Abstract
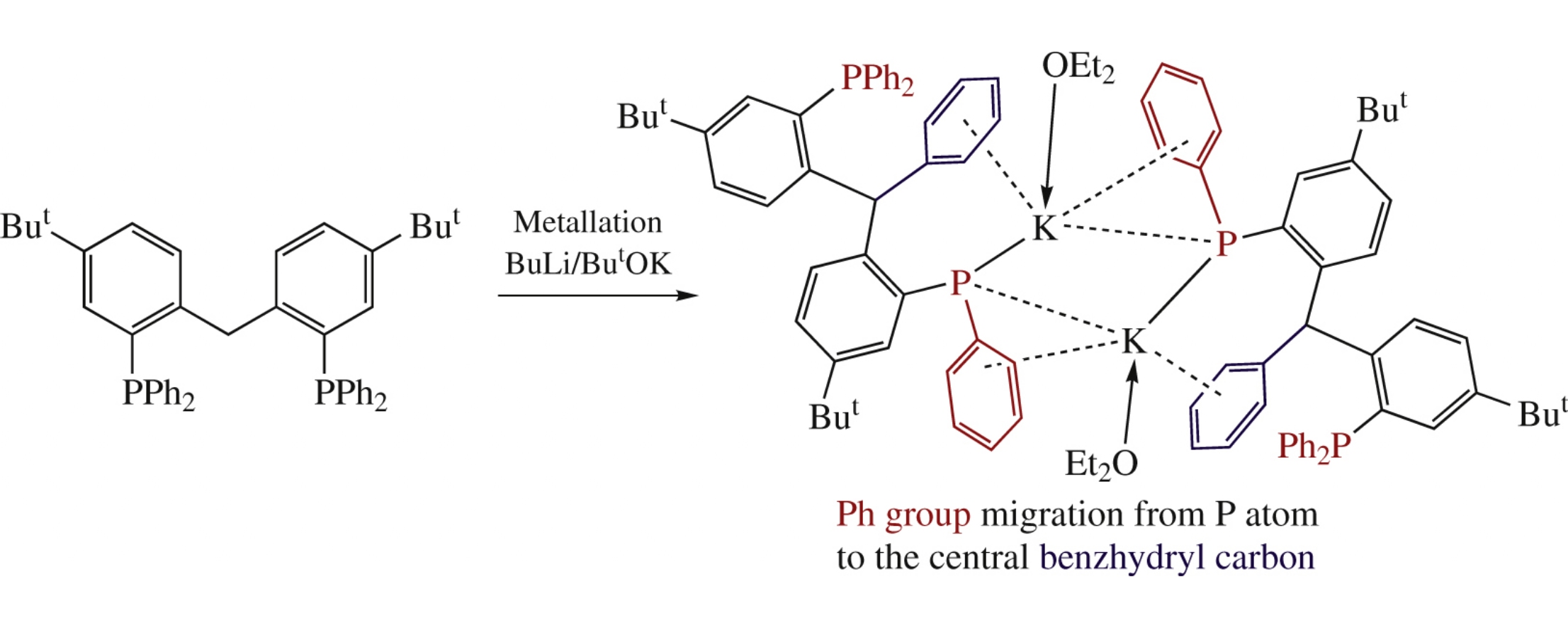
Oxidation of 1,1′-methylenebis[4-tert-butyl-2-(diphenylphosphino) benzene] by H2O2 or S8 in toluene affords two new PCP-type pincer ligands. The methylene group deprotonation in the new ligands as well as in their precursor by n-BuLi or LiCH2SiMe3 failed, while the methylene group in the precursor 1,1′-methylenebis[4-tert-butyl-2-(diphenylphosphino)benzene] was readily metallated by Lochmann–Schlosser superbase to form the unstable potassium complex which in turn was readily transformed into new {5-But-2-[4-But-2-(Ph2P)C6H3(Ph)CH]C6H3P(Ph)K(OEt2)}2 complex due to phenyl group migration.
References
1.
10.1016/j.mencom.2019.05.031_bib0005
The Chemistry of Pincer Compounds,
2007
2.
10.1016/j.mencom.2019.05.031_bib0010
Organometallic Pincer Chemistry,
2013
3.

O'Reilly M.E., Veige A.S.
Chemical Society Reviews,
2014
4.

Goldman A.S., Roy A.H., Huang Z., Ahuja R., Schinski W., Brookhart M.
Science,
2006
5.

Alkane Metathesis by Tandem Alkane-Dehydrogenation–Olefin-Metathesis Catalysis and Related Chemistry
Haibach M.C., Kundu S., Brookhart M., Goldman A.S.
Accounts of Chemical Research,
2012
6.

Zell T., Milstein D.
Accounts of Chemical Research,
2015
7.

Choi J., MacArthur A.H., Brookhart M., Goldman A.S.
Chemical Reviews,
2011
8.

Selander N., Szabó K.J.
Chemical Reviews,
2010
9.
10.1016/j.mencom.2019.05.031_bib0045
Gunanathan
Bifunctional Molecular Catalysis,
2011
10.

Safronov S.V., Koridze A.A.
Russian Chemical Bulletin,
2018
11.

Chakraborty S., Bhattacharya P., Dai H., Guan H.
Accounts of Chemical Research,
2015
12.

