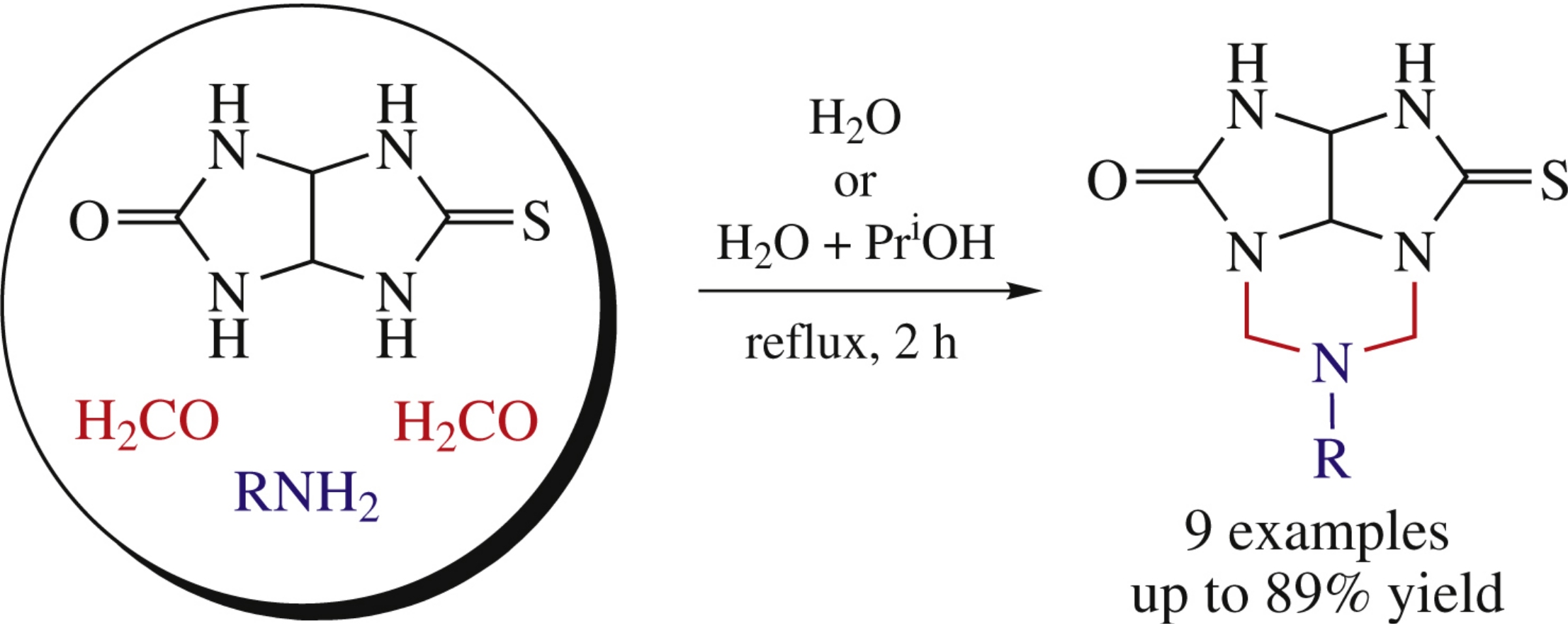
Abstract
A highly selective one-pot synthesis of new 6-substituted 4-thioxohexahydro-5H-2,3,4a,6,7a-pentaazacyclopenta[cd]inden-1(2H)-ones by cycloaminomethylation of semithioglycolurile with formaldehyde and amines is proposed. A mechanism for the process is suggested.
References
1.

Chen Y., She N., Meng X., Yin G., Wu A., Isaacs L.
Organic Letters,
2007
2.

Chen Y., She N., Wu A.
Journal of Chemical Crystallography,
2011
3.
10.1016/j.mencom.2019.05.028_bib0015
Shiri
Synthesis,
2009
4.

Moretti F., Poisson G., Marsura A.
Heteroatom Chemistry,
2016
5.

