Abstract
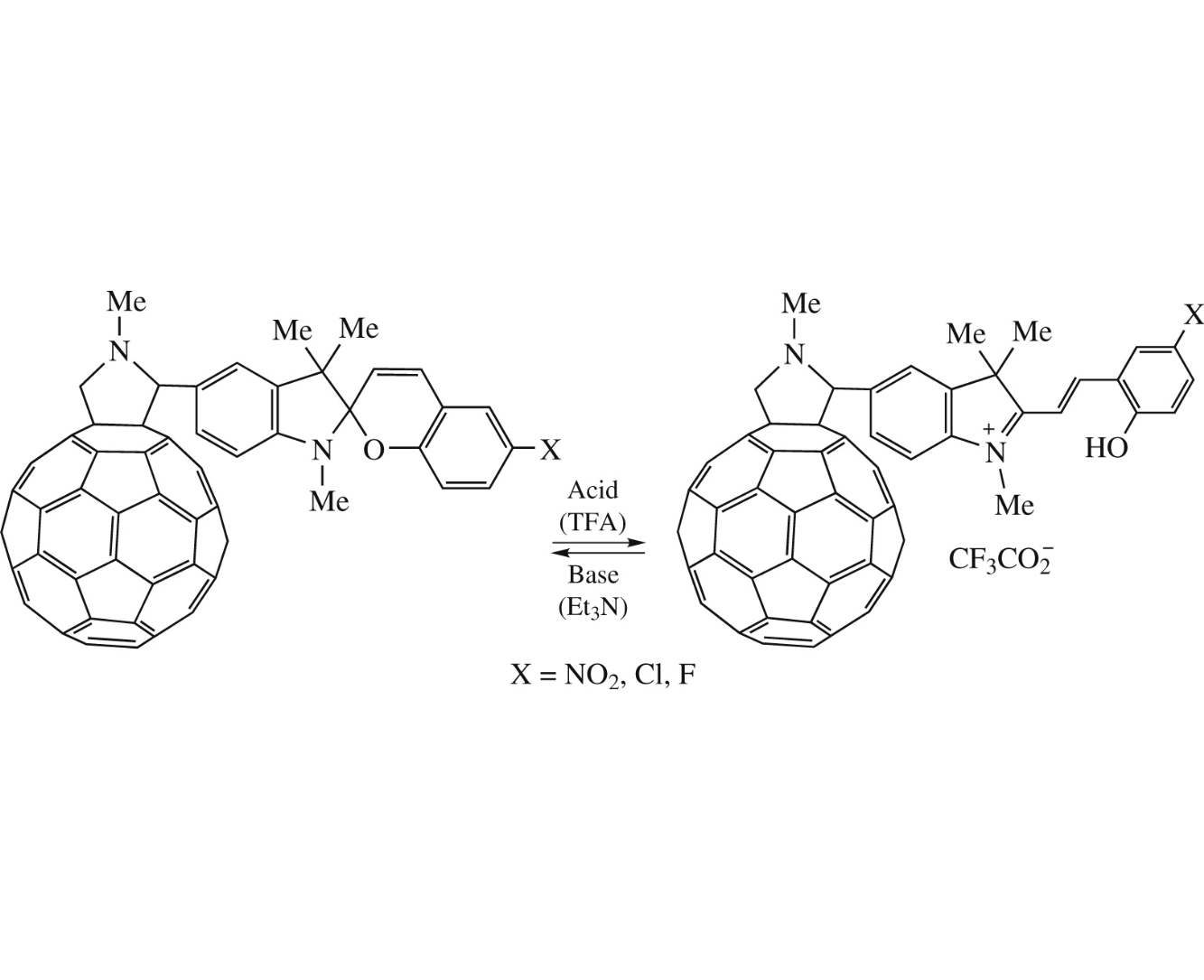
The reversible acid-base isomerization of fullerene C60 hybrid compounds with spiropyrans has been performed for the first time. It has been found that irradiation induces isomerization only for hybrid bearing NO2 group in the photochromic spiropyran part, while the treatment with trifluoroacetic acid induces isomerization for compounds containing NO2, Cl or F, i.e. irrespective of the nature of substituent in the spiropyran moiety.
References
1.

Tuktarov A.R., Khuzin A.A., Dzhemilev U.M.
Russian Chemical Reviews,
2017
2.
3.