Younus H.A., Ahmad N., Su W., Verpoort F.
Coordination Chemistry Reviews,
2014
13.

Deng Q., Melen R.L., Gade L.H.
Accounts of Chemical Research,
2014
14.

Balakrishna M.S.
Polyhedron,
2018
15.
10.1016/j.mencom.2019.05.031_bib0075
Chase
The Privileged Pincer– Metal Platform: Coordination Chemistry & Applications,
2016
16.
10.1016/j.mencom.2019.05.031_bib0080
Pincer and Pincer-Type Complexes: Applications in Organic Synthesis and Catalysis,
2014
17.

Gafurov Z.N., Kagilev A.A., Kantyukov A.O., Balabaev A.A., Sinyashin O.G., Yakhvarov D.G.
Russian Chemical Bulletin,
2018
18.

Abakumov G.A., Piskunov A.V., Cherkasov V.K., Fedushkin I.L., Ananikov V.P., Eremin D.B., Gordeev E.G., Beletskaya I.P., Averin A.D., Bochkarev M.N., Trifonov A.A., Dzhemilev U.M., D'yakonov V.A., Egorov M.P., Vereshchagin A.N., et. al.
Russian Chemical Reviews,
2018
19.

Lesueur W., Solari E., Floriani C., Chiesi-Villa A., Rizzoli C.
Inorganic Chemistry,
1997
20.

Comanescu C.C., Vyushkova M., Iluc V.M.
Chemical Science,
2015
21.

Cui P., Comanescu C.C., Iluc V.M.
Chemical Communications,
2015
22.

Cui P., Iluc V.M.
Chemical Science,
2015
23.

Cui P., Hoffbauer M.R., Vyushkova M., Iluc V.M.
Chemical Science,
2016
24.

Gutsulyak D.V., Piers W.E., Borau-Garcia J., Parvez M.
Journal of the American Chemical Society,
2013
25.
![Electrophilic addition of an osminim-bound chloromethylene ligand to the benzene ring of a triphenylphosphine ligand. Synthesis of metallacyclic and metallabicyclic complexes. X-Ray structures of [OsCl(CO)2(o-PPh2C6H4CHCl)(PPh3)] and [OsCl(CO)2(o-PPh2C6H4CHC6H4PPh2-o)]](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Hoskins S.V., Rckard C.E., Roper W.R.
Journal of the Chemical Society Chemical Communications,
1984
26.

LaPierre E.A., Piers W.E., Spasyuk D.M., Bi D.W.
Chemical Communications,
2016
27.
![Synthesis, structure, and reactions of a bimetallacyclic carbene complex of ruthenium, Br2(CO)[C(C6H4PPh2)2]](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Boyd L.M., Clark G.R., Roper W.R.
Journal of Organometallic Chemistry,
1990
28.

Burford R.J., Piers W.E., Parvez M.
Organometallics,
2012
29.

Reversible Interconversion Between a Monomeric Iridium Hydroxo and a Dinuclear Iridium μ-Oxo Complex
Burford R.J., Piers W.E., Ess D.H., Parvez M.
Journal of the American Chemical Society,
2014
30.

Arras J., Speth H., Mayer H.A., Wesemann L.
Organometallics,
2015
31.

Brock C.P., Schweizer W.B.
Journal of the American Chemical Society,
1985
32.

Bennett M.A., Bhargava S.K., Keniry M.A., Privér S.H., Simmonds P.M., Wagler J., Willis A.C.
Organometallics,
2008
33.

Huang J., Shi M.
Journal of Organic Chemistry,
2003
34.

Albright T.A., Freeman W.J., Schweizer E.E.
Journal of Organic Chemistry,
1975
35.
10.1016/j.mencom.2019.05.031_bib0175
Hays
1964
36.

Bunce R.A., Sullivan J.P.
Synthetic Communications,
1990
37.

Assadourian L., Faure R., Gau G.
Journal of Organometallic Chemistry,
1985
38.

Hill M.S., Hitchcock P.B.
Organometallics,
2001
39.
![Alkali-Metal Alkyl Complexes with the Tridentate Benzhydryl Ligand [2,2′-(4-MeC6H4NMe2)2CH]−](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Khristolyubov D.O., Lyubov D.M., Cherkasov A.V., Fukin G.K., Shavyrin A.S., Trifonov A.A.
Organometallics,
2018
40.

Zabula A.V., Qiao Y., Kosanovich A.J., Cheisson T., Manor B.C., Carroll P.J., Ozerov O.V., Schelter E.J.
Chemistry - A European Journal,
2017
41.

Avent A.G., Bonafoux D., Eaborn C., Hill M.S., Hitchcock P.B., Smith J.D.
Journal of the Chemical Society Dalton Transactions,
2000
42.

Izod K., McFarlane W., Tyson B.V., Clegg W., Harrington R.W., Liddle S.T.
Organometallics,
2003
43.
![Phosphanylborohydrides: First Assessment of the Relative Lewis Basicities of [BH 3 PPh 2 ] – , CH 3 PPh 2 , and HPPh 2](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Dornhaus F., Bolte M., Lerner H., Wagner M.
European Journal of Inorganic Chemistry,
2006