Abstract
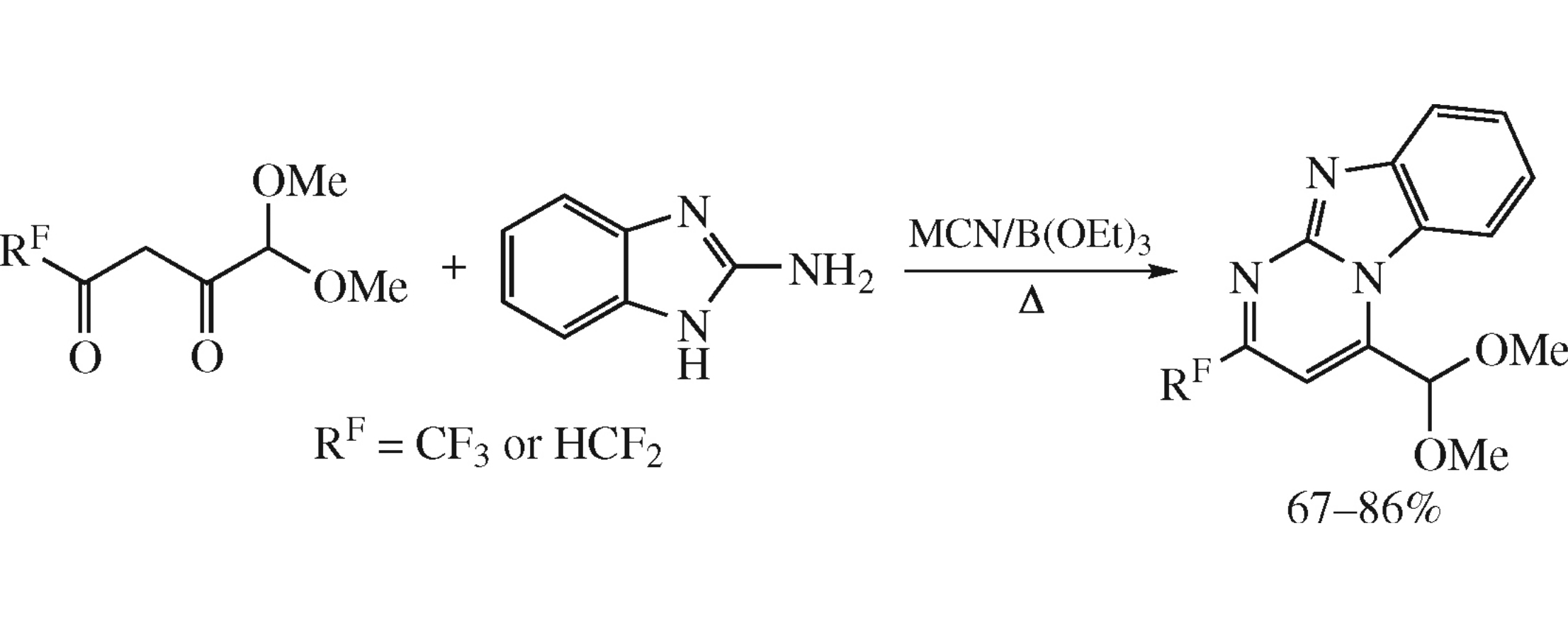
A simple and efficient synthesis of 2-RF-pyrimido[1,2-a]benzimidazole-4-carbaldehyde derivatives comprises the reaction between 3-(polyfluoroacetyl)pyruvaldehyde dimethyl acetals and 2-aminobenzimidazole. The effects of fluorinated substituents in 3-(polyfluoroacetyl)pyruvaldehyde dimethyl acetals and the reaction conditions on regiochemical outcome of this reaction have been estimated.
References
1.
10.1016/j.mencom.2019.05.002_sbref0005a
Algul
Cent. Eur. J. Chem.,
2009
2.
Shah N.M., Joshi H.S.
International Letters of Chemistry Physics and Astronomy,
2014
3.
![One-pot microwave assisted synthesis under green chemistry conditions, antioxidant screening, and cytotoxicity assessments of benzimidazole Schiff bases and pyrimido[1,2-a]benzimidazol-3(4H)-ones](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Neochoritis C.G., Zarganes-Tzitzikas T., Tsoleridis C.A., Stephanidou-Stephanatou J., Kontogiorgis C.A., Hadjipavlou-Litina D.J., Choli-Papadopoulou T.
European Journal of Medicinal Chemistry,
2011
4.
P. Pavlov, B. Winblad, Patent WO 2017168137 A1, 2017.
6.