4.

Zhi J., Baba R., Hashimoto K., Fujishima A.
Chemistry Letters,
1994
5.

Jin Fang Zhi, Baba R., Hashimoto K., Fujishima A.
Journal of Photochemistry and Photobiology A: Chemistry,
1995
6.

Song X., Zhou J., Li Y., Tang Y.
Journal of Photochemistry and Photobiology A: Chemistry,
1995
7.
![SYNTHESIS OF A NEW SPIROPYRAN BASED ON [60]FULLERENE UNIT](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Xu J., Li Y., Zhu D.
Synthetic Communications,
2002
8.

Yagi S., Nakamura S., Watanabe D., Nakazumi H.
Dyes and Pigments,
2009
9.

Darwish T.A., Evans R.A., James M., Malic N., Triani G., Hanley T.L.
Journal of the American Chemical Society,
2010
10.

Darwish T.A., Evans R.A., James M., Hanley T.L.
Chemistry - A European Journal,
2011
11.

Davis D.A., Hamilton A., Yang J., Cremar L.D., Van Gough D., Potisek S.L., Ong M.T., Braun P.V., Martínez T.J., White S.R., Moore J.S., Sottos N.R.
Nature,
2009
12.

Shiraishi Y., Itoh M., Hirai T.
Physical Chemistry Chemical Physics,
2010
13.

Wojtyk J.T., Wasey A., Xiao N., Kazmaier P.M., Hoz S., Yu C., Lemieux R.P., Buncel E.
Journal of Physical Chemistry A,
2007
14.

Darwish N., Aragonès A.C., Darwish T., Ciampi S., Díez-Pérez I.
Nano Letters,
2014
15.

Guo X., Huang L., O'Brien S., Kim P., Nuckolls C.
Journal of the American Chemical Society,
2005
16.

Del Canto E., Flavin K., Natali M., Perova T., Giordani S.
Carbon,
2010
17.

Perry A., Green S.J., Horsell D.W., Hornett S.M., Wood M.E.
Tetrahedron,
2015
18.

Li Y., Duan Y., Zheng J., Li J., Zhao W., Yang S., Yang R.
Analytical Chemistry,
2013
19.

Joo P., Kim B.J., Jeon E.K., Cho J.H., Kim B.
Chemical Communications,
2012
20.

Jang A., Jeon E.K., Kang D., Kim G., Kim B., Kang D.J., Shin H.S.
ACS Nano,
2012
21.

Nahain A., Lee J., Jeong J.H., Park S.Y.
Biomacromolecules,
2013
22.

Sharker S.M., Jeong C.J., Kim S.M., Lee J., Jeong J.H., In I., Lee H., Park S.Y.
Chemistry - An Asian Journal,
2014
23.

Tuktarov A.R., Khuzin A.A., Tulyabaev A.R., Venidictova O.V., Valova T.M., Barachevsky V.A., Khalilov L.M., Dzhemilev U.M.
RSC Advances,
2016
24.

Light-controlled molecular switches based on bistable spirocyclic organic and coordination compounds
Minkin V.I.
Russian Chemical Reviews,
2013
25.
![Inheritance of Photochromic Properties of Nitro-Substituted and Halogenated Spiropyrans Containing the Pyrrolidino[60]fullerene](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Pomogaev V.A., Barachevsky V.A., Tuktarov A.R., Avramov P.V., Artyukhov V.Y.
Journal of Physical Chemistry A,
2018
26.
Tuktarov A.R., Khuzin A.A., Khalilov L.M., Tulyabaev A.R., Akhmetov A.R., Dzhemilev U.M.
Mendeleev Communications,
2015
27.

Tuktarov A.R., Khuzin A.A., Akhmetov A.R., Barachevsky V.A., Venidiktova O.V., Dzhemilev U.M.
Tetrahedron Letters,
2015
28.
Tuktarov A.R., Khuzin A.A., Akhmetov A.R., Khalilov L.M., Tulyabaev A.R., Barachevskii V.A., Venediktova O.V., Dzhemilev U.M.
Mendeleev Communications,
2016
29.

Tuktarov A.R., Akhmetov A.R., Khuzin A.A., Venidiktova O.V., Barachevsky V.A., Dzhemilev U.M.
Russian Journal of Organic Chemistry,
2017