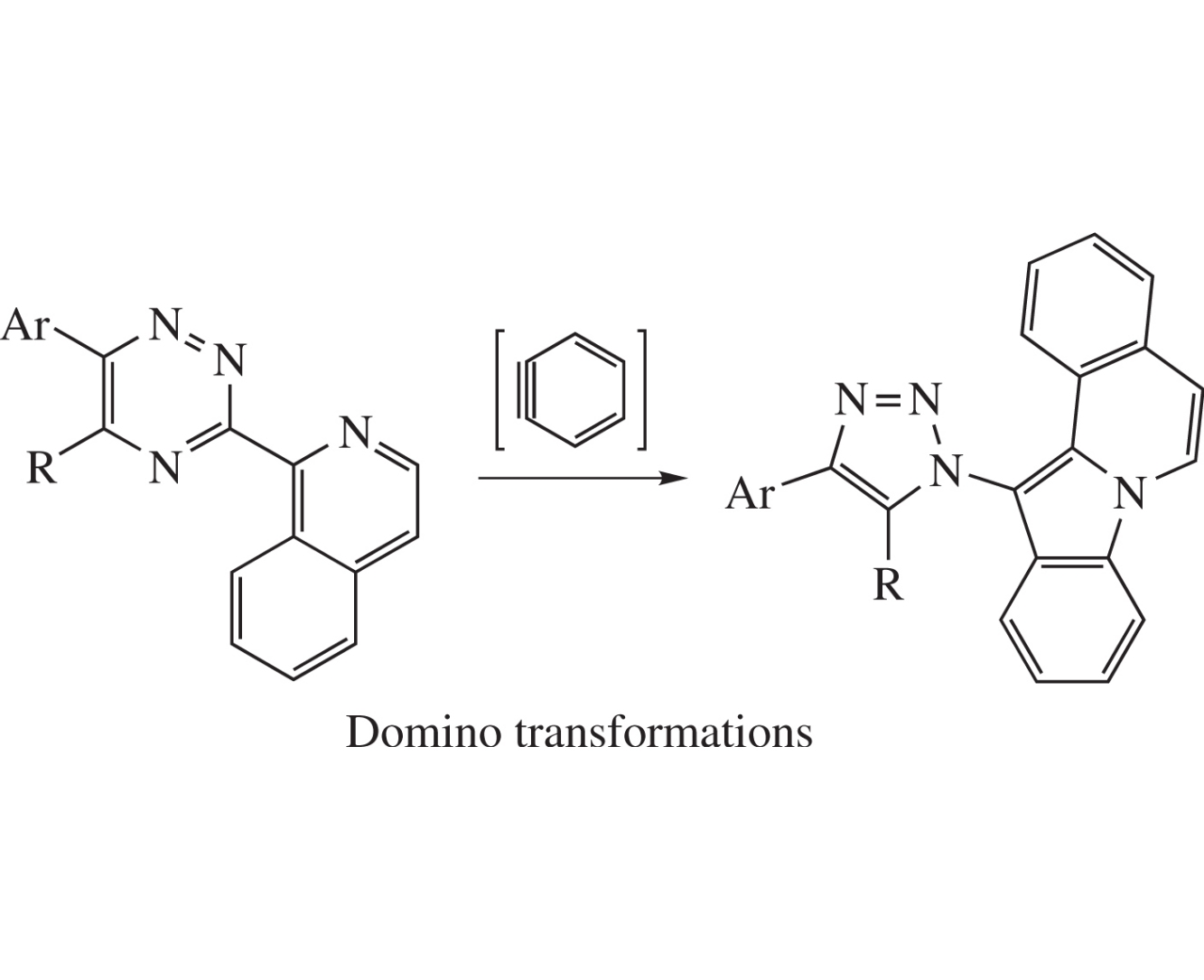
Abstract
12-(1H-1,2,3-Triazol-1-yl)indolo[2,1-a]isoquinolines are prepared in 51–56% yields using a PASE (pot, atom, step, economic)-based approach, namely, by the reaction between available 5-R-6-Ar-3-(isoquinolin-1-yl)-1,2,4-triazines and in situ generated benzyne. A mechanism comprising domino-transformation was suggested, and the structure of one key product was confirmed by a single crystal X-ray diffraction analysis.
References
1.
![C-12-Substituted Indolo[2,1-a]Isoquinolines as Estrogen Receptor Affinic Cytostatic Agents](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Ambros R., Von Angerer S., Wiegrebe W.
Archiv der Pharmazie,
1988
2.
M. Ogawa, T. Morita, N. Iibuchi, H. Suzuki, H. Tsutsui, A. Kano and S. Kato, Patent WO 9954325 A1, 1999.
3.
![Inhibition of Tubulin Polymerization by 5,6-Dihydroindolo[2,1-a]isoquinoline Derivatives](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Goldbrunner M., Loidl G., Polossek T., Mannschreck A., von Angerer E.
Journal of Medicinal Chemistry,
1997
4.
![Synthesis and Antitumor Activity of Methoxy-indolo[2,1-a]isoquinolines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Ambros R., Angerer S.V., Wiegrebe W.
Archiv der Pharmazie,
1988
5.
![A direct synthesis of 5,6-dihydroindolo[2,1-a]isoquinolines that exhibit immunosuppressive activity](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kraus G.A., Gupta V., Kohut M., Singh N.
Bioorganic and Medicinal Chemistry Letters,
2009
6.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Kartsev V.G., Borodkin G.S., Klimenko A.I.
Mendeleev Communications,
2018
7.

