Abstract
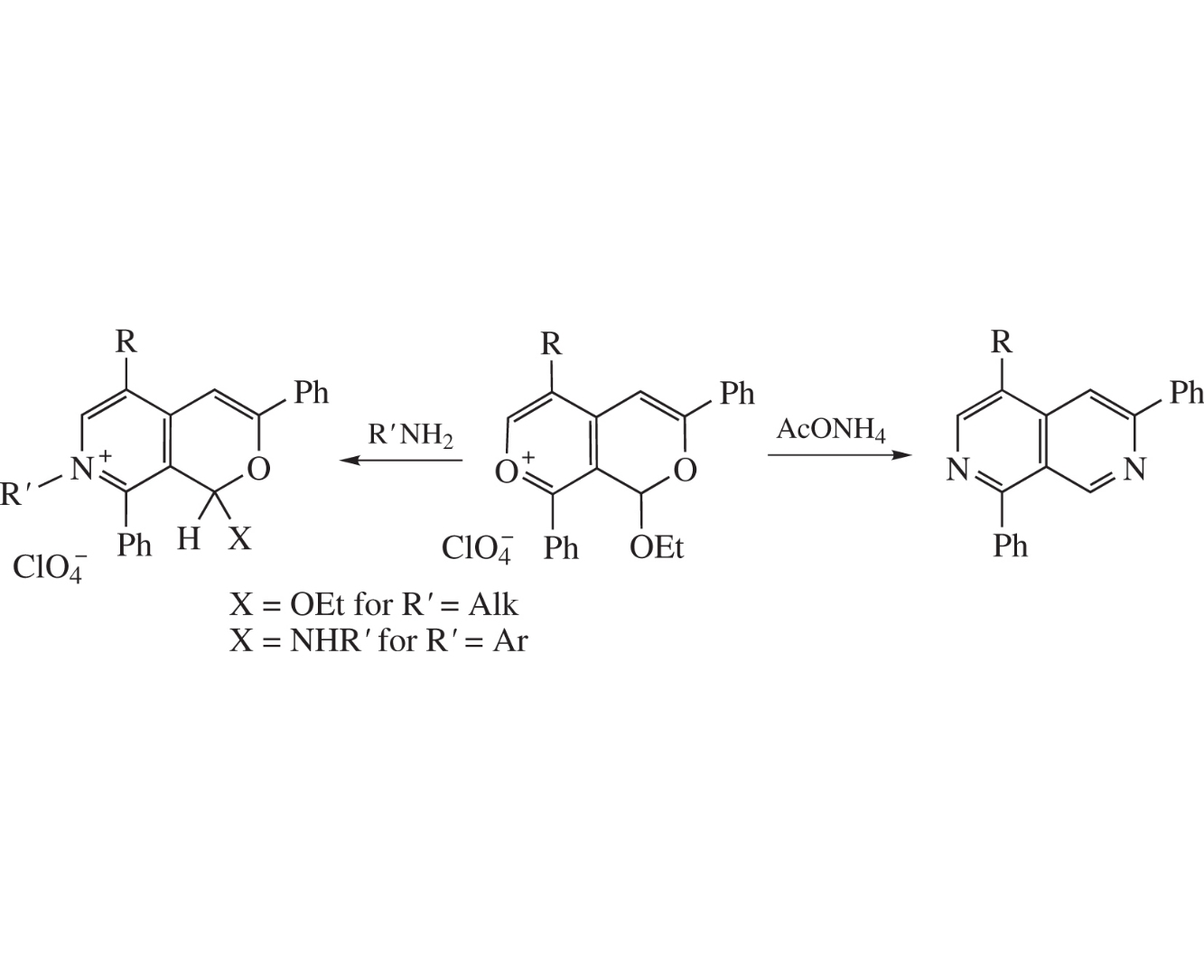
The reaction of 1H-pyrano[3,4-c]pyran-7-ium perchlorates with ammonium acetate gives 2,7-naphthyridines, while in their reaction with primary amines pyrano[3,4-c]pyridinium salts are formed. The structure of 7-[2-(dimethylamino)ethyl]-1-ethoxy-5-methyl-3,8-diphenyl-1H-pyrano[3,4-c]pyridin-7-ium perchlorate was established by X-ray diffraction.
References
1.
![Unexpected pyrylium to pyrylium domino transformation. Synthesis of pyrano[3,4-c]pyran-7-ium cation and its recyclization to 2,7-naphthyridine derivative](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Suzdalev K.F., Krachkovskaya A.V., Kletskii M.E., Burov O.N., Tatarov A.V., Kurbatov S.V.
Chemistry of Heterocyclic Compounds,
2017
2.
10.1016/j.mencom.2019.07.026_sbref0010a
Balaban
1982
3.

Toma C., Balaban A.T.
Tetrahedron,
1966
4.
10.1016/j.mencom.2019.07.026_sbref0010c
Heidarizadeh
Orient. J. Chem.,
2011
5.

Kwak W., Kim J., Ryu K., Cho Y., Jeon S., Moon C.
Biological and Pharmaceutical Bulletin,
2005
6.

Gross H., Goeger D.E., Hills P., Mooberry S.L., Ballantine D.L., Murray T.F., Valeriote F.A., Gerwick W.H.
Journal of Natural Products,
2006
7.
10.1016/j.mencom.2019.07.026_sbref0025a
Wójcicka
Acta Pol. Pharm.,
2015
8.
![Discovery of Dibenzo[c,f][2,7]naphthyridines as Potent and Selective 3-Phosphoinositide-Dependent Kinase-1 Inhibitors](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Gopalsamy A., Shi M., Boschelli D.H., Williamson R., Olland A., Hu Y., Krishnamurthy G., Han X., Arndt K., Guo B.
Journal of Medicinal Chemistry,
2007
9.

Kluza J., Mazinghien R., Degardin K., Lansiaux A., Bailly C.
European Journal of Pharmacology,
2005
10.

Kang K., Kim H., Kim K.I., Yang Y., Yoon D., Kim J., Ryu J., Noh E., Jeon S., Lim J.
Biochemical Pharmacology,
2008
11.

Nguyen S.T., Kwasny S.M., Ding X., Cardinale S.C., McCarthy C.T., Kim H., Nikaido H., Peet N.P., Williams J.D., Bowlin T.L., Opperman T.J.
Bioorganic and Medicinal Chemistry,
2015
12.

Sjuts H., Vargiu A.V., Kwasny S.M., Nguyen S.T., Kim H., Ding X., Ornik A.R., Ruggerone P., Bowlin T.L., Nikaido H., Pos K.M., Opperman T.J.
Proceedings of the National Academy of Sciences of the United States of America,
2016
13.

Cragg G.M., Newman D.J.
Biochimica et Biophysica Acta - General Subjects,
2013
14.

Becke A.D.
Physical Review A,
1988
15.

Becke A.D.
Journal of Chemical Physics,
1993
16.

Lee C., Yang W., Parr R.G.
Physical Review B,
1988
17.

Stevens J., Schweizer M., Rauhut G.
Journal of the American Chemical Society,
2001
18.
10.1016/j.mencom.2019.07.026_bib0055
Frisch
Gaussian 03, Revision C.02,
2004
19.

Bernhard Schlegel H.
Theoretical Chemistry Accounts,
1984
20.

Tomasi J., Mennucci B., Cammi R.
Chemical Reviews,
2005