Abstract
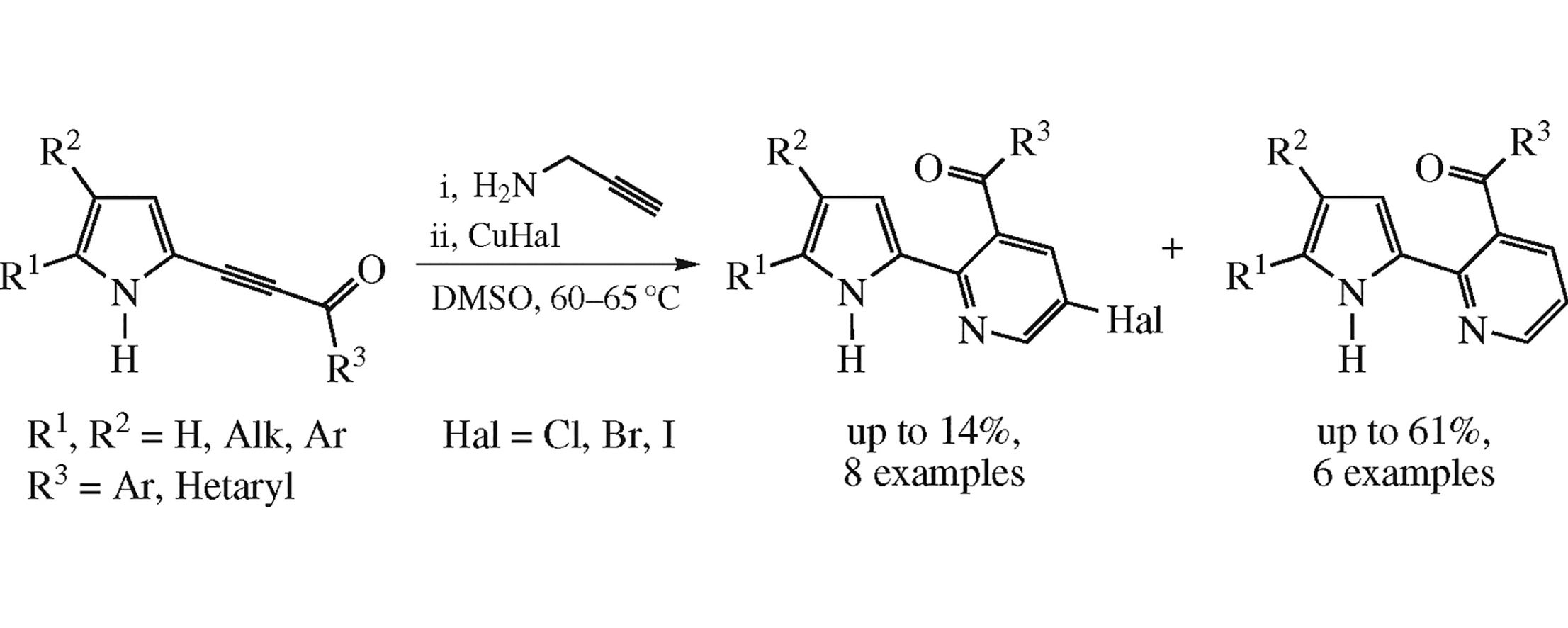
Available NH-2-acylethynylpyrroles undergo annulation/aromatization with propargylamine in the presence of 1 equiv. of CuHal (Hal=Cl, Br, I) at 60–65°C to afford 3-acyl-2-(pyrrol-2-yl)pyridines and their 5-halo analogues in 28–61 and 5–14% yields, respectively. The latter are assumed to be resulted from halohydrogenation of the intermediate Cu-pyrrolylpropargyl amino enones.
References
1.

Baumann M., Baxendale I.R., Ley S.V., Nikbin N.
Beilstein Journal of Organic Chemistry,
2011
2.

Baumann M., Baxendale I.R.
Beilstein Journal of Organic Chemistry,
2013
3.
10.1016/j.mencom.2019.05.003_bib0015
Altaf
J. Drug Des. Med. Chem.,
2015
4.

Gholap S.S.
European Journal of Medicinal Chemistry,
2016
5.

L.S. Kishbaugh T.
Current Topics in Medicinal Chemistry,
2016
6.
10.1016/j.mencom.2019.05.003_bib0030
Kaur
J. Pharm. Chem. Chem. Sci.,
2017
7.

Sessler J.L., Camiolo S., Gale P.A.
Coordination Chemistry Reviews,
2003
8.

Gale P.A.
Accounts of Chemical Research,
2006
9.

Hang X., Fleetham T., Turner E., Brooks J., Li J.
Angewandte Chemie - International Edition,
2013
10.
![Benzodithiophene based ternary copolymer containing covalently bonded pyrrolo[3,4-c]pyrrole-1,3-dione and benzothiadiazole for efficient polymer solar cells utilizing high energy sunlight](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Tamilavan V., Kim S., Agneeswari R., Lee D.Y., Cho S., Jin Y., Jeong J., Park S.H., Hyun M.H.
Organic Electronics,
2016
11.

Canjeevaram Balasubramanyam R.K., Kandjani A.E., Jones L.A., Periasamy S.R., Wong S., Narayan R., Bhargava S.K., Ippolito S.J., Basak P.
Advanced Electronic Materials,
2018
12.

Sobenina L.N., Sagitova E.F., Markova M.V., Ushakov I.A., Ivanov A.V., Trofimov B.A.
Tetrahedron Letters,
2018
13.

Trofimov B.A., Stepanova Z.V., Sobenina L.N., Mikhaleva A.I., Ushakov I.A.
Tetrahedron Letters,
2004
14.
Trofimov B.A., Stepanova Z.V., Sobenina L.N., Mikhaleva A.I., Sinegovskaya L.M., Potekhin K.A., Fedyanin I.V.
Mendeleev Communications,
2005
15.
10.1016/j.mencom.2019.05.003_bib0075
Trofimov
2009