Abstract
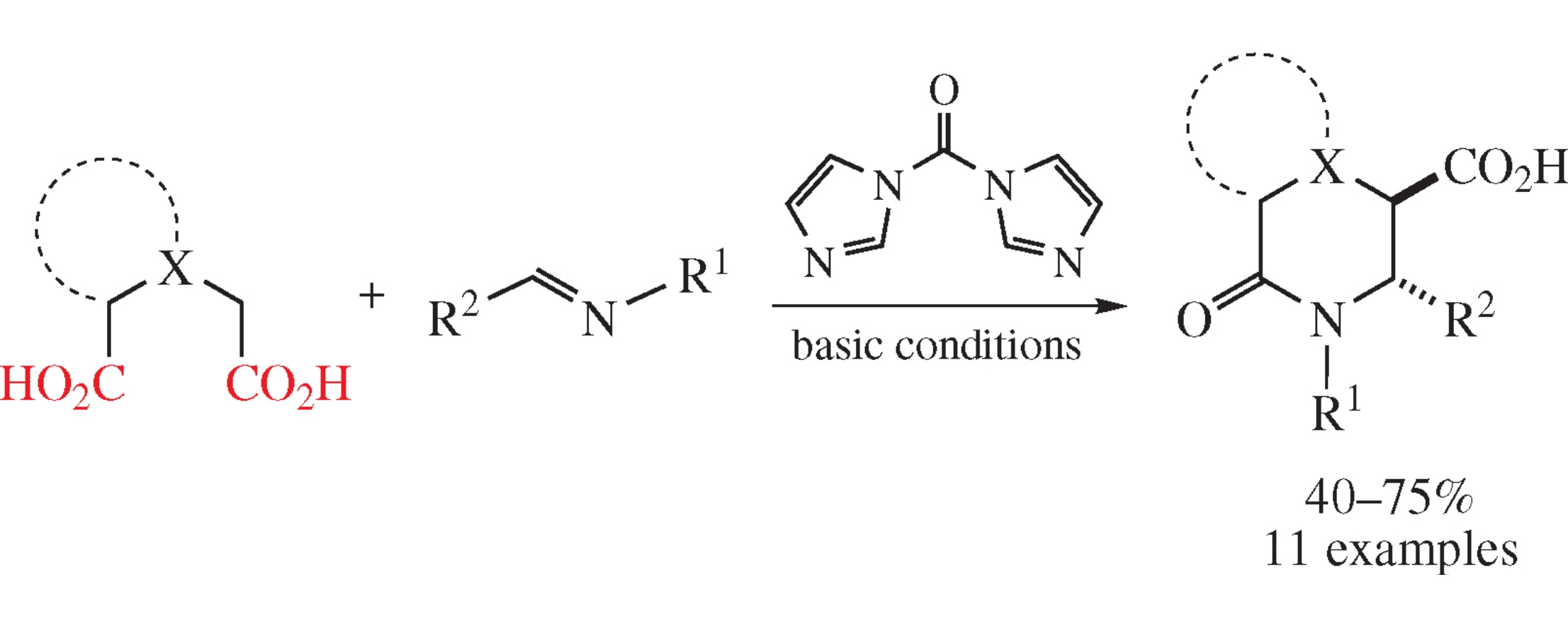
A novel protocol for the Castagnoli–Cushman reaction of dicarboxylic acids and imines comprises the use of 1,1′-carbonyldiimidazole as the cyclodehydrating agent to in situ produce the intermediate anhydrides. In contrast to previously developed procedure involving the use of acetic anhydride, the current protocol allows one to utilize substrates prone to acylation or acid-promoted transformations, which significantly broadens the reaction scope of lactams to be obtained.
References
1.

Krasavin M., Dar’in D.
Tetrahedron Letters,
2016
2.

Dar’in D., Bakulina O., Chizhova M., Krasavin M.
Organic Letters,
2015
3.

Lepikhina A., Dar’in D., Bakulina O., Chupakhin E., Krasavin M.
ACS Combinatorial Science,
2017
4.

Adamovskyi M.I., Ryabukhin S.V., Sibgatulin D.A., Rusanov E., Grygorenko O.O.
Organic Letters,
2016
5.

Krasavin M., Bakulina O., Dar’in D.
Synlett,
2017
6.

Pommier Y.
Chemical Reviews,
2009
7.

González-López M., Shaw J.T.
Chemical Reviews,
2009
8.

Staab H.A., Walther G., Rohr W.
Chemische Berichte,
1962
9.

Melillo D.G., Larsen R.D., Mathre D.J., Shukis W.F., Wood A.W., Colleluori J.R.
Journal of Organic Chemistry,
1987
10.

Kantin G., Chupakhin E., Dar'in D., Krasavin M.
Tetrahedron Letters,
2017
11.

Hauptschein M., Stokes C.S., Nodiff E.A.
Journal of the American Chemical Society,
1952
12.

McMaster L., Ahmann F.F.
Journal of the American Chemical Society,
1928
13.

Fieser L.F., Daudt W.H.
Journal of the American Chemical Society,
1941
14.

Storr T.E., Cully S.J., Rawling M.J., Lewis W., Hamza D., Jones G., Stockman R.A.
Bioorganic and Medicinal Chemistry,
2015
15.
![Enantiospecific synthesis of the trans-9-[3-(3,5-dimethyl-1-piperazinyl)propyl]carbazoles](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Harfenist M., Hoerr D.C., Crouch R.
Journal of Organic Chemistry,
1985
16.

Kim J., Jang D.O.
Synthetic Communications,
2001
17.
10.1016/j.mencom.2019.05.016_bib0085
Parvin
Lett. Org. Chem.,
2017
18.

Dar'in D., Bakulina O., Nikolskaya S., Gluzdikov I., Krasavin M.
RSC Advances,
2016
19.

Pohmakotr M., Yotapan N., Tuchinda P., Kuhakarn C., Reutrakul V.
Journal of Organic Chemistry,
2007
20.

Vara Y., Bello T., Aldaba E., Arrieta A., Pizarro J.L., Arriortua M.I., Lopez X., Cossío F.P.
Organic Letters,
2008