Nowicka A., Liszkiewicz H., Nawrocka W., Wietrzyk J., Kempińska K., Dryś A.
Open Chemistry,
2014
7.
10.1016/j.mencom.2019.05.002_sbref0025b
Nawrocka
Pol. J. Chem.,
2006
8.

Filler R., Saha R.
Future Medicinal Chemistry,
2009
9.

Wang B., Wang L., Jiang B., Wang S., Wu N., Li X., Shi D.
Mini-Reviews in Medicinal Chemistry,
2017
10.
Palysaeva N.V., Boltacheva N.S., Slepukhin P.A., Pervova M.G., Filyakova V.I., Sheremetev A.B., Charushin V.N.
Mendeleev Communications,
2018
11.
10.1016/j.mencom.2019.05.002_sbref0030d
Bégué
Bioorganic and Medicinal Chemistry of Fluorine,
2008
12.
Muzalevskiy V.M., Mamedzade M.N., Chertkov V.A., Bakulev V.A., Nenajdenko V.G.
Mendeleev Communications,
2018
13.
10.1016/j.mencom.2019.05.002_sbref0030f
Bégué
Fluorine and Health. Molecular Imaging, Biomedical Materials and Pharmaceuticals,
2008
14.

Zhou Y., Wang J., Gu Z., Wang S., Zhu W., Aceña J.L., Soloshonok V.A., Izawa K., Liu H.
Chemical Reviews,
2016
15.
Dutov M.D., Kachala V.V., Ugrak B.I., Korolev V.A., Popkov S.V., Aleksanyan D.R., Rusina O.N., Aleksanyan K.G., Koshelev V.N.
Mendeleev Communications,
2018
16.
Ivanova A.E., Burgart Y.V., Saloutin V.I., Orshanskaya Y.R., Zarubaev V.V.
Mendeleev Communications,
2018
17.
![Herbizide, 2. Mitt. 2-Trifluormethylpyrimido[1,2-a]benzimidazole](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Kreutzberger A., Leger M.
Archiv der Pharmazie,
1982
18.
![Synthesis of pyrimido[1,2-a]benzimidazoles from ethyl 2-ethoxymethylidene-3-oxo-3-(polyfluoroalkyl)propionates](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Goryaeva M.V., Burgart Y.V., Saloutin V.I.
Russian Journal of Organic Chemistry,
2010
19.
![Regiodirected synthesis of polyfluoro-alkylated pyrimido[1,2-a]benzimidazoles](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Goryaeva M.V., Burgart Y.V., Saloutin V.I., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2012
20.
10.1016/j.mencom.2019.05.002_sbref0035d
Krasovsky
Synthesis,
2002
21.

Krasovsky A.L., Moiseev A.M., Nenajdenko V.G., Balenkova E.S.
Synthesis,
2002
22.
![Synthesis and Characterization of Some Novel 2-(Trifluoromethyl)pyrimido[1,2-a]benzimidazoles and Pyrimido[1,2-a]benzimidazol-2H)-ones of Biological Interest](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Echevarria A., Zanatta N., Amaral S., Esteves-Souza A., Brondani P., Flores D., Bonacorso H., Flores A., Martins M.
Synthesis,
2006
23.

Kawase M., Hirabayashi M., Saito S., Yamamoto K.
Tetrahedron Letters,
1999
24.

Bouillon J., Janousek Z., Viehe H.G., Tinant B., Declercq J.
Journal of the Chemical Society Perkin Transactions 1,
1995
25.

Chizhov D.L., Belyaev D.V., Yachevskii D.S., Rusinov G.L., Chupakhin O.N., Charushin V.N.
Journal of Fluorine Chemistry,
2017
26.

Chizhov D.L., Pervova M.G., Samorukova M.A., Khmara E.F., Filyakova V.I., Saloutin V.I., Charushin V.N.
Journal of Fluorine Chemistry,
2011
27.

Fustero S., Román R., Sanz-Cervera J.F., Simón-Fuentes A., Cuñat A.C., Villanova S., Murguía M.
Journal of Organic Chemistry,
2008