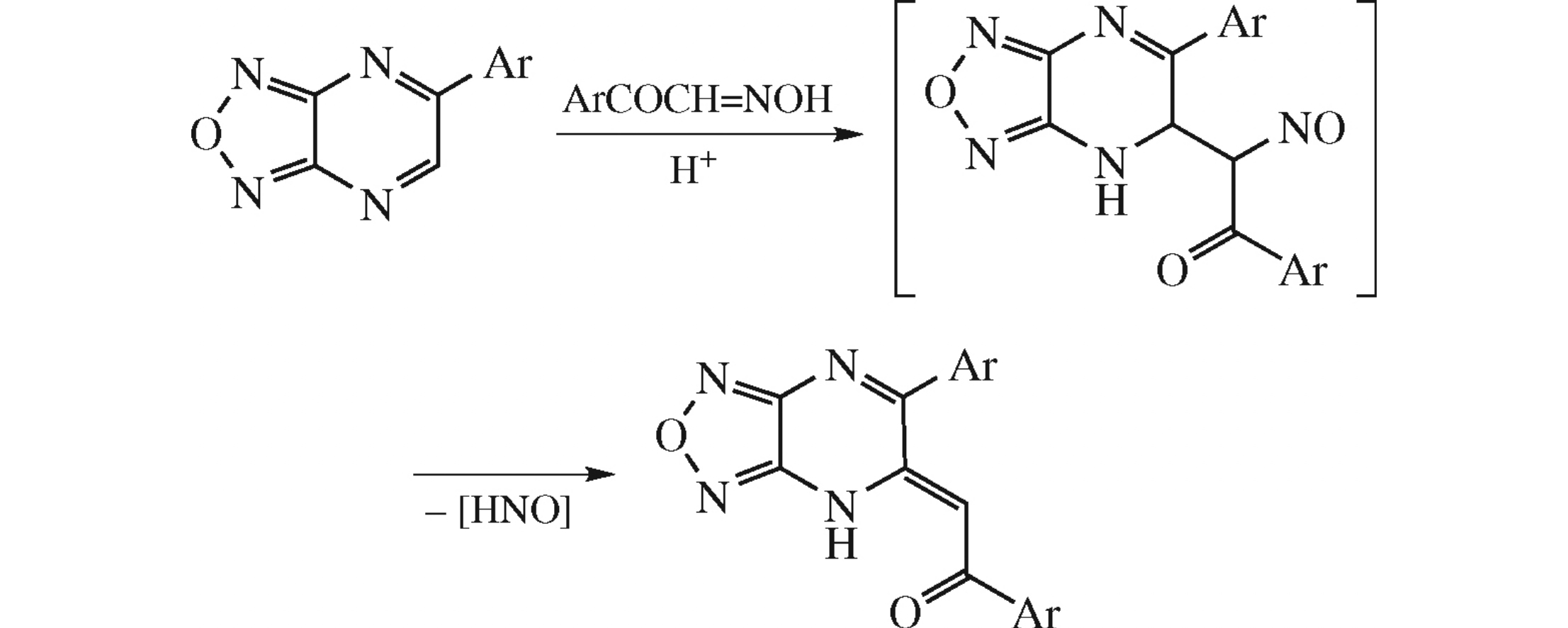
Abstract
Reaction of arylglyoxal oximes ArCOCH=NOH (Ar=4-MeOC6H4, Ph) with 5-arylfurazanopyrazines proceeds as vicarious nucleophilic substitution of hydrogen in pyrazine ring with the elimination of hyponitrous acid, affording 5-(aroylmethylidene)-6-aryl-4H-furazano[3,4-b]pyrazines. Structure of the product was confirmed by X-ray diffraction.
References
1.
![Advances in the chemistry of furazano[3,4-b]pyrazines and their analogues](/storage/images/resized/9Mus3KG1Tkd7Bwaurt8H3RwWh0CxRlGoO6ng9UK1_small_thumb.webp)
Sheremetev A.B., Yudin I.L.
Russian Chemical Reviews,
2003
2.