Ahmed E., Briseno A.L., Xia Y., Jenekhe S.A.
Journal of the American Chemical Society,
2008
8.

Wang L., Qu X., Li Z., Peng W.
Tetrahedron Letters,
2015
9.

Morimoto K., Hirano K., Satoh T., Miura M.
Organic Letters,
2010
10.

Zheng L., Hua R.
Chemistry - A European Journal,
2014
11.
![Ruthenium(II)-Catalyzed Indolo[2,1-a]isoquinolines Synthesis by Tandem C–H Allylation and Oxidative Cyclization of 2-Phenylindoles with Allyl Carbonates](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
12.

Alam K., Hong S.W., Oh K.H., Park J.K.
Angewandte Chemie - International Edition,
2017
13.

Ausekle E., Ejaz S.A., Khan S.U., Ehlers P., Villinger A., Lecka J., Sévigny J., Iqbal J., Langer P.
Organic and Biomolecular Chemistry,
2016
14.

Li S., Xia Y., Hu F., Liu C., Su F., Dong L.
Chemistry - An Asian Journal,
2016
15.

Florentino L., Aznar F., Valdés C.
Chemistry - A European Journal,
2013
16.
![Palladium and Copper Cocatalyzed Tandem N–H/C–H Bond Functionalization: Synthesis of CF3-Containing Indolo- and Pyrrolo[2,1-a]isoquinolines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Sun L., Liao Z., Tang R., Deng C., Zhang X.
Journal of Organic Chemistry,
2012
17.

Joshi M., Patel M., Tiwari R., Verma A.K.
Journal of Organic Chemistry,
2012
18.
![A Copper-Catalyzed Tandem Synthesis of Indolo- and Pyrrolo[2,1-a]isoquinolines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Verma A., Kesharwani T., Singh J., Tandon V., Larock R.
Angewandte Chemie - International Edition,
2009
19.
![Indolo[2,1-a]isoquinolines. Syntheses, steroid hormone receptor binding affinities, and cytostatic activity](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Ambros R., Schneider M.R., Von Angerer S.
Journal of Medicinal Chemistry,
1990
20.
![A one-pot conversion of ortho-alkynyl benzaldehydes into indolo[2,1-a]isoquinolines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kraus G.A., Beasley J.
Tetrahedron Letters,
2013
21.
![Indolopyridines with a hetero atom at a position of fusion. 5. Catalytic synthesis of indolo[2,1-a]isoquinoline by pyrolysis of benzylisoquinolines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Soldatova S.A., Rodriguez Alarcon J.A., Soldatenkov A.T.
Chemistry of Heterocyclic Compounds,
1994
22.

Boente J.M., Castedo L., de Lera A.R., Saá J.M., Suau R., Vidal M.C.
Tetrahedron Letters,
1983
23.
Gvozdev V.D., Shavrin K.N., Baskir E.G., Egorov M.P., Nefedov O.M.
Mendeleev Communications,
2017
24.

Waldmann H., Eberhardt L., Wittstein K., Kumar K.
Chemical Communications,
2010
25.
![Synthesis of indolo[2,1-a]isoquinolines via a triazene-directed C-H annulation cascade.](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Sun H., Wang C., Yang Y., Chen P., Wu Y., Zhang X., Huang Y.
Journal of Organic Chemistry,
2014
26.

Wang Q., Huang L., Wu X., Jiang H.
Organic Letters,
2013
27.

Sanz R.
Organic Preparations and Procedures International,
2008
28.
10.1016/j.mencom.2019.07.002_sbref0130b
Sanz
An Overview in Arene Chemistry: Reaction Mechanisms and Methods for Aromatic Compounds,
2015
29.

