Abstract
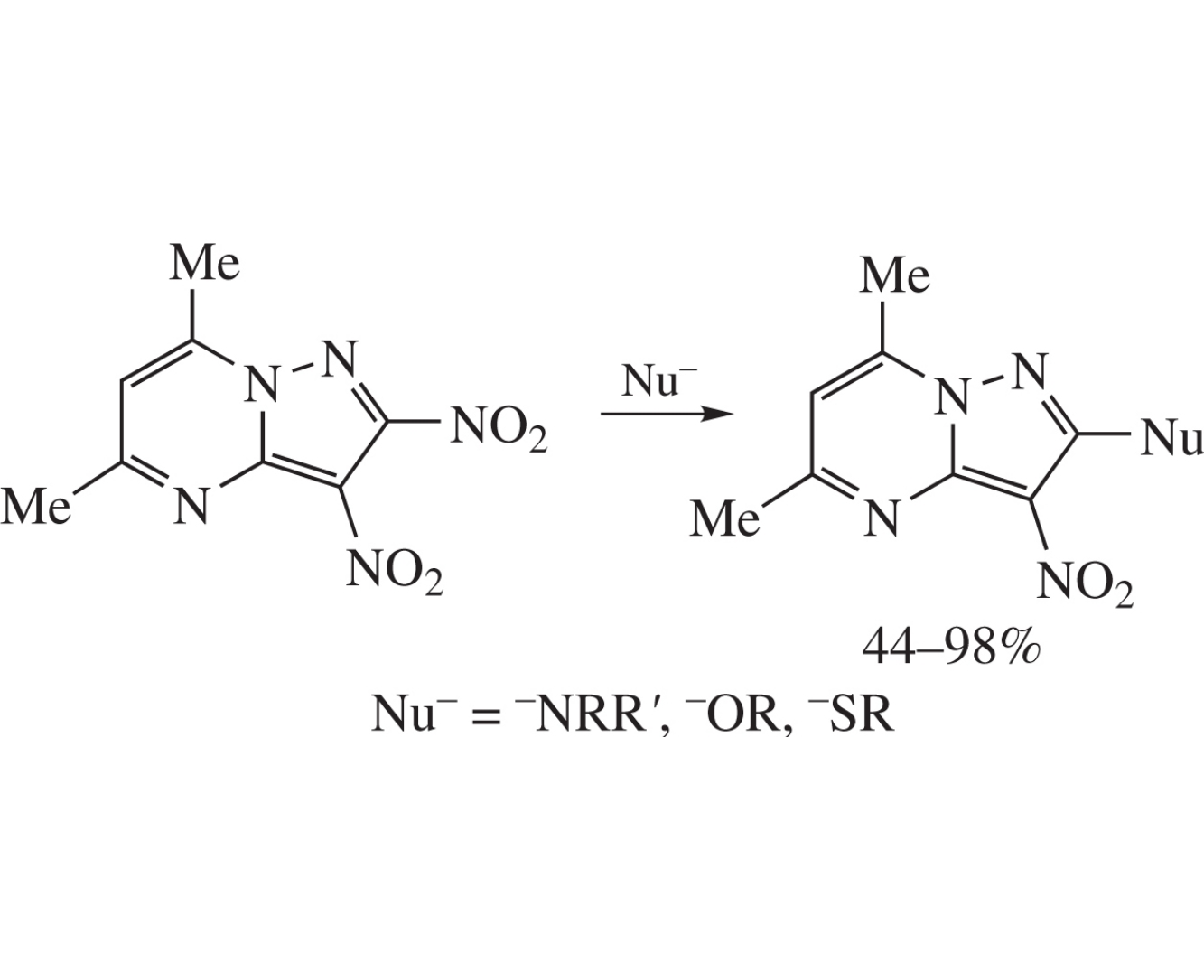
5,7-Dimethyl-2,3-dinitropyrazolo[1,5-a]pyrimidine in reaction with N-, S- and O-nucleophiles under mild conditions undergoes regioselective nucleophilic substitution of the 2-positioned nitro group, which provides an access to a library of 2-R-3-nitropyrazolo[1,5-a]pyrimidines.
References
1.

