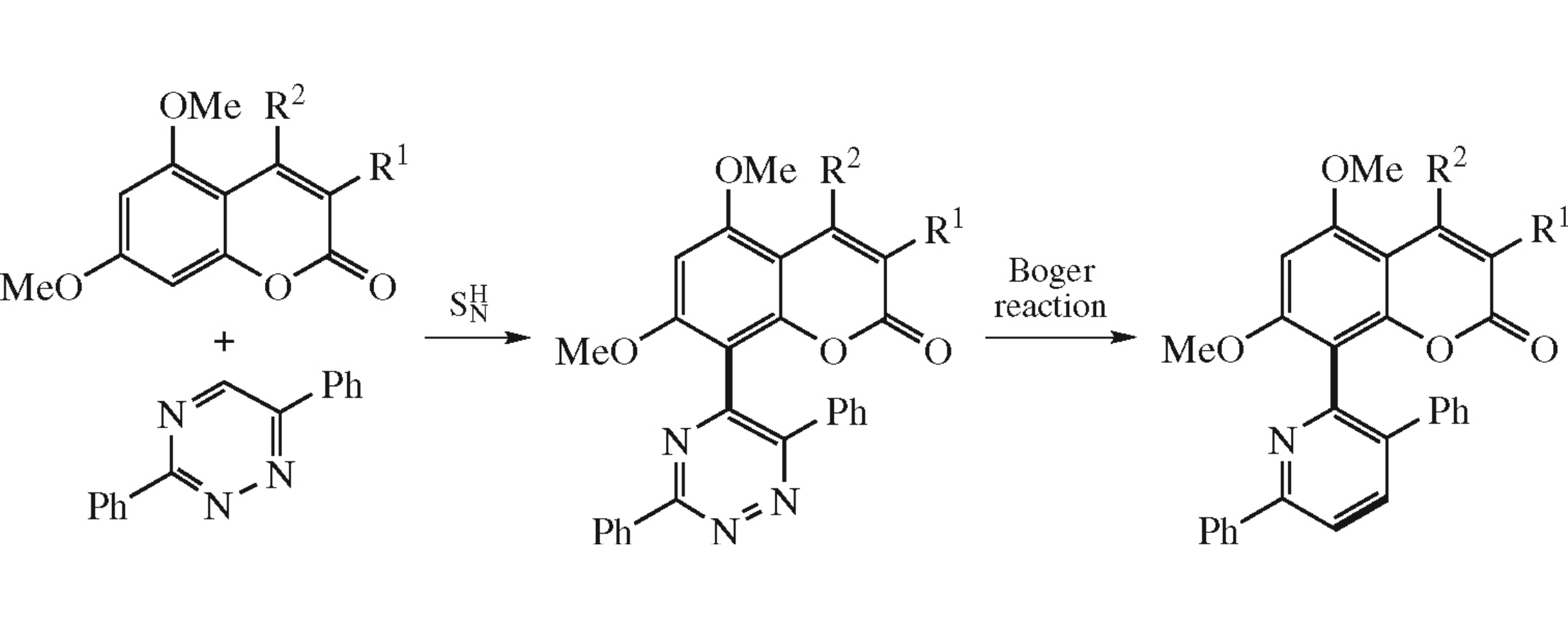
Abstract
5,7-Dimethoxy-8-(3,6-diphenylpyridin-2-yl)coumarins were obtained from 5,7-dimethoxycoumarins and 3,6-diphenyl-1,2,4-triazines via the protocol comprising aromatic \( \mathrm{S_N^H} \) substitution in the triazine ring followed by the Boger transformation of formed triazine moiety into the pyridine one. The advantages of the suggested method are simple procedures, high yields, and the absence of transition-metal catalysts.
References
1.
10.1016/j.mencom.2019.05.019_sbref0005a
Patel
Arkivoc,
2010
2.

Moffett R.B.
Journal of Medicinal Chemistry,
1964
3.

Sreenivasulu B., Sundaramurthy V., Rao N.V.
Proceedings of the Indian Academy of Sciences - Section A,
1974
4.

Lad H., Giri R., Chovatiya Y., Brahmbhatt D.
Journal of the Serbian Chemical Society,
2015
5.
10.1016/j.mencom.2019.05.019_sbref0010b
Brahmbhatt
Indian J. Chem., Sect. B,
2007
6.
10.1016/j.mencom.2019.05.019_sbref0010c
Brahmbhatt
Indian J. Chem., Sect. B,
2001
7.
10.1016/j.mencom.2019.05.019_sbref0015a
Charushin
2014
8.