Peña D., Escudero S., Pérez D., Guitián E., Castedo L.
Angewandte Chemie - International Edition,
1998
30.
![Palladium-Promoted [2 + 2 + 2] Cocyclization of Arynes and Unsymmetrical Conjugated Dienes: Synthesis of Justicidin B and Retrojusticidin B](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Patel R.M., Argade N.P.
Organic Letters,
2012
31.

Gilmore C.D., Allan K.M., Stoltz B.M.
Journal of the American Chemical Society,
2008
32.

Gonsalves A.M., Pinho e Melo T.M., Gilchrist T.L.
Tetrahedron,
1992
33.
![[4 + 2]‐Cycloadditionen mit inversem Elektronenbedarf, XVII. Oxepin und 2,7‐Dimethyloxepin als Dienophile bei Diels‐Alder‐Cycloadditionen mit inversem Elektronenbedarf](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Dhar R., Hühnermann W., Kämpchen T., Overheu W., Seitz G.
Chemische Berichte,
1983
34.

Kopchuk D.S., Nikonov I.L., Zyryanov G.V., Kovalev I.S., Rusinov V.L., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2014
35.

Kopchuk D.S., Chepchugov N.V., Taniya O.S., Khasanov A.F., Giri K., Kovalev I.S., Santra S., Zyryanov G.V., Majee A., Rusinov V.L., Chupakhin O.N.
Tetrahedron Letters,
2016
36.

Li J., Gao J., Xiong W., Zhang Q.
Tetrahedron Letters,
2014
37.
![A one-pot approach to 10-(1 H -1,2,3-triazol-1-yl)pyrimido[1,2- a ]indoles via aryne-mediated transformations of 3-(pyrimidin-2-yl)-1,2,4-triazines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kopchuk D.S., Chepchugov N.V., Khasanov A.F., Kovalev I.S., Santra S., Nosova E.V., Zyryanov G.V., Majee A., Rusinov V.L., Chupakhin O.N.
Tetrahedron Letters,
2016
38.
![Benzyne-mediated rearrangement of 3-(2-pyridyl)-1,2,4-triazines into 10-(1H-1,2,3-triazol-1-yl)pyrido[1,2-a]indoles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Nikonov I.L., Kopchuk D.S., Kovalev I.S., Zyryanov G.V., Khasanov A.F., Slepukhin P.A., Rusinov V.L., Chupakhin O.N.
Tetrahedron Letters,
2013
39.

Kopchuk D.S., Nikonov I.L., Khasanov A.F., Giri K., Santra S., Kovalev I.S., Nosova E.V., Gundala S., Venkatapuram P., Zyryanov G.V., Majee A., Chupakhin O.N.
Organic and Biomolecular Chemistry,
2018
40.
Kopchuk D.S., Nikonov I.L., Zyryanov G.V., Nosova E.V., Kovalev I.S., Slepukhin P.A., Rusinov V.L., Chupakhin O.N.
Mendeleev Communications,
2015
41.

Bachollet S.P., Volz D., Fiser B., Münch S., Rönicke F., Carrillo J., Adams H., Schepers U., Gómez-Bengoa E., Bräse S., Harrity J.P.
Chemistry - A European Journal,
2016
42.
![Identification of trans-4-[1-[[7-fluoro-2-(1-methyl-3-indolyl)-6-benzoxazolyl]acetyl]-(4S)-fluoro-(2S)-pyrrolidinylmethoxy]cyclohexanecarboxylic acid as a potent, orally active VLA-4 antagonist](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Setoguchi M., Iimura S., Sugimoto Y., Yoneda Y., Chiba J., Watanabe T., Muro F., Iigo Y., Takayama G., Yokoyama M., Taira T., Aonuma M., Takashi T., Nakayama A., Machinaga N., et. al.
Bioorganic and Medicinal Chemistry,
2012
43.
![(Benzo[h])Quinolinyl-Substituted Monoazatriphenylenes: Synthesis and Photophysical Properties](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Kopchuk D.S., Khasanov A.F., Kovalev I.S., Kim G.A., Nikonov I.L., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2014
44.

Kozhevnikov D.N., Shabunina O.V., Kopchuk D.S., Slepukhin P.A., Kozhevnikov V.N.
Tetrahedron Letters,
2006
45.

Shabunina O.V., Starnovskaya E.S., Shtaits Y.K., Kopchuk D.S., Kovalev I.S., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Russian Journal of Organic Chemistry,
2018
46.

Kopchuk D.S., Krinochkin A.P., Kozhevnikov D.N., Slepukhin P.A.
Polyhedron,
2016
47.

Kozhevnikov V.N., Shabunina O.V., Kopchuk D.S., Ustinova M.M., König B., Kozhevnikov D.N.
Tetrahedron,
2008
48.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
49.
10.1016/j.mencom.2019.07.002_bib0210
Sheldrick
2008