Abstract
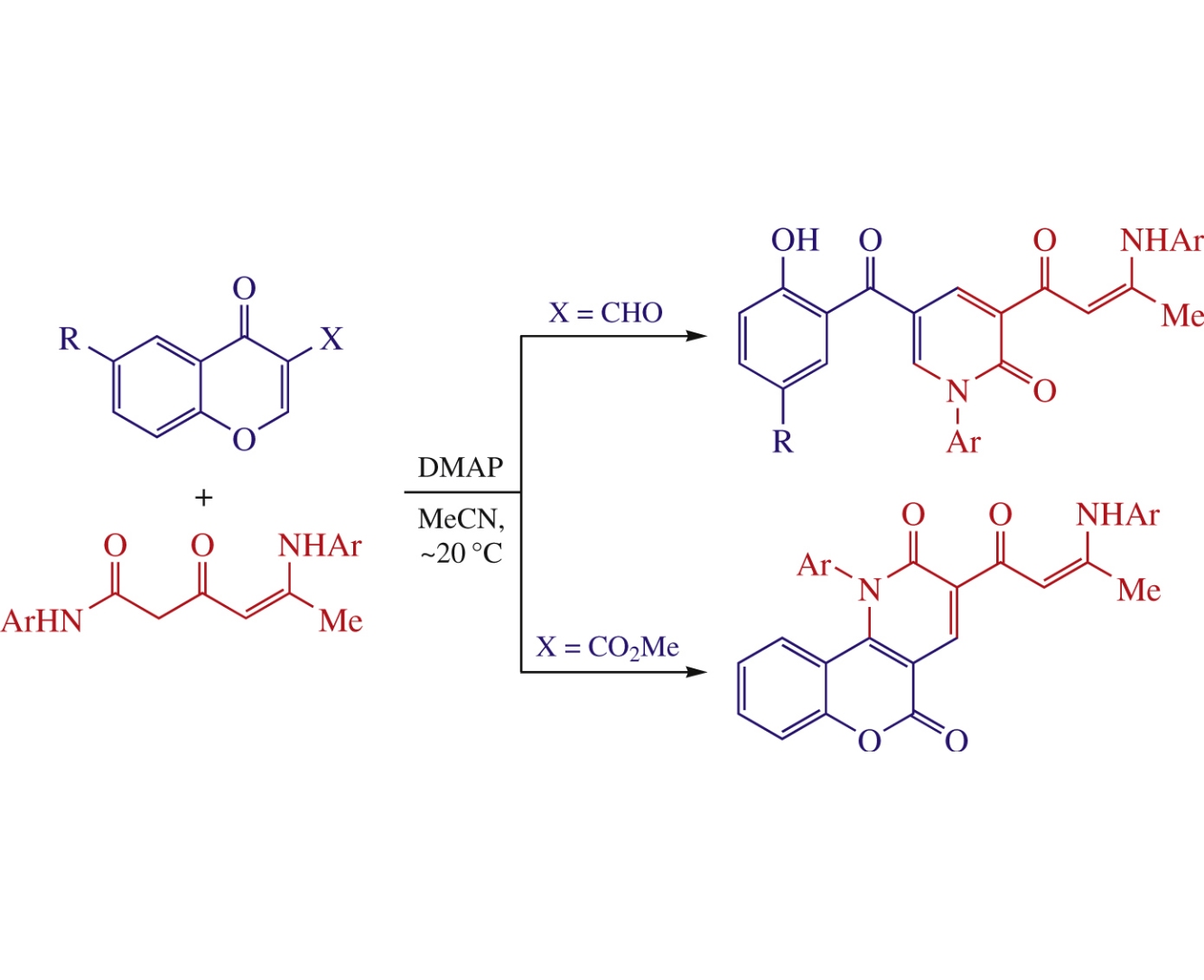
N-Aryl-5-arylamino-3-oxohex-4-enamides as 1,3-C,N-di-nucleophiles react regioselectively with 3-formyl or 3-methoxycarbonyl chromones in the presence of DMAP in MeCN to form 2-pyridones (34–43%) or chromeno[4,3-b]pyridine-2,5-diones (54–73%) bearing the amino enone moiety.
References
1.

