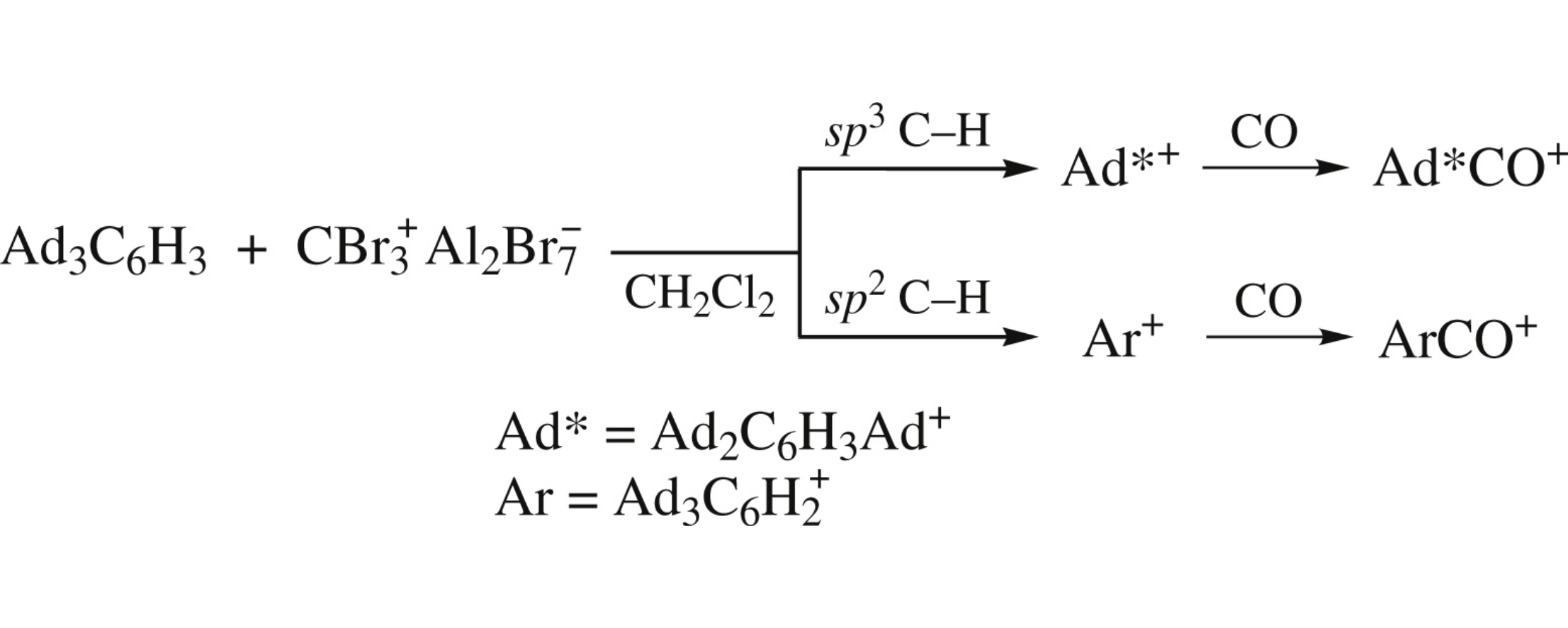
Abstract
The DFT B3LYP-/6-311+G* calculations of the reactions beween 1,3,5-Ad3C6H3 (Ad=1-adamantyl) and CBr3+Al2Br7− with or without CO reveal that only sp3 C–H route occurs.
References
1.

Perego C., Ingallina P.
Catalysis Today,
2002
2.
10.1016/j.mencom.2019.05.035_sbref0005b
Pazur
Ionic Polymerization and Related Processes,
1999
3.
10.1016/j.mencom.2019.05.035_bib0010
Friedel–Crafts and Related Reactions,
1963
4.
10.1016/j.mencom.2019.05.035_bib0015
Taylor
Electrophilic Aromatic Substitution,
1990
5.

Rueping M., Nachtsheim B.J.
Beilstein Journal of Organic Chemistry,
2010
6.
10.1016/j.mencom.2019.05.035_bib0025
Arene Chemistry: Reaction Mechanisms and Methods for Aromatic Compounds,
2015
7.

Naredla R.R., Klumpp D.A.
Chemical Reviews,
2013
8.
10.1016/j.mencom.2019.05.035_bib0035
Olah
Superacid Chemistry,
2009
9.

Milanesi S., Fagnoni M., Albini A.
Chemical Communications,
2002
10.

Nicolaides A., Smith D.M., Jensen F., Radom L.
Journal of the American Chemical Society,
1997
11.

Bernardi F., Grandinetti F., Guarino A., Robb M.A.
Chemical Physics Letters,
1988
12.

Klippenstein S.J.
International Journal of Mass Spectrometry and Ion Processes,
1997
13.

Wright T.G., Panov S.I., Miller T.A.
Journal of Chemical Physics,
1995
14.

Schleyer P.V., Kos A.J., Raghavaohari K.
Journal of the Chemical Society Chemical Communications,
1983
15.

Protti S., Dichiarante V., Dondi D., Fagnoni M., Albini A.
Chemical Science,
2012
16.

Borisov Y.A., Akhrem I.S.
Journal of Molecular Catalysis A Chemical,
2017
17.
10.1016/j.mencom.2019.05.035_bib0050
Borisov
J. Mol. Catal. A: Chem.,
2018
18.

Schleyer P.V., Watts W.E., Fort R.C., Comisarow M.B., Olah G.A.
Journal of the American Chemical Society,
1964
19.

Harding M.E., Gauss J., Schleyer P.V.
Journal of Physical Chemistry A,
2011
20.

Laube T.
Accounts of Chemical Research,
1995
21.

Orlinkov A.V., Akhrem I.S., Vol'pin M.E.
Russian Chemical Reviews,
1991
22.
10.1016/j.mencom.2019.05.035_bib0070
Le Carpentier
1972
23.
10.1016/j.mencom.2019.05.035_bib0075
Le Carpentier
Compt. Rend. Chim.,
1967
24.

Boer F.P.
Journal of the American Chemical Society,
1968
25.

Boer F.P.
Journal of the American Chemical Society,
1966
26.
10.1016/j.mencom.2019.05.035_bib0090
Chevrier
1972
27.

Chevrier B., Le Carpentier J.M., Weiss R.
Journal of the American Chemical Society,
1972
28.
10.1016/j.mencom.2019.05.035_bib0100
Glasstone
The Theory of Rate Processes. The Kinetics of Chemical Reactions, Viscosity, Diffusion and Electro-chemical Phenomena,
1941