Abstract
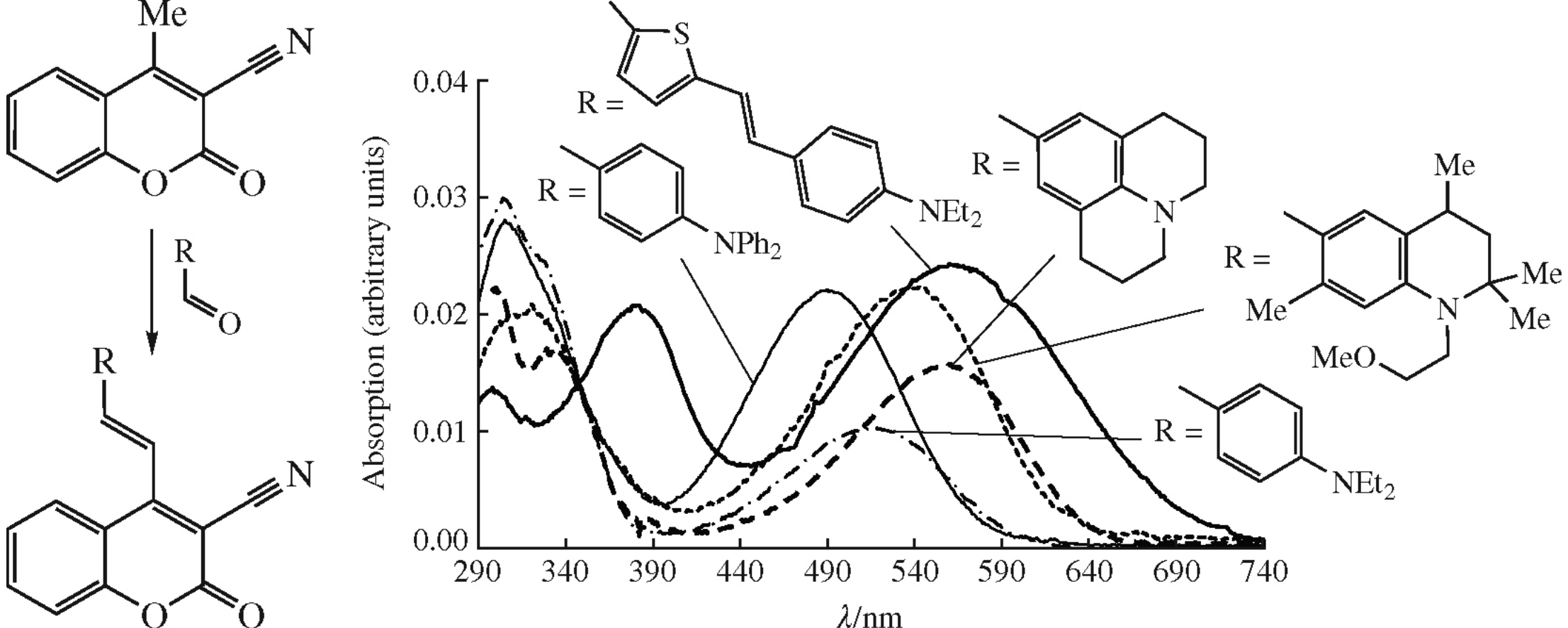
New derivatives of 2-oxo-4-vinyl-2H-chromene-3-carbonitrile have been synthesized by the Knoevenagel reaction of 4-methyl-2-oxo-2H-chromene-3-carbonitrile with aromatic and heteroaromatic aldehydes. The first hyperpolarizability β for the obtained compounds was calculated using M05-2X functional and 6-31+G(d) basis set; their optical properties and solvatochromism in solvents of various polarity were examined. The crystal structure for one representative of the synthesized compounds was determined by XRD.
References
1.

Liu J., Xu G., Liu F., Kityk I., Liu X., Zhen Z.
RSC Advances,
2015
2.

Wu W., Qin J., Li Z.
Polymer,
2013
3.

Shi Z., Luo J., Huang S., Polishak B.M., Zhou X., Liff S., Younkin T.R., Block B.A., Jen A.K.
Journal of Materials Chemistry A,
2012
4.

Dalton L.R., Harper A.W., Ghosn R., Steier W.H., Ziari M., Fetterman H., Shi Y., Mustacich R.V., Jen A.K., Shea K.J.
Chemistry of Materials,
1995
5.

Zhang C., Dalton L.R., Oh M., Zhang H., Steier W.H.
Chemistry of Materials,
2001
6.

Peng C., Wu J., Liu J., Qiu L., Liu X., Bo S., Zhen Z.
Polymer Chemistry,
2013
7.

Suponitsky K.Y., Timofeeva T.V., Antipin M.Y.
Russian Chemical Reviews,
2006
8.