Torres M., Gil S., Parra M.
Current Organic Chemistry,
2005
2.

Heravi M.M., Hamidi H.
Journal of the Iranian Chemical Society,
2012
3.
Fisyuk A.S., Poendaev N.V., Bundel’ Y.G.
Mendeleev Communications,
1998
4.

Mohammadi Ziarani G., Lashgari N., Moradi R., Zandiyeh M.
Heterocycles,
2018
5.

Usachev B.I.
Journal of Fluorine Chemistry,
2015
6.

Kudyakova Y.S., Bazhin D.N., Goryaeva M.V., Burgart Y.V., Saloutin V.I.
Russian Chemical Reviews,
2014
7.
Kornev M.Y., Moshkin V.S., Eltsov O.S., Sosnovskikh V.Y.
Mendeleev Communications,
2016
8.
Lyubchanskaya V.M., Mukhanova T.I., Panisheva E.K., Alekseeva L.M., Granik V.G.
Mendeleev Communications,
1995
9.

Guo Z., Zhu W., Tian H.
Chemical Communications,
2012
10.

Schreiner E., Richter F., Nerdinger S.
Topics in Heterocyclic Chemistry,
2016
11.

Reis J., Gaspar A., Milhazes N., Borges F.
Journal of Medicinal Chemistry,
2017
12.

Keri R.S., Budagumpi S., Pai R.K., Balakrishna R.G.
European Journal of Medicinal Chemistry,
2014
13.

Obydennov D.L., Khammatova L.R., Eltsov O.S., Sosnovskikh V.Y.
Organic and Biomolecular Chemistry,
2018
14.
Ali T.E., Ibrahim M.A., El-Gohary N.M., El‐Kazak A.M.
European Journal of Chemistry,
2013
15.
Ghosh C.K., Chakraborty A.
Arkivoc,
2015
16.
Obydennov D.L., Khammatova L.R., Sosnovskikh V.Y.
Mendeleev Communications,
2017
17.

Shanks B.H., Keeling P.L.
Green Chemistry,
2017
18.

Chia M., Schwartz T.J., Shanks B.H., Dumesic J.A.
Green Chemistry,
2012
19.

Schwartz T.J., Johnson R.L., Cardenas J., Okerlund A., Da Silva N.A., Schmidt-Rohr K., Dumesic J.A.
Angewandte Chemie - International Edition,
2014
20.

Schwartz T.J., O’Neill B.J., Shanks B.H., Dumesic J.A.
ACS Catalysis,
2014
21.
Obydennov D.L., El-Tantawy A.I., Sosnovskikh V.Y.
Mendeleev Communications,
2019
22.

Moreno-Mañas M., Pleixats R.
Advances in Heterocyclic Chemistry,
1992
23.

Bio-based triacetic acid lactone in the synthesis of azaheterocyclesviaa ring-opening transformation
Obydennov D.L., El-Tantawy A.I., Sosnovskikh V.Y.
New Journal of Chemistry,
2018
24.

Obydennov D.L., El-Tantawy A.I., Sosnovskikh V.Y.
Journal of Organic Chemistry,
2018
25.

Barkov A.Y., Korotaev V.Y., Kutyashev I.B., Sosnovskikh V.Y.
Tetrahedron,
2016
26.
10.1016/j.mencom.2019.03.041_sbref0050a
Ryabukhin
Synlett,
2004
27.

Sosnovskikh V.Y., Irgashev R.A., Kodess M.I.
Tetrahedron,
2008
28.

Kornev M.Y., Sosnovskikh V.Y.
Chemistry of Heterocyclic Compounds,
2016
29.

Klutchko S., Shavel J., Von Strandtmann M.
Journal of Organic Chemistry,
1974