Stancl M., Khan M.S., Sindelar V.
Tetrahedron,
2011
6.

Hofmann J., Just G., Moya D., Ostermann S., Pritzkow W., Visothea M.P.
Journal für praktische Chemie,
1990
7.
Zharkov M.N., Kuchurov I.V., Fomenkov I.V., Zlotin S.G., Tartakovsky V.A.
Mendeleev Communications,
2015
8.

Cui K., Xu G., Xu Z., Wang P., Xue M., Meng Z., Li J., Wang B., Ge Z., Qin G.
Propellants, Explosives, Pyrotechnics,
2014
9.

Kravchenko A.N., Chikunov I.E., Lyssenko K.A., Baranov V.V.
Chemistry of Heterocyclic Compounds,
2014
10.

Barsegyan Y.A., Baranov V.V., Kravchenko A.N.
Chemistry of Heterocyclic Compounds,
2017
11.
![A Method for Synthesizing Partially Substituted Cucurbit[n]uril](/storage/images/resized/MjH1ITP7lMYGxeqUZfkt2BnVLgjkk413jwBV97XX_small_thumb.webp)
Day A., Arnold A., Blanch R.
Molecules,
2003
12.

Barrow S.J., Kasera S., Rowland M.J., del Barrio J., Scherman O.A.
Chemical Reviews,
2015
13.
Gazieva G.A., Kravchenko A.N., Chegaev K.Y., Strelenko Y.A., Lebedev O.V.
Mendeleev Communications,
2000
14.
10.1016/j.mencom.2019.05.028_bib0070
Wanger
Organic Nanoreactors,
2016
15.
A. Aumueller, P. Neumann, H. Trauth, Patent EP 0272588 A1, 1988.
16.
Anikina L.V., Vikharev Y.B., Baranov V.V., Malyshev O.R., Kravchenko A.N.
Mendeleev Communications,
2018
17.
![Efficient method for the synthesis of 1,3-unsubstituted 2-imino-5-oxooctahydroimidazo[4,5-d]imidazolium iodides based on thioglycolurils](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Baranov V.V., Yatsenko E.L., Melnikova E.K., Nelyubina Y.V., Kravchenko A.N.
Chemistry of Heterocyclic Compounds,
2019
18.

Singh M., Solel E., Keinan E., Reany O.
Chemistry - A European Journal,
2014
19.

Solel E., Singh M., Reany O., Keinan E.
Physical Chemistry Chemical Physics,
2016
20.

Cow C.N., Harrison P.H.
Journal of Organic Chemistry,
1997
21.

Gazieva G.A., Anikina L.V., Nechaeva T.V., Pukhov S.A., Karpova T.B., Popkov S.V., Nelyubina Y.V., Kolotyrkina N.G., Kravchenko A.N.
European Journal of Medicinal Chemistry,
2017
22.

Gazieva G.A., Nechaeva T.V., Kostikova N.N., Sigay N.V., Serkov S.A., Popkov S.V.
Russian Chemical Bulletin,
2018
23.

New access to thioglycolurils by condensation of 4,5-dihydroxyimidazolidin-2-ones(thiones) with HSCN
Baranov V.V., Nelyubina Y.V., Kravchenko A.N., Kolotyrkina N.G., Biriukova K.A.
Tetrahedron Letters,
2015
24.
![Synthesis, Structure, and Biological Activity of 2,6-Disubstituted 2,3a,4a,6,7a,8a-Hexaazaperhydrocyclopenta[ def ]fluorene-4-thioxo-8-ones](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Barsegyan Y., Baranov V., Kravchenko A., Strelenko Y., Anikina L., Karnoukhova V., Kolotyrkina N.
Synthesis,
2018
25.

Mock W.L., Manimaran T., Freeman W.A., Kuksuk R.M., Maggio J.E., Williams D.H.
Journal of Organic Chemistry,
1985
26.

Ali-Asgari S., Moradi M., Maskani E., Mirtamizdoust B.
Chemistry of Natural Compounds,
2017