclic anticancer drug, prospidium chloride, was comparatively evaluated in water and two other non-aqueous solvents, formamide and ethylene glycol, forming the three dimensional H-bond network. Standard enthalpies and heat capacities of solution, as well as, enthalpic parameters of solute–solute pair interactions were estimated at 298 and 318K and compared with the available values for some ionic species. A linear correlation between the energetics of ion–ion interactions and the reciprocal dielectric constant of the medium for tetraethylammonium bromide and prospidium chloride was revealed.
References
1.
10.1016/j.mencom.2019.07.029_bib0005
Sharp
Encyclopedia of Life Sciences,
2001
2.
10.1016/j.mencom.2019.07.029_bib0010
Robinson
Water in Biology, Chemistry and Physics. Experimental Overviews and Computational Methodologies,
1996
3.

Chumaevskii N.A., Rodnikova M.N.
Journal of Molecular Liquids,
2003
4.

Chandler D.
Nature,
2005
5.
10.1016/j.mencom.2019.07.029_bib0025
Kessler
Solvophobic Effects,
1994
6.

Craft Q.D., Wood R.H.
Journal of Solution Chemistry,
1977
7.

Sjoeberg M., Silveston R., Kronberg B.
Langmuir,
1993
8.

Chumaevskii N.A., Rodnikova M.N., Barthel J.
Journal of Molecular Liquids,
2004
9.

Rodnikova M.N., Idiyatullin Z.S., Barthel J., Solonina I.A., Sirotkin D.A.
Journal of Molecular Liquids,
2017
10.

Kustov A.V., Smirnova N.L.
Journal of Physical Chemistry B,
2011
11.
10.1016/j.mencom.2019.07.029_bib0055
Grohn
Cancer Treat. Rep.,
1984
12.

Soldatski I.L., Onufrieva E.K., Steklov A.M., Schepin N.V.
Laryngoscope,
2005
13.

Rojo J.M., Ojeda G., Portolés M.P., Portolés A.
Chemotherapy,
1983
14.

Kustov A.V., Antonova O.A., Smirnova N.L., Kladiev A.A., Kladiev A.A., Kudayarova T.V., Gruzdev M.S., Berezin D.B.
Journal of Molecular Liquids,
2018
15.

Kustov A.V., Belykh D.V., Smirnova N.L., Venediktov E.A., Kudayarova T.V., Kruchin S.O., Khudyaeva I.S., Berezin D.B.
Dyes and Pigments,
2018
16.

Korolev V.P., Antonova O.A., Smirnova N.L., Kustov A.V.
Journal of Thermal Analysis and Calorimetry,
2010
17.

Korolev V.P., Antonova O.A., Smirnova N.L., Kustov A.V.
Journal of Thermal Analysis and Calorimetry,
2010
18.

Kustov A.V., Smirnova N.L., Antonova O.A., Korolev V.P.
Russian Journal of Physical Chemistry A,
2008
19.

Gallagher K.R., Sharp K.A.
Journal of the American Chemical Society,
2003
20.

Kustov A.V., Smirnova N.L., Antonova O.A.
Journal of Chemical Thermodynamics,
2019
21.

Huang N., Schlesinger D., Nordlund D., Huang C., Tyliszczak T., Weiss T.M., Acremann Y., Pettersson L.G., Nilsson A.
Journal of Chemical Physics,
2012