Ramsey S.J., Attkins N.J., Fish R., van der Graaf P.H.
British Journal of Pharmacology,
2011
2.

Petroski R.E., Pomeroy J.E., Das R., Bowman H., Yang W., Chen A.P., Foster A.C.
Journal of Pharmacology and Experimental Therapeutics,
2006
3.
J. P. Dusza, A.S. Tomcufcik and J. D. Albright, Patent US 4654347, 1987.
4.

El-Naggar M., Hassan A., Awad H., Mady M.
Molecules,
2018
5.
![Synthesis andin vitroanticancer activity of pyrazolo[1,5-a]pyrimidines and pyrazolo[3,4-d][1,2,3]triazines](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Hassan A.S., Moustafa G.O., Awad H.M.
Synthetic Communications,
2017
6.
![Synthesis and biological evaluation of chalcone-linked pyrazolo[1,5-a]pyrimidines as potential anticancer agents](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Bagul C., Rao G.K., Makani V.K., Tamboli J.R., Pal-Bhadra M., Kamal A.
MedChemComm,
2017
7.
10.1016/j.mencom.2019.07.025_sbref0015a
Mohamed
Pharma Chem.,
2018
8.
![An efficient synthesis of pyrazolo[1,5-a]pyrimidines and evaluation of their antimicrobial activity](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
DESHMUKH S., DINGORE K., GAIKWAD V., JACHAK M.
Journal of Chemical Sciences,
2016
9.

Mackman R.L., Sangi M., Sperandio D., Parrish J.P., Eisenberg E., Perron M., Hui H., Zhang L., Siegel D., Yang H., Saunders O., Boojamra C., Lee G., Samuel D., Babaoglu K., et. al.
Journal of Medicinal Chemistry,
2015
10.

McCarthy M.J., Pagba C.V., Prakash P., Naji A.K., van der Hoeven D., Liang H., Gupta A.K., Zhou Y., Cho K., Hancock J.F., Gorfe A.A.
ACS Omega,
2019
11.
![Discovery of Pyrazolo[1,5-a]pyrimidine TTK Inhibitors: CFI-402257 is a Potent, Selective, Bioavailable Anticancer Agent](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Liu Y., Laufer R., Patel N.K., Ng G., Sampson P.B., Li S., Lang Y., Feher M., Brokx R., Beletskaya I., Hodgson R., Plotnikova O., Awrey D.E., Qiu W., Chirgadze N.Y., et. al.
ACS Medicinal Chemistry Letters,
2016
12.
![Identification, Synthesis, and Biological Evaluation of 6-[(6R)-2-(4-Fluorophenyl)-6-(hydroxymethyl)-4,5,6,7-tetrahydropyrazolo[1,5-a]pyrimidin-3-yl]-2-(2-methylphenyl)pyridazin-3(2H)-one (AS1940477), a Potent p38 MAP Kinase Inhibitor](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Asano T., Yamazaki H., Kasahara C., Kubota H., Kontani T., Harayama Y., Ohno K., Mizuhara H., Yokomoto M., Misumi K., Kinoshita T., Ohta M., Takeuchi. M.
Journal of Medicinal Chemistry,
2012
13.
![Discovery of pyrazolo[1,5-a]pyrimidine-3-carbonitrile derivatives as a new class of histone lysine demethylase 4D (KDM4D) inhibitors](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fang Z., Wang T., Li H., Zhang G., Wu X., Yang L., Peng Y., Zou J., Li L., Xiang R., Yang S.
Bioorganic and Medicinal Chemistry Letters,
2017
14.
![Synthesis and SAR of a New Series of COX-2-Selective Inhibitors: Pyrazolo[1,5-a]pyrimidines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Almansa C., de Arriba A.F., Cavalcanti F.L., Gómez L.A., Miralles A., Merlos M., García-Rafanell J., Forn J.
Journal of Medicinal Chemistry,
2001
15.

