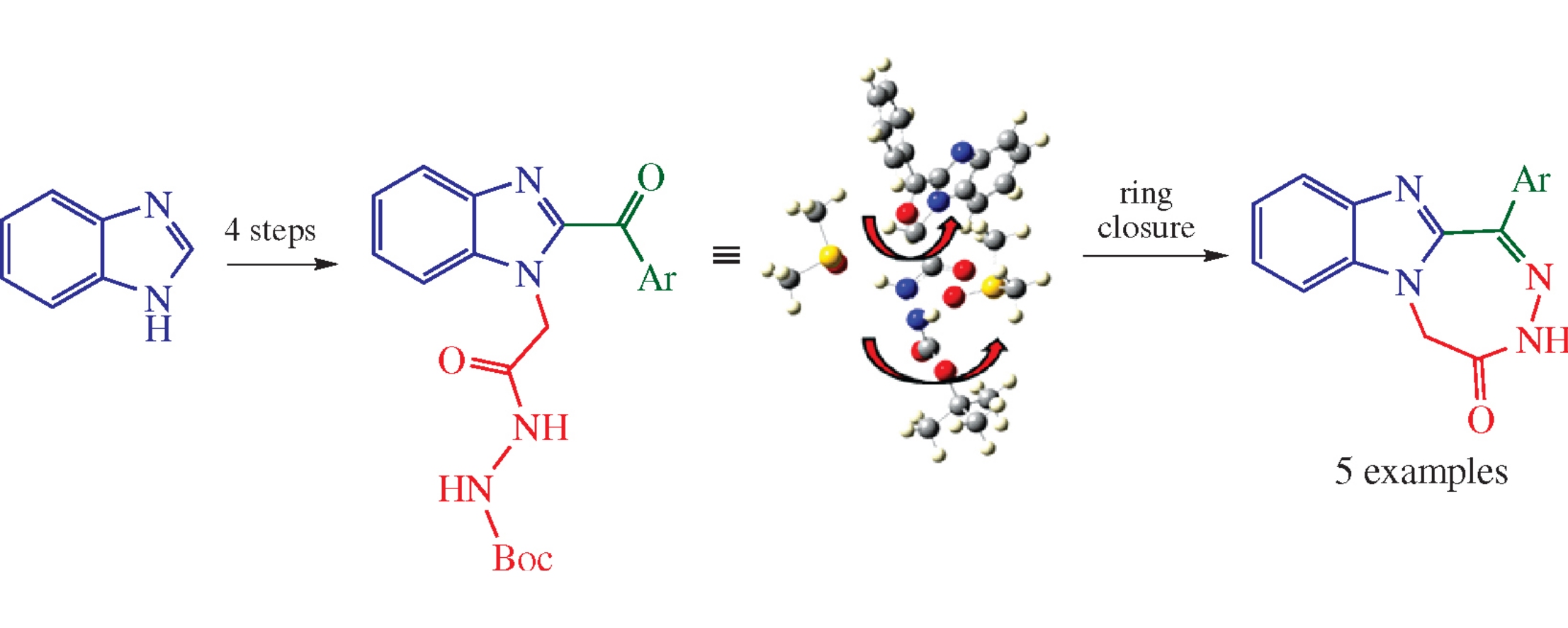
Abstract
3H-[1,2,5]Triazepino[5,4-a]benzimidazol-4(5H)-ones were obtained in five steps involving C-acylation of benzimidazole, its N-alkylation with ethyl bromoacetate, the ester hydrolysis, condensation with BocNHNH2, and the acid-catalyzed heterocyclization of thus obtained 2-(2-aroyl-1H-benzimidazol-1-yl)-N′-(tert-butoxycarbonyl)acetohydrazides. The geometry of tert-butyl carbazate rotamers was estimated with quantum chemical calculations.
References
1.

Noman L., Oke-Altuntas F., Zellagui A., Sahin Yaglioglu A., Demirtas I., M Cardoso S., Akkal S., Gherraf N., Rhouati S.
Natural Product Research,
2017
2.

Townsend L.B., Revankar G.R.
Chemical Reviews,
1970
3.
Koshchienko Y.V., Kuz’menko T.A., Morkovnik A.S., Kiskin M.A., Alexandrov G.G.
Mendeleev Communications,
2016
4.
Gvozdev V.D., Shavrin K.N., Baskir E.G., Egorov M.P., Nefedov O.M.
Mendeleev Communications,
2017
5.
Vchislo N.V., Verochkina E.A., Larina L.I., Vashchenko A.V., Chuvashev Y.A.
Mendeleev Communications,
2017
6.

Bansal Y., Silakari O.
Bioorganic and Medicinal Chemistry,
2012
7.

Keri R.S., Hiremathad A., Budagumpi S., Nagaraja B.M.
Chemical Biology and Drug Design,
2014
8.

Volk B., Milen M., Földesi T., Dancsó A., Simig G.
Synlett,
2015
9.
![Efficient synthesis of a new compound family, 9-aryl-5 H -imidazo[2,1- d ][1,2,5]triazepin-6(7 H )-ones](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Földesi T., Dancsó A., Simig G., Volk B., Milen M.
Tetrahedron,
2016
10.

Földesi T., Dancsó A., Volk B., Milen M.
Tetrahedron,
2017
11.

Yang H., Murigi F.N., Wang Z., Li J., Jin H., Tu Z.
Bioorganic and Medicinal Chemistry Letters,
2015
12.

Milen M., Grün A., Bálint E., Dancsó A., Keglevich G.
Synthetic Communications,
2010
13.

Guerret P., Ancher J., Langlois M.
Journal of Heterocyclic Chemistry,
1983
14.
10.1016/j.mencom.2019.05.017_bib0070
Greene
Protective Groups in Organic Synthesis,
1999