Shi Y., Frattarelli D., Watanabe N., Facchetti A., Cariati E., Righetto S., Tordin E., Zuccaccia C., Macchioni A., Wegener S.L., Stern C.L., Ratner M.A., Marks T.J.
Journal of the American Chemical Society,
2015
9.

Briers D., De Cremer L., Koeckelberghs G., Foerier S., Verbiest T., Samyn C.
Macromolecular Rapid Communications,
2007
10.

Pereverzev Y.V., Gunnerson K.N., Prezhdo O.V., Sullivan P.A., Liao Y., Olbricht B.C., Akelaitis A.J., Jen A.K., Dalton L.R.
Journal of Physical Chemistry C,
2008
11.

Cho M.J., Lim J.H., Hong C.S., Kim J.H., Lee H.S., Choi D.H.
Dyes and Pigments,
2008
12.
A. Kaneko, M. Tsukase, M. Satou, US Patent 9798044B2, 2017.
13.
D. Huang, B. Chen, US Patent 6750603B2, 2004.
14.

Tolmachev A.I., Kachkovskii A.D., Kudinova M.A., Kurdiukov V.V., Ksenzov S., Schrader S.
Dyes and Pigments,
2007
15.
10.1016/j.mencom.2019.05.020_bib0075
Chudov
XII Russian Conference ‘Technologies and Materials for Extreme Conditions (Laser Technologies Electric Current Sources and Materials)’,
2017
16.
10.1016/j.mencom.2019.05.020_bib0080
Levchenko
VII International Conference on Photonics and Information Optics,
2018
17.

Bochkov A.Y., Akchurin I.O., Dyachenko O.A., Traven V.F.
Chemical Communications,
2013
18.
![Novel push-pull fluorescent dyes – 7-(diethylamino)furo- and thieno[3,2-c]coumarins derivatives: structure, electronic spectra and TD-DFT study](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Akchurin I.O., Yakhutina A.I., Bochkov A.Y., Solovjova N.P., Medvedev M.G., Traven V.F.
Journal of Molecular Structure,
2018
19.
10.1016/j.mencom.2019.05.020_bib0095
Sharma
International Journal of Environmental Science: Development and Monitoring (IJESDM),
2013
20.

Reynolds G.A., Drexhage K.H.
Optics Communications,
1975
21.

Hara K., Sato T., Katoh R., Furube A., Ohga Y., Shinpo A., Suga S., Sayama K., Sugihara H., Arakawa H.
Journal of Physical Chemistry B,
2002
22.

Chen Q., Wu N., Liu Y., Li X., Liu B.
RSC Advances,
2016
23.
![Reactivity of condensed thiophenes in the Diels-Alder reaction: The reactivity of 3-aminothieno [3,4:3`,4`] benzo [b] pyranone; 3-aminothieno [3,4-c] quinoline and of 5-amino-7-substituted thieno [3,4-d] pyridazinone toward electron-poor olefins and acetylenes](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Al-Omran F., Mohammed Abdel Khalik M., Al-Awadhi H., Elnagdi M.H.
Tetrahedron,
1996
24.

Elnagdi M.H., Al-Awadi N.A., Kumar A., Khalik M.A.
Heteroatom Chemistry,
2001
25.

Levchenko K.S., Chudov K.A., Zinoviev E.V., Lyssenko K.A., Demin D.U., Poroshin N.O., Shmelin P.S., Grebennikov E.P.
Tetrahedron Letters,
2018
26.

Liu F., Xiao H., Yang Y., Wang H., Zhang H., Liu J., Bo S., Zhen Z., Liu X., Qiu L.
Dyes and Pigments,
2016
27.

Reichardt C.
Chemical Reviews,
1994
28.

Suponitsky K.Y., Tafur S., Masunov A.E.
Journal of Chemical Physics,
2008
29.

Suponitsky K.Y., Liao Y., Masunov A.E.
Journal of Physical Chemistry A,
2009
30.

Suponitsky K.Y., Masunov A.E.
Journal of Chemical Physics,
2013
31.
Suponitsky K.Y., Masunov A.E., Antipin M.Y.
Mendeleev Communications,
2009
32.

Cossi M., Barone V.
Journal of Chemical Physics,
2001
33.

Rao V.P., Jen A.K., Wong K., Drost K.J., Mininni R.M.
Proceedings of SPIE - The International Society for Optical Engineering,
1993
34.

Clays K., Persoons A.
Physical Review Letters,
1991
35.

Clays K., Persoons A.
Review of Scientific Instruments,
1992
36.

Kinnibrugh T., Bhattacharjee S., Sullivan P., Isborn C., Robinson B.H., Eichinger B.E.
Journal of Physical Chemistry B,
2006
37.
Suponitsky K.Y., Masunov A.E., Antipin M.Y.
Mendeleev Communications,
2008