Patnaik S., Zheng W., Choi J.H., Motabar O., Southall N., Westbroek W., Lea W.A., Velayati A., Goldin E., Sidransky E., Leister W., Marugan J.J.
Journal of Medicinal Chemistry,
2012
16.

Qu C., Ding M., Zhu Y., Lu Y., Du J., Miller M., Tian J., Zhu J., Xu J., Wen M., Er-Bu A., Wang J., Xiao Y., Wu M., McManus O.B., et. al.
Journal of Medicinal Chemistry,
2017
17.

Blad C.C., van Veldhoven J.P., Klopman C., Wolfram D.R., Brussee J., Lane J.R., IJzerman A.P.
Journal of Medicinal Chemistry,
2012
18.
![2-Substituted 5,6-dimethyl-3-phenylsulfonyl-pyrazolo[1,5-a]pyrimidines: New series of highly potent and specific serotonin 5-HT6 receptor antagonists](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Ivachtchenko A.V., Golovina E.S., Kadieva M.G., Koryakova A.G., Mitkin O.D., Tkachenko S.E., Kysil V.M., Okun I.
European Journal of Medicinal Chemistry,
2011
19.
![An insight on synthetic and medicinal aspects of pyrazolo[1,5-a]pyrimidine scaffold](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Cherukupalli S., Karpoormath R., Chandrasekaran B., Hampannavar G.A., Thapliyal N., Palakollu V.N.
European Journal of Medicinal Chemistry,
2017
20.
![Medicinal attributes of pyrazolo[1,5-a]pyrimidine based scaffold derivatives targeting kinases as anticancer agents](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Ismail N.S., Ali G.M., Ibrahim D.A., Elmetwali A.M.
Future Journal of Pharmaceutical Sciences,
2016
21.
![Synthesis of 4,4'-dinitro-1H,1'H-[3,3'-bipyrazole]-5,5'-diamine](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Shkineva T.K., Kormanov A.V., Boldinova V.N., Vatsadze I.A., Dalinger I.L.
Chemistry of Heterocyclic Compounds,
2018
22.

Dalinger I.L., Vatsadze I.A., Shkineva T.K., Popova G.P., Shevelev S.A., Nelyubina Y.V.
Journal of Heterocyclic Chemistry,
2013
23.

Dalinger I., Vatsadze I., Shkineva T., Popova G., Shevelev S.
Synthesis,
2012
24.

Dalinger I.L., Vatsadse I.A., Shkineva T.K., Popova G.P., Ugrak B.I., Shevelev S.A.
Russian Chemical Bulletin,
2010
25.
Starosotnikov A.M., Shkaev D.V., Bastrakov M.A., Fedyanin I.V., Shevelev S.A., Dalinger I.L.
Mendeleev Communications,
2018
26.

Dalinger I.L., Suponitsky K.Y., Shkineva T.K., Lempert D.B., Sheremetev A.B.
Journal of Materials Chemistry A,
2018
27.

Dalinger I.L., Kormanov A.V., Suponitsky K.Y., Muravyev N.V., Sheremetev A.B.
Chemistry - An Asian Journal,
2018
28.

Dalinger I.L., Serushkina O.V., Muravyev N.V., Meerov D.B., Miroshnichenko E.A., Kon'kova T.S., Suponitsky K.Y., Vener M.V., Sheremetev A.B.
Journal of Materials Chemistry A,
2018
29.
Zlotin S.G., Churakov A.M., Dalinger I.L., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2017
30.

Zaitsev A.A., Dalinger I.L., Shevelev S.A.
Russian Chemical Reviews,
2009
31.

Makarov V.A., Anisimova O.S., Granik V.G.
Chemistry of Heterocyclic Compounds,
1997
32.

Solov'eva N.P., Makarov V.A., Granik V.G.
Chemistry of Heterocyclic Compounds,
1997