Abstract
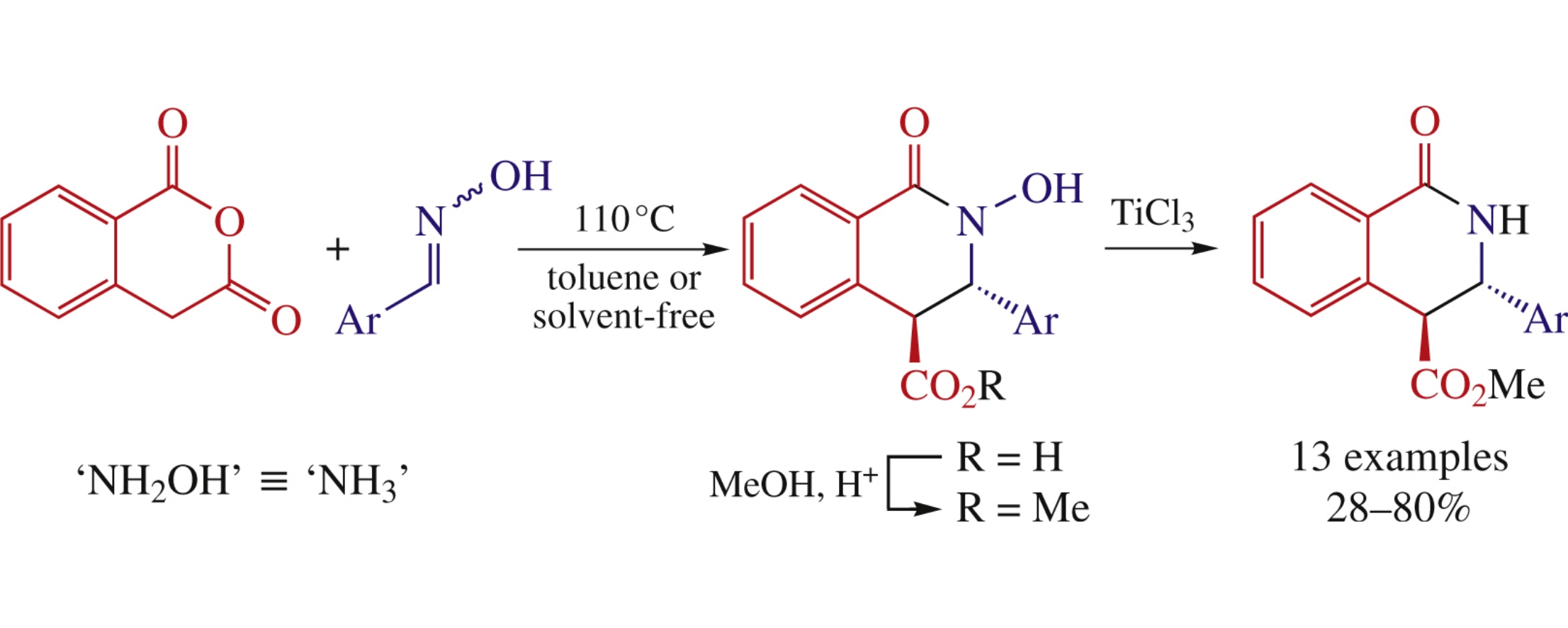
A novel synthetic protocol towards trans-NH-tetrahydroisoquinolonic acid esters is based on the Castagnoli–Cushman reaction between aromatic aldehyde oximes and homophthalic anhydride, followed by esterification and TiCl3-promoted reduction. The scope of the method with respect to the aromatic portion (both electron-rich and electron-deficient) is broader compared to the earlier described approaches, which makes it a suitable synthetic strategy for the structure–activity exploration.
References
1.

Dar’in D., Bakulina O., Chizhova M., Krasavin M.
Organic Letters,
2015
2.

Krasavin M., Dar’in D.
Tetrahedron Letters,
2016
3.

Pommier Y., Cushman M.
Molecular Cancer Therapeutics,
2009
4.

Guranova N., Dar’in D., Krasavin M.
Synthesis,
2018
5.
C. Adams, G.M. Ksander, J. Papillon, Patent WO 2007117982 A3, 2007.
6.

Cushman M., Chen J.K.
Journal of Organic Chemistry,
1987
7.
![Catalytic Enantioselective Synthesis of Lactams through Formal [4+2] Cycloaddition of Imines with Homophthalic Anhydride](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Jarvis C.L., Hirschi J.S., Vetticatt M.J., Seidel D.
Angewandte Chemie - International Edition,
2017
8.

Ghorbani-Choghamarani A., Hajjami M., Norouzi M., Abbasityula Y., Eigner V., Dušek M.
Tetrahedron,
2013
9.

Bakulina O., Bannykh A., Dar'in D., Krasavin M.
Chemistry - A European Journal,
2017
10.

Robinson C.H., Gnoj O., Mitchell A., Wayne R., Townley E., Kabasakalian P., Oliveto E.P., Barton D.H.
Journal of the American Chemical Society,
1961
11.

Mccord T.J., Crawford C.P., Rabon J.A., Gage L.D., Winter J.M., Davis A.L.
Journal of Heterocyclic Chemistry,
1982
12.

Zhai L., Tian X., Wang C., Cui Q., Li W., Huang S., Yu Z., Hong R.
Angewandte Chemie - International Edition,
2017
13.

Yamada K., Kishikawa K., Yamamoto M.
Journal of Organic Chemistry,
1987
14.

Ouyang J., Yan R., Mi X., Hong R.
Angewandte Chemie - International Edition,
2015
15.

Kimmel K.L., Weaver J.D., Lee M., Ellman J.A.
Journal of the American Chemical Society,
2012
16.

Mattingly P.G., Miller M.J.
Journal of Organic Chemistry,
1980
17.
10.1016/j.mencom.2019.05.033_bib0085
Gundersen
Encyclopedia of Reagents for Organic Synthesis,
2007