Chupakhin O.N., Postovskii I.Y.
Russian Chemical Reviews,
1976
9.

Kozhevnikov D.N., Rusinov V.L., Chupakhin O.N.
Advances in Heterocyclic Chemistry,
2002
10.
10.1016/j.mencom.2019.05.019_sbref0025a
Li
Name Reactions,
2014
11.

Pabst G.R., Pfüller O.C., Sauer J.
Tetrahedron,
1999
12.

Rykowski A., Branowska D., Kielak J.
Tetrahedron Letters,
2000
13.
Kopchuk D.S., Kovalev I.S., Khasanov A.F., Zyryanov G.V., Slepukhin P.A., Rusinov V.L., Chupakhin O.N.
Mendeleev Communications,
2013
14.

Prokhorov A.M., Kozhevnikov D.N.
Chemistry of Heterocyclic Compounds,
2012
15.

Kozhevnikov V.N., Kozhevnikov D.N., Nikitina T.V., Rusinov V.L., Chupakhin O.N., Zabel M., König B.
Journal of Organic Chemistry,
2003
16.

Kozhevnikov D.N., Kozhevnikov V.N., Prokhorov A.M., Ustinova M.M., Rusinov V.L., Chupakhin O.N., Aleksandrov G.G., König B.
Tetrahedron Letters,
2006
17.

Prokhorov A.M., Kozhevnikov D.N., Rusinov V.L., Chupakhin O.N., Glukhov I.V., Antipin M.Y., Kazheva O.N., Chekhlov A.N., Dyachenko O.A.
Organometallics,
2006
18.
Kovalev I.S., Kopchuk D.S., Khasanov A.F., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Mendeleev Communications,
2014
19.

Shtaitz Y.K., Savchuk M.I., Starnovskaya E.S., Krinochkin A.P., Kopchuk D.S., Santra S., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
AIP Conference Proceedings,
2019
20.

Kopchuk D.S., Chepchugov N.V., Kovalev I.S., Santra S., Rahman M., Giri K., Zyryanov G.V., Majee A., Charushin V.N., Chupakhin O.N.
RSC Advances,
2017
21.

Kopchuk D.S., Krinochkin A.P., Starnovskaya E.S., Shtaitz Y.K., Khasanov A.F., Taniya O.S., Santra S., Zyryanov G.V., Majee A., Rusinov V.L., Chupakhin O.N.
ChemistrySelect,
2018
22.

Savchuk M.I., Starnovskaya E.S., Shtaitz Y.K., Kopchuk D.S., Nosova E.V., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Russian Journal of General Chemistry,
2018
23.

Kopchuk D.S., Nikonov I.L., Khasanov A.F., Giri K., Santra S., Kovalev I.S., Nosova E.V., Gundala S., Venkatapuram P., Zyryanov G.V., Majee A., Chupakhin O.N.
Organic and Biomolecular Chemistry,
2018
24.

Sharghi H., Jokar M.
Heterocycles,
2007
25.

IINUMA M., TANAKA T., HAMADA K., MIZUNO M., ASAI F.
Chemical and Pharmaceutical Bulletin,
2011
26.

Alkoxypsoralens, Novel Nonpeptide Blockers of Shaker-Type K+ Channels: Synthesis and Photoreactivity
Wulff H., Rauer H., Düring T., Hanselmann C., Ruff K., Wrisch A., Grissmer S., Hänsel W.
Journal of Medicinal Chemistry,
1998
27.

Saraswathi T.V., Srinivasan V.R.
Tetrahedron,
1977
28.

Khalymbadzha I., Chupakhin O., Fatykhov R., Charushin V., Schepochkin A., Kartsev V.
Synlett,
2016
29.

Khalymbadzha I., Fatykhov R., Chupakhin O., Charushin V., Tseitler T., Sharapov A., Inytina A., Kartsev V.
Synthesis,
2018
30.

Kumar N.S., Shafikov M.Z., Whitwood A.C., Donnio B., Karadakov P.B., Kozhevnikov V.N., Bruce D.W.
Chemistry - A European Journal,
2016
31.

Shafikov M.Z., Kozhevnikov D.N., Bodensteiner M., Brandl F., Czerwieniec R.
Inorganic Chemistry,
2016
32.

Krayushkin M.M., Sedishev I.P., Yarovenko V.N., Zavarzin I.V., Kotovskaya S.K., Kozhevnikov D.N., Charushin V.N.
Russian Journal of Organic Chemistry,
2008
33.

Kozhevnikov V.N., Ustinova M.M., Slepukhin P.A., Santoro A., Bruce D.W., Kozhevnikov D.N.
Tetrahedron Letters,
2008