Abstract
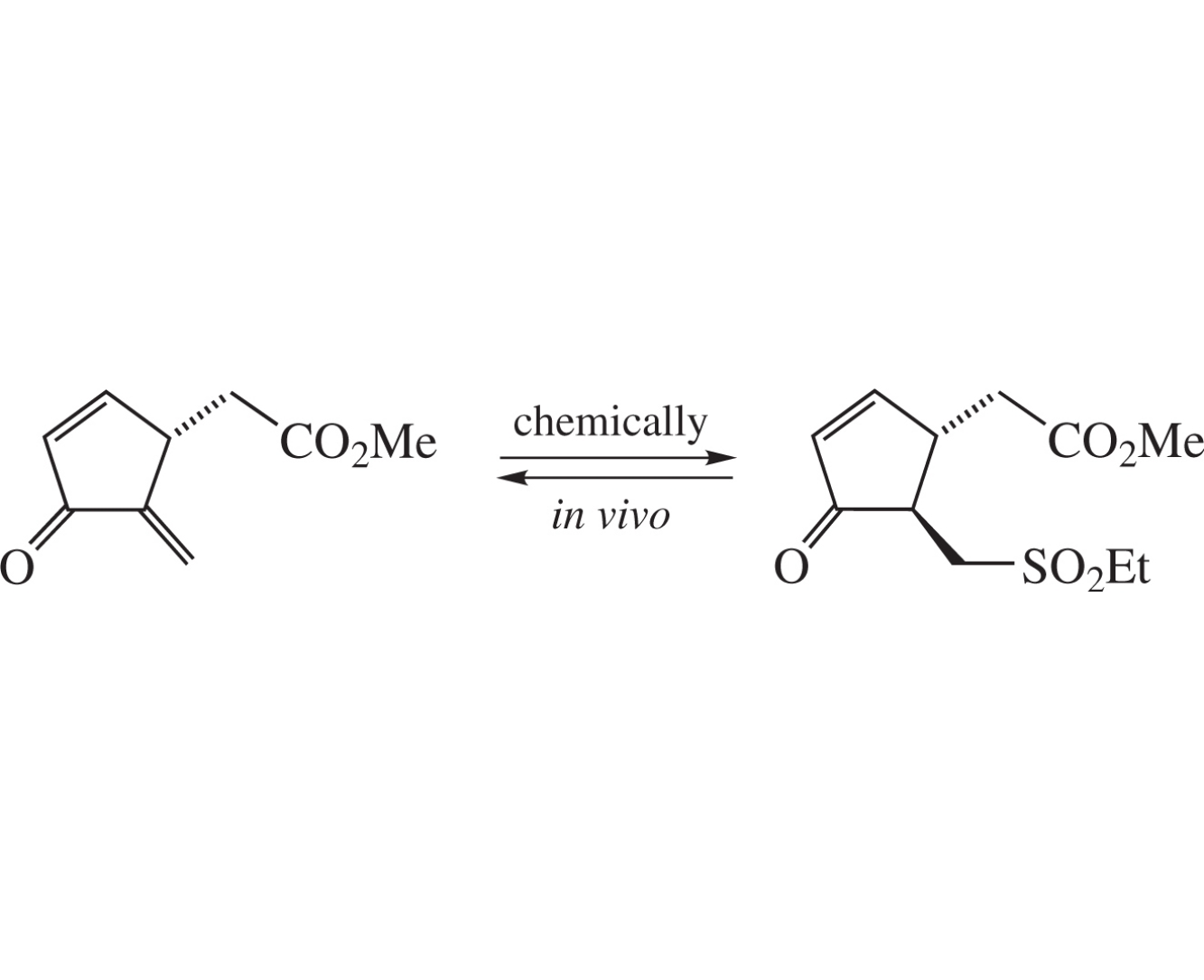
Unstable methyl (5-methylidene-4-oxocyclopent-2-en-1-yl)acetate, having a ring moiety of J-type prostaglandins (PGJs) and similar cytotoxicity, reacts with EtSH and other thiols with formation of mono-and bis-adducts, which have been further converted by oxidation with mCPBA into corresponding stable mono-sulfones possessing cytotoxic effect most probably due to in vivo regeneration of the starting dienone. Thus, the derived sulfones can be used as transport forms for original dienone as a pharmacologically important moiety of Δ12-PGJ2 and similar prostaglandins.
References
1.

Straus D.S., Glass C.K.
Medicinal Research Reviews,
2001
2.

Loza V.V., Gimazetdinov A.M., Miftakhov M.S.
Russian Journal of Organic Chemistry,
2018
3.

Das S., Chandrasekhar S., Yadav J.S., Grée R.
Chemical Reviews,
2007
4.

Conti M.
Anti-Cancer Drugs,
2006
5.

Fukushima S., Kishimoto S., Takeuchi Y., Fukushima M.
Advanced Drug Delivery Reviews,
2000
6.

Pica F., Palamara A.T., Rossi A., De Marco A., Amici C., Santoro M.G.
Antimicrobial Agents and Chemotherapy,
2000
7.

Shibata T., Kondo M., Osawa T., Shibata N., Kobayashi M., Uchida K.
Journal of Biological Chemistry,
2002
8.

Egger J., Fischer S., Bretscher P., Freigang S., Kopf M., Carreira E.M.
Organic Letters,
2015
9.

Pande V., Ramos M.J.
Bioorganic and Medicinal Chemistry Letters,
2005
10.

Suzuki M., Mori M., Niwa T., Hirata R., Furuta K., Ishikawa T., Noyori R.
Journal of the American Chemical Society,
1997
11.
10.1016/j.mencom.2019.07.003_bib0055
Fukushima
Eicosanoids,
1990
12.

Nicolaou K.C., Pulukuri K.K., Yu R., Rigol S., Heretsch P., Grove C.I., Hale C.R., ElMarrouni A.
Chemistry - A European Journal,
2016
13.

Kudva A.K., Kaushal N., Mohinta S., Kennett M.J., August A., Paulson R.F., Prabhu K.S.
PLoS ONE,
2013
14.

Nicolaou K.C., Pulukuri K.K., Rigol S., Heretsch P., Yu R., Grove C.I., Hale C.R., ElMarrouni A., Fetz V., Brönstrup M., Aujay M., Sandoval J., Gavrilyuk J.
Journal of the American Chemical Society,
2016
15.

Uchida K., Shibata T.
Chemical Research in Toxicology,
2007
16.

Scher J.U., Pillinger M.H.
Clinical Immunology,
2005
17.
Gimazetdinov A.M., Khalfitdinova L.A., Miftakhov M.S.
Mendeleev Communications,
2013
18.
10.1016/j.mencom.2019.07.003_bib0090
Tolstikov
J. Org. Chem. USSR (Engl. Transl.),
1984
19.
10.1016/j.mencom.2019.07.003_bib0095
Kato
Cancer Res.,
1986
20.

Noyori R., Suzuki M.
Science,
1993