Abstract
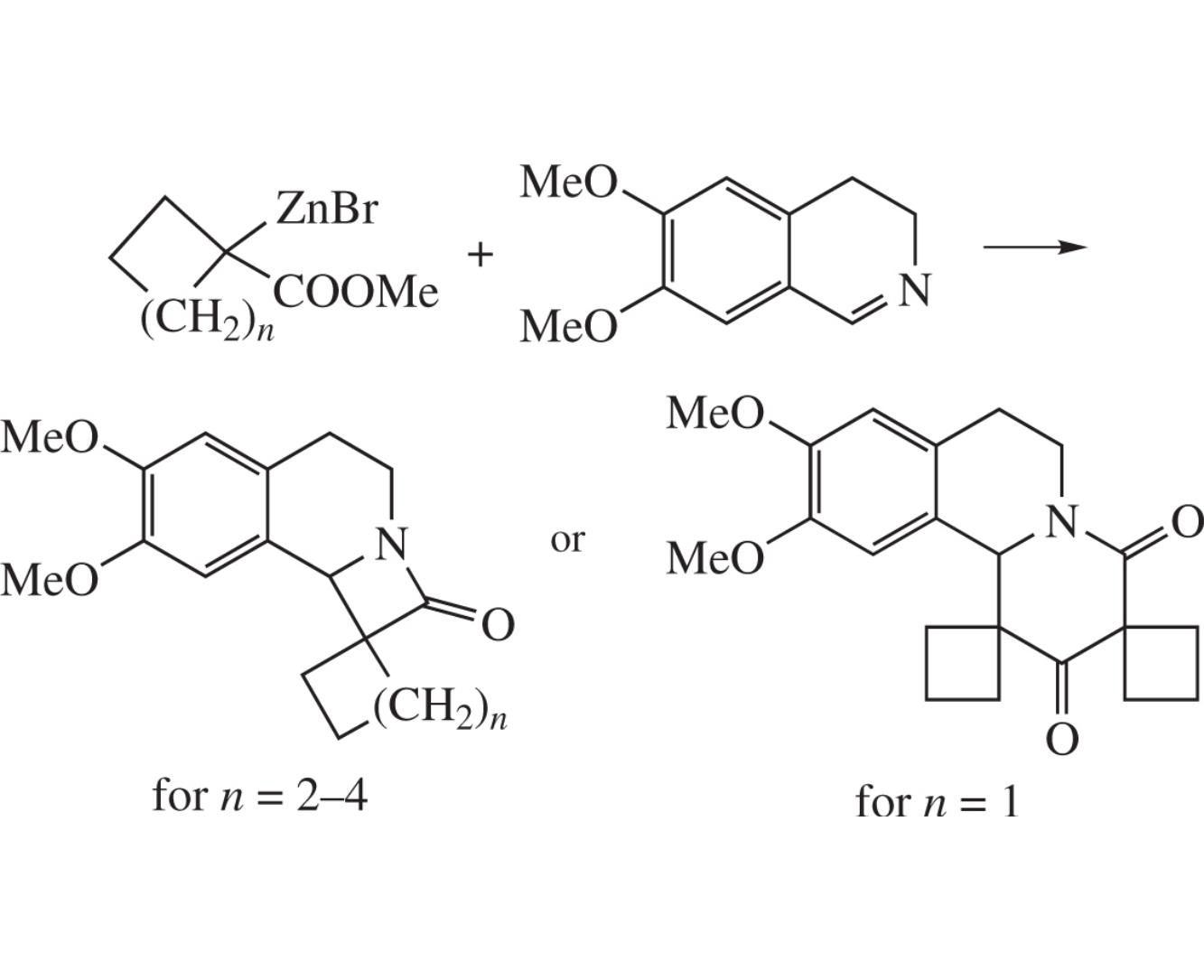
The reaction of 5–7-membered methyl 1-bromocycloalkanecarboxylates with zinc and 6,7-dimethoxy-3,4-dihydroisoquinoline leads to 7,8-dimethoxy-5,9b-dihydro-2H,4H-spiro{azeto[2,1-a]isoquinoline-1,1′-cycloalkan}-2-ones. In the case of 1-bromocyclobutanecarboxylate, 9′,10′-dimethoxy-7′,11b′-dihydro-2′H,4′H,6′H-dispiro{cyclobutane-1,1′-pyrido[2,1-a]isoquinoline-3′,1″-cyclobutane}-2′,4′-dione is formed. The structures of the products were confirmed by X-ray diffraction.
References
1.

Shchepin V.V., Melekhin V.S., Kirillov N.F.
Russian Journal of Organic Chemistry,
2007
2.

Wu G., Tormos W.
Journal of Organic Chemistry,
1997
3.

Benfatti F., Cardillo G., Gentilucci L., Tolomelli A.
European Journal of Organic Chemistry,
2007
4.

Ruchirawat S., Thasana N., Bjerke-Kroll B.
Synlett,
2008
5.

Bulletin of the Korean Chemical Society,
2007
6.

Hussey A.S., Newman M.S.
Journal of the American Chemical Society,
1948
7.

Mineno M., Sawai Y., Kanno K., Sawada N., Mizufune H.
Journal of Organic Chemistry,
2013
8.
![Synthesis of 16-aryl-15-oxadispiro[5.1.5.3]hexadecane-7,14-diones by reformatsky reaction](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Kirillov N.F., Nikiforova E.A., Vasyanin A.N., Dmitriev M.V.
Russian Journal of Organic Chemistry,
2015
9.
10.1016/j.mencom.2019.07.011_bib0045
Melekhin
2014
10.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007