Abstract
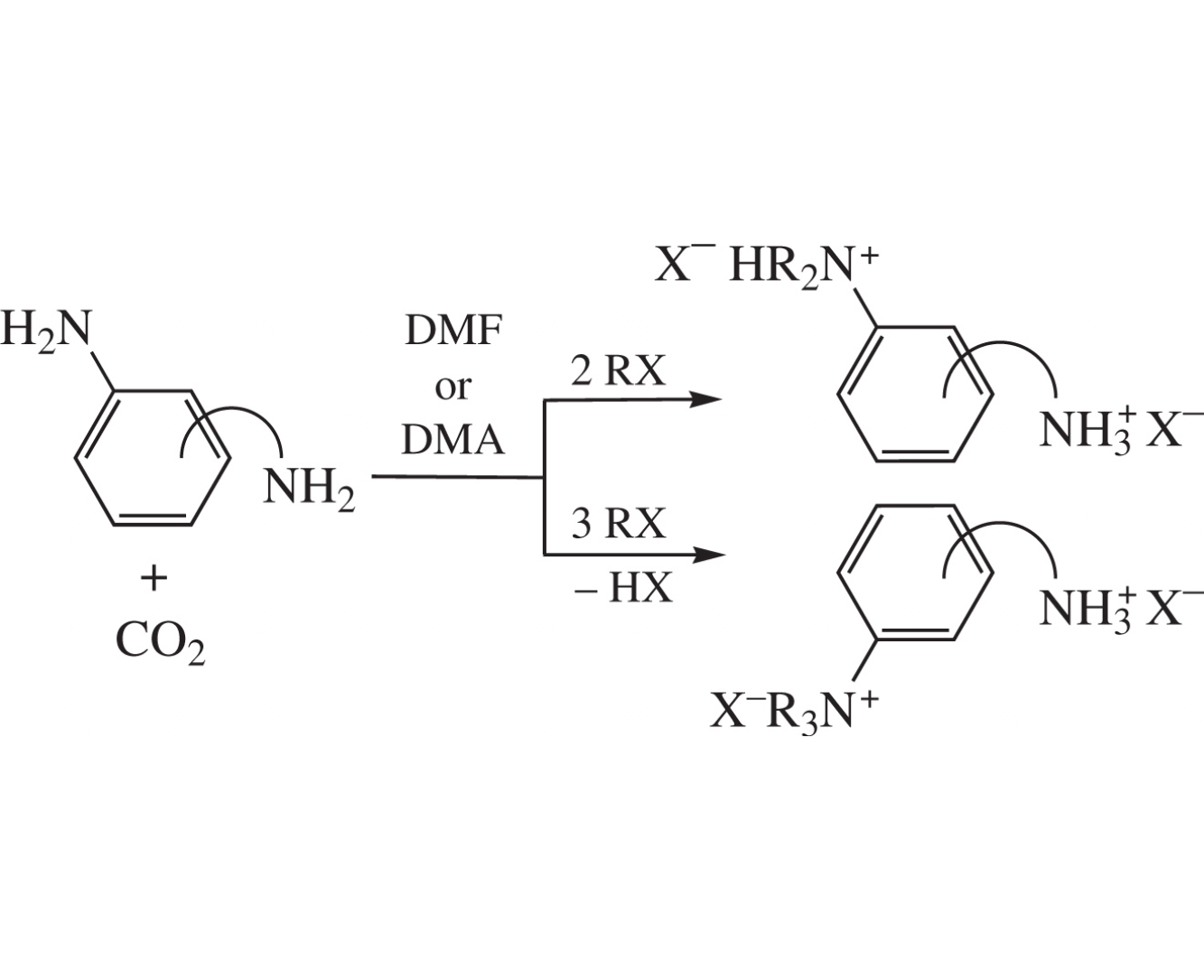
Selective Hofmann alkylation at arylamino group in carbon dioxide medium was demonstrated on model diamines containing aliphatic and aromatic primary amino groups, namely, 2-H2NC6H4CH2NH2, 3-H2NC6H4CH(Me)NH2, and 4-H2NC6H4CH2CH2NH2. Depending on the spatial factors, di- or trialkylarylammonium derivatives are selectively formed in amide solvents (DMF, DMA) without additional base.
References
1.

Xie X., Liotta C.L., Eckert C.A.
Industrial & Engineering Chemistry Research,
2004
2.

Wittmann K., Wisniewski W., Mynott R., Leitner W., Kranemann C.L., Rische T., Eilbracht P., Kluwer S., Ernsting J.M., Elsevier C.J.
Chemistry - A European Journal,
2001
3.

Peeters A., Ameloot R., De Vos D.E.
Green Chemistry,
2013
4.

Ethier A., Switzer J., Rumple A., Medina-Ramos W., Li Z., Fisk J., Holden B., Gelbaum L., Pollet P., Eckert C., Liotta C.
Processes,
2015
5.

Fürstner A., Ackermann L., Beck K., Hori H., Koch D., Langemann K., Liebl M., Six C., Leitner W.
Journal of the American Chemical Society,
2001
6.

Mohammed F., Kitchens C.
Molecules,
2015
7.

Speckmeier E., Klimkait M., Zeitler K.
Journal of Organic Chemistry,
2018
8.

Riemer D., Hirapara P., Das S.
ChemSusChem,
2016
9.
10.1016/j.mencom.2019.07.028_bib0015
Quaranta
Carbon Dioxide as Chemical Feedstock,
2010
10.

Hampe E.M., Rudkevich D.M.
Tetrahedron,
2003
11.

Masuda K., Ito Y., Horiguchi M., Fujita H.
Tetrahedron,
2005
12.

Maran F., Celadon D., Severin M.G., Vianello E.
Journal of the American Chemical Society,
1991
13.

Sosnovsky G., Konieczny M.
Zeitschrift fur Naturforschung - Section B Journal of Chemical Sciences,
1978
14.

Diesendruck C., Arava S.
Synthesis,
2017
15.

BENINGTON F., MORIN R.D., CLARK L.C.
Journal of Organic Chemistry,
1956