Beebe X., Nilius A.M., Merta P.J., Soni N.B., Bui M.H., Wagner R., Beutel B.A.
Bioorganic and Medicinal Chemistry Letters,
2003
3.
![Structure-activity relationships of furazano[3,4-b]pyrazines as mitochondrial uncouplers.](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kenwood B.M., Calderone J.A., Taddeo E.P., Hoehn K.L., Santos W.L.
Bioorganic and Medicinal Chemistry Letters,
2015
4.
K. Lai, K.J. Wierenga, M. Tang, US Patent 8357701B2, 2013.
5.
H. Paulus, US Patent 7553832B2, 2009.
6.
D.E. Fisher, R. Haq, H.R.V. Widlund, US Patent 2014/0296223A1, 2014.
7.

De Meester J.W., Van Der Plas H.C., Middelhoven W.J.
Journal of Heterocyclic Chemistry,
1987
8.

Coogan N.T., Chimes M.A., Raftery J., Mocilac P., Denecke M.A.
Journal of Organic Chemistry,
2015
9.

Lazarenko V., Dorovatovskii P., Zubavichus Y., Burlov A., Koshchienko Y., Vlasenko V., Khrustalev V.
Crystals,
2017
10.

Evans P.
Acta Crystallographica Section D Biological Crystallography,
2005
11.

Battye T.G., Kontogiannis L., Johnson O., Powell H.R., Leslie A.G.
Acta Crystallographica Section D Biological Crystallography,
2011
12.

Winn M.D., Ballard C.C., Cowtan K.D., Dodson E.J., Emsley P., Evans P.R., Keegan R.M., Krissinel E.B., Leslie A.G., McCoy A., McNicholas S.J., Murshudov G.N., Pannu N.S., Potterton E.A., Powell H.R., et. al.
Acta Crystallographica Section D Biological Crystallography,
2011
13.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015
14.
![Synthesis of furazano[3,4-b]pyrazine derivatives](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Eremeev A.V., Andrianov V.G., Piskunova I.P.
Chemistry of Heterocyclic Compounds,
1978
15.
10.1016/j.mencom.2019.05.018_bib0055
1979
16.

Möhrle H., Keller G.
Zeitschrift fur Naturforschung - Section B Journal of Chemical Sciences,
1999
17.
10.1016/j.mencom.2019.05.018_sbref0060b
Hahn
Pol. J. Chem.,
1986
18.
19.

Hammouda M., Abou Zeid Z.M., Metwally M.A.
Chemistry of Heterocyclic Compounds,
2008
20.
![Reactions of pyrazinium salts with phenols:from σH-adducts to SN Hproducts and transformations into benzo[b]furans](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Verbitskiy E.V., Kvashnin Y.A., Slepukhin P.A., Kuchin A.V., Rusinov G.L., Chupakhin O.N., Charushina V.N.
Russian Chemical Bulletin,
2011
21.
![9-Ethyl-3-{6-(het)aryl-[1,2,5]oxadiazolo[3,4-b]pyrazin-5-yl}-9H-carbazoles: synthesis and study of sensitivity to nitroaromatic compounds](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Verbitskiy E.V., Kvashnin Y.A., Baranova A.A., Yakovleva Y.A., Khokhlov K.О., Rusinov G.L., Charushin V.N.
Russian Chemical Bulletin,
2018
22.

Goliński J., Makosza M.
Tetrahedron Letters,
1978
23.

Ma̧kosza M., Wojciechowski K.
Chemical Reviews,
2004
24.
10.1016/j.mencom.2019.05.018_sbref0070c
Terrier
Modern Nucleophilic Aromatic Substitution,
2013
25.

Chupakhin O.N., Charushin V.N.
Tetrahedron Letters,
2016
26.
![Direct arylalkenylation of furazano[3,4- b ]pyrazines via a new C–H functionalization protocol](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kazin N.A., Kvashnin Y.A., Irgashev R.A., Dehaen W., Rusinov G.L., Charushin V.N.
Tetrahedron Letters,
2015