Abstract
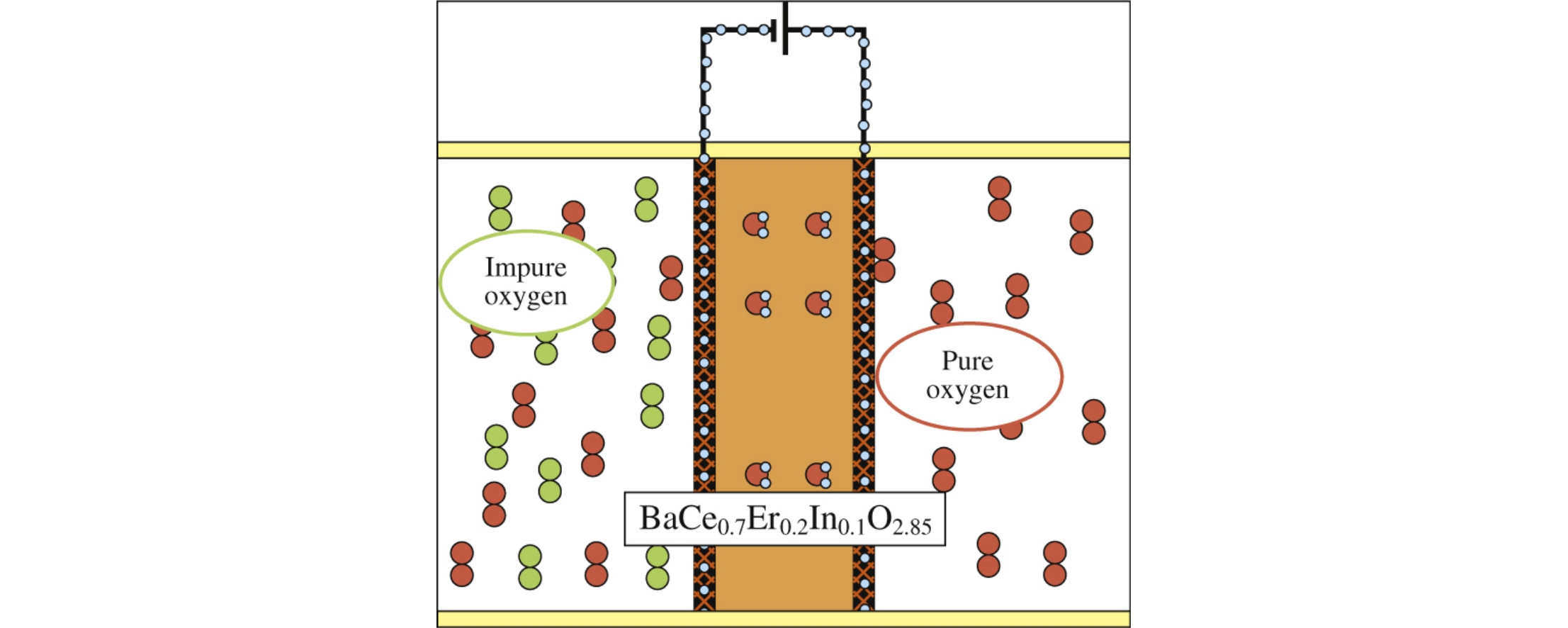
The new compound BaCe0.7Er0.2In0.1O2.85 was synthesized as a representative of promising class of solid state ionic conductors. Thermodynamic functions needed to improve and extend devices portfolio with a special emphasis on the heat capacity and phase transition have been revealed. It has been experimentally established that there is no phase transition for BaCe0.7Er0.2In0.1O2.85 in the temperature range of 191–695K, which can give advantage to the practical application of this compound in comparison with materials possessing the phase transitions.
References
1.
Orlov A.V., Vinokurov A.L., Vanetsev A.S., Tretyakov Y.D., Koltsov A.V., Gavrilov K.L., Levi-Setti R.
Mendeleev Communications,
2004
2.

Guan Q., Wang H., Miao H., Sheng L., Li H.
Ceramics International,
2017
3.
Stenina I.A., Kislitsyn M.N., Pinus I.Y., Yaroslavtsev A.B.
Mendeleev Communications,
2004
4.

Sun L.
International Journal of Electrochemical Science,
2018
5.
Trofimov T.I., Samsonov M.D., Lee S.C., Myasoedov B.F., Wai C.M.
Mendeleev Communications,
2001
6.

Zhang W., Yuan M., Wang H., Liu J.
Journal of Alloys and Compounds,
2016
7.
Bondarenko G.N., Beletskaya I.P.
Mendeleev Communications,
2015
8.
Vasiliev R.B., Rumyantseva M.N., Dorofeev S.G., Potashnikova Y.M., Ryabova L.I., Gaskov A.M.
Mendeleev Communications,
2004
9.

Matskevich N.I., Matskevich M.Y., Wolf T., Bryzgalova A.N., Chupakhina T.I., Anyfrieva O.I.
Journal of Alloys and Compounds,
2013
10.
Greish A.A., Glukhov L.M., Finashina E.D., Kustov L.M., Sung J., Choo K., Kim T.
Mendeleev Communications,
2010
11.

Venkata Krishnan R., Nagarajan K., Vasudeva Rao P.R.
Journal of Nuclear Materials,
2001
12.

Saha R., Babu R., Nagarajan K., Mathews C.K.
Journal of Nuclear Materials,
1989
13.

Matskevich N.I., Wolf T., Adelmann P., Semerikova A.N., Anyfrieva O.I.
Thermochimica Acta,
2015
14.

Matskevich N.I.
Journal of Thermal Analysis and Calorimetry,
2007
15.

Giannici F., Longo A., Balerna A., Kreuer K., Martorana A.
Chemistry of Materials,
2007
16.

Zhao F., Liu Q., Wang S., Brinkman K., Chen F.
International Journal of Hydrogen Energy,
2010
17.
Matskevich N.I., Wolf T., Merz M., Adelmann P., Anyfrieva O.I., Matskevich M.Y.
Mendeleev Communications,
2018
18.

Matskevich N.I., Wolf T., Le Tacon M., Adelmann P., Stankus S.V., Samoshkin D.A., Tkachev E.N.
Journal of Thermal Analysis and Calorimetry,
2017
19.

Matskevich N.I., Wolf T., Pischur D.P., Kozlova S.G., Gelfond N.V., Vyazovkin I.V., Chernov A.A.
Journal of Thermal Analysis and Calorimetry,
2018
20.
10.1016/j.mencom.2019.05.038_bib0100
Gurvich
Thermodynamic Properties of Individual Substances,
1989
21.

Matskevich N.I., Wolf T., Matskevich M.Y.
Journal of Chemical Thermodynamics,
2018